Nitrous acid, HNO₂, can be made by reacting N₂O3 with water as depicted below in a balanced chemical equation: N₂O3 + H₂O → 2HNO₂ If 20.0 grams of N₂O3 were reacted with an excess of water and only 19.5 grams of HNO₂ actually formed, what would be the percent yield of this reaction? Molar Mass of N₂O3 = 76.01 g/mole Molar Mass HNO₂ = 47.013 g/mole Note: Give your answer in percentage, not fraction, form (e.g if it were 25.4%, enter 25.4, NOT 0.254) PLEASE CLEARLY SHOW YOUR WORK FOR THIS AND SUBMIT IT
Nitrous acid, HNO₂, can be made by reacting N₂O3 with water as depicted below in a balanced chemical equation: N₂O3 + H₂O → 2HNO₂ If 20.0 grams of N₂O3 were reacted with an excess of water and only 19.5 grams of HNO₂ actually formed, what would be the percent yield of this reaction? Molar Mass of N₂O3 = 76.01 g/mole Molar Mass HNO₂ = 47.013 g/mole Note: Give your answer in percentage, not fraction, form (e.g if it were 25.4%, enter 25.4, NOT 0.254) PLEASE CLEARLY SHOW YOUR WORK FOR THIS AND SUBMIT IT
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter3: Stoichiometry
Section: Chapter Questions
Problem 7RQ: Consider the hypothetical reaction between A2 and AB pictured below. What is the balanced equation?...
Related questions
Question
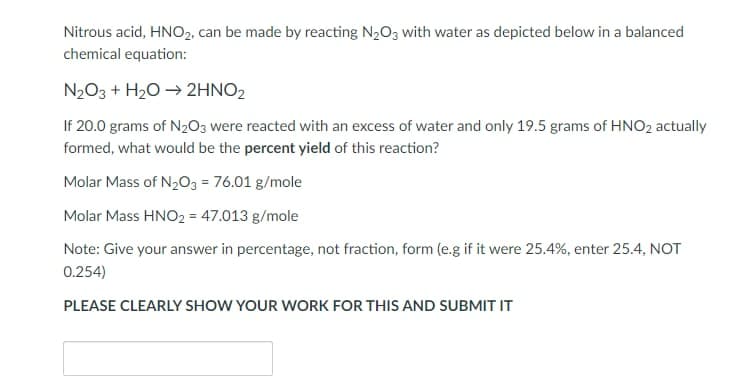
Transcribed Image Text:Nitrous acid, HNO₂, can be made by reacting N₂O3 with water as depicted below in a balanced
chemical equation:
N₂O3 + H₂O → 2HNO2
If 20.0 grams of N₂O3 were reacted with an excess of water and only 19.5 grams of HNO₂ actually
formed, what would be the percent yield of this reaction?
Molar Mass of N₂O3 = 76.01 g/mole
Molar Mass HNO2 = 47.013 g/mole
Note: Give your answer in percentage, not fraction, form (e.g if it were 25.4%, enter 25.4, NOT
0.254)
PLEASE CLEARLY SHOW YOUR WORK FOR THIS AND SUBMIT IT
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning