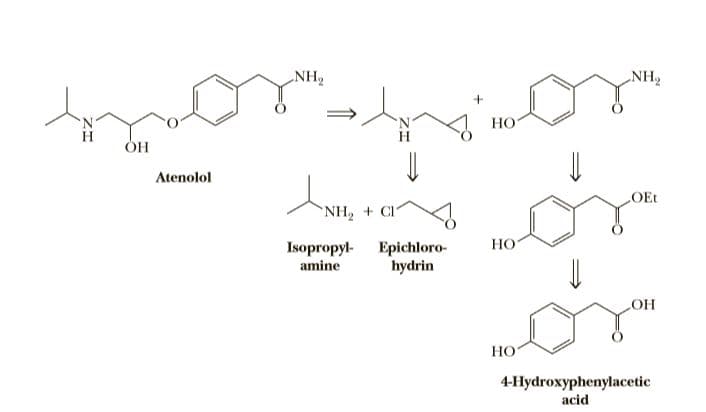
Side effects of propranolol include disturbances of the central nervous system (CNS), such as fatigue, sleep disturbances (including insomnia and nightmares), and depression. Pharmaceutical companies wondered if this drug could be redesigned to eliminate or at least reduce these side effects. Propranolol, it was reasoned, enters the CNS by passive diffusion because of the lipidlike character of its naphthalene ring. The challenge, then, was to design a more hydrophilic drug that does not cross the blood-brain barrier but still retains a b-adrenergic antagonist property. A product of this research is atenolol, a potent b-adrenergic blocker that is hydrophilic enough that it crosses the blood-brain barrier to only a very limited extent. Atenolol is now one of the most widely used b-blockers.
Q. Is atenolol chiral? If so, which of the possible stereoisomers are formed in this synthesis?
Step by step
Solved in 3 steps with 3 images






