Q: Answer all questions: 1. Consider cyclobutadiene molecule (structure is shown below) has a point…
A:
Q: Assign priorities to the groups in each set
A: The rules that must be followed while prioritizing the substituent group for R-S configurations are-…
Q: Find out all the energies and the complete molecular orbitals for 1,3 butadiene system.
A:
Q: What is the energy order of MOs (list lowest energy level first) in an MO diagram of the species…
A: Molecular orbital : When atomic orbital overlaps to form new set of same number of orbitals are…
Q: Antibonding MOs always have at least one node. Can abonding MO have a node? If so, draw an example.
A: When two atomic orbitals overlap, they form two molecular orbitals, of which one is bonding and one…
Q: sO2 belongs to which point group? C2v .A O C2h .BO Doh.cO C2.DO
A:
Q: What is the point group of SbF, ? C1 O C3h C3 O C3v
A: The given molecule is SbF3.
Q: (b) Work out the point group of- (i) CIF;; (ü) IF5.
A:
Q: Reduce the following reducible representation: C3v E 2C3 3σv 6 0 -2
A: The following question is related to group theory. point table is given of point group C3v in the…
Q: Use as a basis the valence pz orbitals on each atom in BF3 toUse as a basis the valence pz orbitals…
A:
Q: Draw a pz and a dz2 orbital and assign their point groups
A: Px is symmetrical and dz2 is symmetrical
Q: Stability by Hyperconjugation: A
A: Stability of alkenes can be expained by hydperconjugation Displacement alpha C-H sigma electrons…
Q: MOT diagram for N2
A: The electronic configuration of N- atom can be written asN (7) : 1s2 2s2 2p3N (7) : 1s2 2s2 2p3
Q: Which of the following molecule(s) belong to CooV point group? 1. CO 2. H2 3. HCI 4. 02 * O 1 and 3…
A: We have to ask which of the molecule belong to C∞V point Group 1. CO 2. H2 3. HCl 4. O2…
Q: Irreducible representation for cH2cl2
A: The point group is C2v. Therefore, we need to use the correct character table for the transformable…
Q: Determine the point group of the following orbital:
A: The given orbital is f0 orbital
Q: What is the energy order of MOs (list lowest energy level first) in an MO diagram of the species B2-…
A: The answer to the following question is-
Q: Bro 1) The orbital potential energies for the 4s and 4p of Br are -18.65 ev and -12.49ev,…
A:
Q: Produce an MO diagram for N2 and determine the overlap integral for the HOMO and LUMO orbitals using…
A: solution- MO diagram for N2 is given below;
Q: NO3- (d3h) MO diagram.. needed in irreducible form
A:
Q: diato anion selenium bromide (SeBr-) |(A) What is the basis set for the molecular orbital of SeBr-?…
A: Explanation Molecular energy diagram Diatomic molecule with two non-identical atoms are called as…
Q: determine the symmetry adapted linear combinations of borane BH3
A:
Q: Which of the following would be expected to have a dipole moment of zero on the basis of symmetry?…
A: The molecule that is expected to have a dipole moment of zero on the basis of symmetry has to be…
Q: Draw an MO diagram for the pi-bonding in 1,3-butadiene (C2h symmetry). Draw a picture of each MO…
A: M.O CAN BE OBTAINED FROM NOMOF PARTICIPATING ORBITALS,HERE TOTAL 4 ATOM PARTICIPATED SO 4 M.O WILL…
Q: The correct MO description of NO+ is Group of answer choices BO of 3 and paramagnetic BO of 2 and…
A: The bond order indicates how many chemical bonds exist between two atoms. The formula for bond order…
Q: 5) Examine if CH4 molecule possesses an S4 or not. Show what are the associated symmetry operations…
A: Ch4 belongs to the TD group and posses an S4 improper rotation.
Q: Based on the symmetry considerations used for homonucelar diatomic molecules, consider the C2H4 a.…
A: When symmetry of two orbitals matches, then bonding orbitals are formed. When symmetry of two…
Q: The types of symmetry operations for AsClz are: Select one: A. E, C3, C2 oy B. E, C3, C2 S2 C. E,…
A: We have to predict the symmetry elements in given compound.
Q: (b) Work out the point group of (i) CIF;; (ü) IF5.
A: To determine point group of a molecule , we first calculate the symmetry elements present in the 3D…
Q: Determine the point group for each compölünd
A:
Q: Hydrogen sulfide (H2S) is a toxic gas, often emitted into the atmosphere as an industrial pollutant.…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Which of the following would be expected to have a dipole moment of zero on the basis of symmetry?…
A: All symmetrical molecules have diople moment equal to zero and are considered as non-polar whereas…
Q: (a) Complete the following table: Molecule/lon Geometry of molecule Key symmetry elements Point…
A: 1) B(F)(Cl)(Br) The given compound has 3 bond pairs and 0 lone pairs on the central atom. Thus it is…
Q: For each molecule (a)–(f), indicate how many differentelectron-domain geometries are consistent with…
A: The table below describes the electron and molecular geometries,
Q: Using appropriate diagram(s), show which of the symmetry operation(s) does the [SO4]2- ion have: (i)…
A:
Q: The bond in H–CI can be explained by the overlap between an s orbital from hydrogen and a p orbital…
A: (a)
Q: Use group theory to derive the MO diagram for the pi system in 1,3-butadiene. Sketch the shapes of…
A:
Q: Predict the valence electron configuration and the totalbond order for the molecule I2. Will I2 be…
A: Valance shell electrons in I2 molecule:14. For this, we have to write the electronic configuration…
Q: Sketch the MO diagram for 1,3 budadiene. Identify the HOMO and LUMO along with all symmetric MO’s,…
A: MO diagrams: These diagram gives information regarding the linear combination of atomic orbitals and…
Q: (a) Draw the orbital picture for :C=0: showing the explicit overlap of the contributing AOs. (b) How…
A: Molecular orbitals are formed by the Linear Combination of Atomic Orbitals (LCAO). Number of…
Q: The bond in Cl2 can be explained by the end-on overlap between two p AOs, as shown inthis diagram.…
A: The bond in Cl2 can be explained by the end-on overlap between two p AOs, as shown in this diagram.…
Q: 1. Where are the symmetrical classifcations on the molecules? 2. What is the point group for each…
A: Following are the symmetrical classification, point group and chirality of the given compounds.
Q: Using the appropriate MO diagram determine the bond orders for: a) F22+ b) F2-
A: By using the electronic configuration, σ2s2 σ*2s2 σ2pz2 π2px2 π2py2 π*2px1 π *2py1 , the MO diagram…
Q: StectureD is more stable. Deaw the Newman projections for these two Chairs?
A:
Step by step
Solved in 2 steps with 1 images

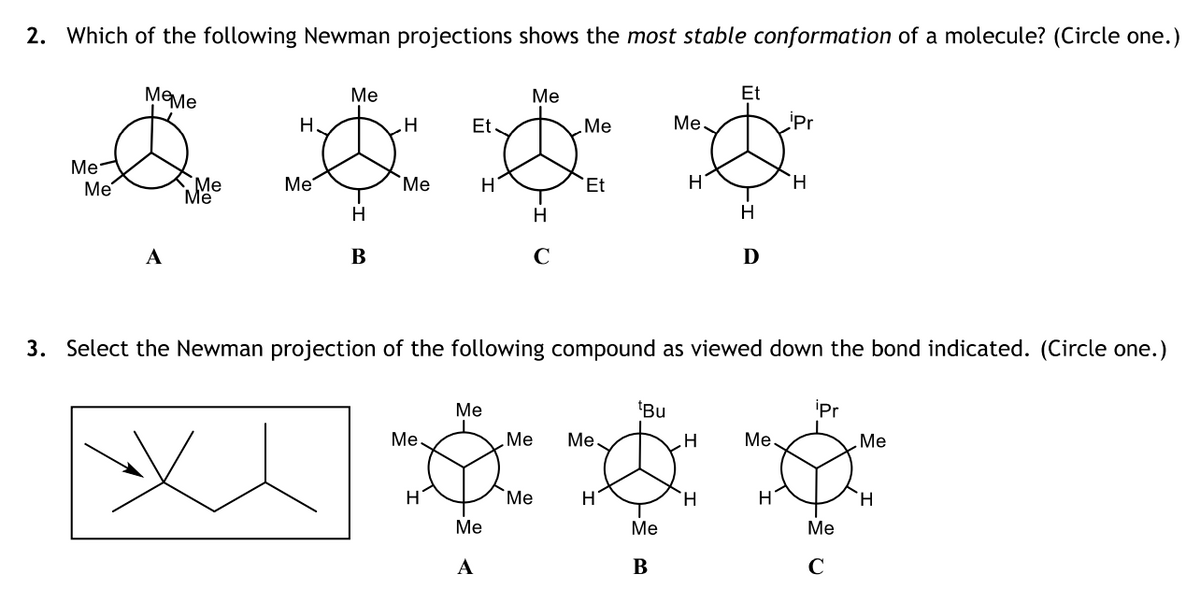
- Solve correctly please. Which of the following Newman projections is the least stable?Draw an exact 3-D representation of the molecule from the model.Consider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.
- Consider the Newman projection below. a. Draw a full Lewis structure of this molecule with R1=Me,R2=Et , and R3=iPr . b. Given the sizes of these R groups (R3R2R1) , does the Newman projection above show thelowest potential energy conformation of this bond? If not, draw a Newman projectionshowing the lowest P.E. conformation (sighting down this same bond). c. To draw a Newman projection in the lowest P.E. conformation, the following rule of thumbusually applies: Place the largest group on the front carbon anti to the largest group on theback carbon. Is your answer to the previous question consistent with this rule of thumb?Build a model of methylcyclohexane, and use the model to complete the following Newmanprojections of methylcyclohexane in the chair conformation: a. When the methyl group is in an axial or equatorial (circle one) position, the molecule is inits lowest potential energy conformation. b. Label one Newman projection above anti and the other gauche to describe the relationshipbetween the methyl group and C3 of the ring. c. In general, which is a lower PE conformation, anti or gauche? d. Explain how your answer to b and c provide an explanation for why it is more favorable fora large group to be in an equatorial than an axial position.Then draw the most stable and least stable Newman projection conformation from the C4-C5 bond in the molecule above
- assign R and S for fisher projection and show assigned priorities as well as bond lines. Thank you in advance!is left or right more stable? Please provide an explanation as to why. Thank you!Please look at the picture as it is related on my question. When do we decide that it is CH3 or H3C that we put while we are drawing the Newman projection?

