Q: 7) Write the IUPAC/common name of the following compounds: A) HCOO-Li B) CH3CH₂CH₂COO NH4+ 8) Write…
A:
Q: The amount of product that can be produced depends only on the amount of the limiting reactant…
A:
Q: It is observed that 8.7x10^-4 mol of SrF2 will dissolve in 1.0 L of water. Use this data to…
A:
Q: 4HCL+02= 2H20 +2CL2 If 02 is increased, what would be the effect on the equilibrium? Select the…
A:
Q: A solution is prepared that is initially 0.49M in pyridine (C5H₂N), a weak base, and 0.40M in…
A:
Q: Black Forest Biologicals, a biotech startup, has a promising Alzheimer's drug candidate Compound…
A:
Q: At a potential of -1.0 V (versus SCE), carbon tetrachloride in methanol is reduced to chloroform at…
A: Given information: At - 1.0 V (versus SCE) 2 CC14 + 2 H+ + 2e- + 2 Hg(l) ----> 2 CHC13 +…
Q: Q14: If you are given 3 samples of polypropylene (pp), one isotactic, one syndiotactic and one a…
A:
Q: On the sketch indicate a. the signs of the electrodes Mg(s) + 2Ag+ (aq) → Mg+2 (aq) + 2Ag(s)
A: Recall the given redox reaction, Mg s + 2 Ag+ aq → Mg+2 aq + 2 Ag…
Q: Give the enol formation(s) of the following dicarbonyl compound
A: Here we are required to draw the enol form of given compound
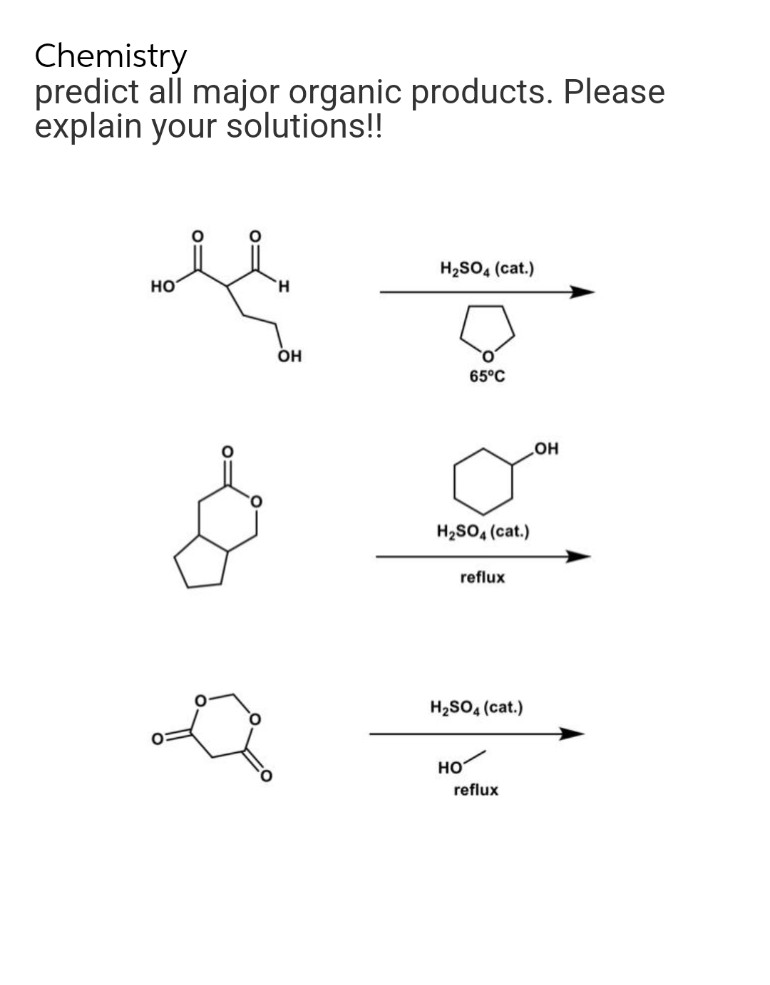
Q: Q3/ Synthesis the following compounds using any organic or inorganic reagents from ethylacetoacetate…
A:
Q: A certain compound, X. with an initial concentration of 0.79 M undergoes a zeroth order reaction…
A: The half life of the reaction is time taken by the reaction to complete half of the reactant.
Q: please answer, d, e, f, g, h, i
A: Esters are alkyl derivatives of carboxylic acids. IUPAC name of ester is alkyl alkanoate as it…
Q: Materials engineers often create new alloys in an effort to improve the properties of an existing…
A: Given, Formula of the alloy Zn0.838Cd0.162O
Q: ----) A vessel has content 0.2 of C₂H6 & 0.6 Oxygen as at 2000K, find the time to consumption all…
A:
Q: Given the balanced redox reaction is as follows: 2Cr(OH)3(s) + 6ClO-(aq) → 2CrO42-(aq) + 3Cl2(g) +…
A:
Q: (SHOW CALCULATIONS) Find q when 50.0 g of water (c = 4.184 J/g-K) is heated from 40.0°C to 100.0°C.
A:
Q: Choices 0.20 mol/L H₂SO3(aq) 0.10 mol/L H₂SO4 0.20 mol/L H₂SO3(aq) Stronger Acid 0.10 mol/L H₂SO4
A: The acid which have higher H+ ions in given amount of water will have more stronger acid as compare…
Q: Click the "draw structure" button to launch the drawing utility. Draw the structure of a molecule…
A:
Q: A nitrile compound is prepared from cyclopentene. The following is an analysis of the disconnection…
A: The synthesis steps indicate the reaction of alkene i.e. cyclopentene with peracetic acid to form…
Q: 54. How and why do chemical reaction rates depend on temperature?
A: The rate of the reaction depends on the temperature. For more than one question, we are asked to…
Q: Calculate the enthalpy change for the following chemical reaction: Tip: make sure the reaction is…
A:
Q: Deduce the correct maximum wave length (max the following compound
A:
Q: 3. The oxo-ions of p-block elements are usually aggressive oxidizing agents while thos elements are…
A: We must choose an oxo-ion which matcjes with the charge and valency of phosphate ion, so that it can…
Q: Br ... Yuilll o CO₂H Br ||||| c CO₂H
A: The given two compounds are not constitutional isomer constitutional isomer and can be explained by…
Q: Raul law expressions and parameter description
A: Raoults law states that for any volatile substance the partial pressure is exerted by vapour or gas…
Q: Which type of photon is emitted when an electron in a hydrogen atom drops from the n = 2 to the n =…
A:
Q: Suggest a reasonable Geranylgeranylpyrophosphate. GPP معة biosynthesis of diterpene OPP…
A: By ionizing, the diphosphate functionality, followed by a 2,3 double bond isomerization, a tertiary…
Q: 1. A. Complete/Completing analysis B. Trace analysis C. Proximate analysis D. Ultimate analysis 2.…
A: Few types of chemical analysis are given. In proximate analysis,technique used to analyse compound…
Q: Solid phosphorus (P)and chlorine (C1₂) gas react to form solid phosphorus pentachloride (PC15).…
A: Given Number of moles of P=13.0 mol Number of moles of Cl2= 7.0 mol Number of moles of PCl5= ?
Q: Aniline is a weak base. The pH of a 0.010 M aqueous solution of aniline is 8.32. What is the percent…
A: The percentage of dissociation of aniline can be calculated as follows
Q: 1. Calculate the normality of potassium permanganate (MW: 158.034 g/mol) if 0.2142 g of sodium…
A: Dear student, since the given question is multiple question hence I solved first question according…
Q: Determine the volume (in mL) of water that needs to be added to 25.0 mL of a 0.250 M NaBr to produce…
A:
Q: If the bicarbonate ion (HCO3-) concentration in a sample of blood is 0.135 M, determine the carbonic…
A:
Q: An equilibrium mixture contains 0.550 mol of each of the products (carbon dioxide and hydrogen gas)…
A:
Q: how to draw chair conformations as consituinal isomers?
A: Constitutional isomers are structural isomers the compounds that share same molecular formula but…
Q: Predict the major organic product of the reaction of 2-methyl-1-propene or 2-methylprop-1-ene and…
A: Here we have to write the major product formed in the following given reaction.
Q: Indicate which of the following Ti3+ compounds exhibits the wavelength absorption shortest wave in…
A:
Q: consider 0.10 M aqueous solutions of each of the following substances: HlO, NH4ClO4, HClO4, KlO,…
A: Given : Ka for HIO = 2.3 x 10-11 Ka for HClO4 = 107 Kb for NH3 = 1.8 x 10-5
Q: An analytical chemist is titrating 92.3 mL of a 0.6600M solution of ethylamine (C₂H5NH₂) with a…
A:
Q: Give the IUPAC name for each compound. OH a. CH3CH(CH₂)4CH3 (select) OH L (CH3CH₂)2CHCHCH₂CH3…
A: Here we have to write IUPAC name of the following given compounds.
Q: Be sure to answer all parts. Give the structure corresponding to each IUPAC name. a. 1,3-heptadiene…
A:
Q: The weedkiller propanil used in the rice fields is an amide. Write the analysis and synthesis this…
A: Given in following question strecture of a propanilus3d in rice fields is na amide . Write the…
Q: Research mass spectrometry and spectrophotometers. How has the use of mass spectrometry advanced our…
A: Mass spectroscopy has an impeccable importance in the real world. Few are listed as follows-
Q: Q9: Polystyrene is soluble in benzene while poly (vinyl alcohol) is soluble in water. how will a…
A:
Q: Propyl-3-ethyl pentanoate WHAT IS THE 1.Common name 2.Condensed formula 3. Type of Compound and…
A: The condensed formula represents the symbols of atoms, listed in order as they are shown in the…
Q: Draw intermediates 1 and 2 given the following chemical reaction
A: Here we are required to find thr intermediate 1 and 2 for the given chemical reaction.
Q: How many grams of a solution that is 5.20 obtain 18.0 % sucrose by mass are needed to g of sucrose
A: The mass of solute in gram present in 100 g solution is known as the mass percent concentration of a…
Q: pls answer d, e , f
A: See below
Q: Determine the volume (in mL) of water that needs to be added to 25.0 mL of a 0.250 M NaBr to produce…
A:
Step by step
Solved in 4 steps with 4 images

- Provide complete solutions.Determine the mole fraction of CH3OH in a solution prepared by dissolving 36 g of alcohol in 1000g of H₂0.A. 55.55molesB. 0.02molesC. 1.125molesD. None of the aboveEthanol is metabolized in most people at a constant rate of about 120mg per hour per kg body weight, regardless of its concentration. If a 70kg person were at twice the legal limit (160mg/100ml), how long would it take for their blood alcohol level to fall below the legal limit?
- A. FERMENTATION OF CANE SUGAR1. In a 500 mL Florence flask, place 50 gms of brown cane sugar and 1 gram ofdisodium phosphate.2. Dissolve 1 teaspoon of yeast in 15 mL of water. Add this to the sugar in the flasktogether with 150 mL water. Stir until the sugar is dissolved.3. Close the flask with a rubber stopper provided with a delivery tube so that theevolved gas will bubble through a solution of clear limewater.4. HAVE THE SET – UP CHECKED BY YOUR INSTRUCTOR.5. Keep the set – up inside the locker for 5 – 6 days at room temperature.Fermentation Set – up: ________________________________Write the equation for the hydrolysis of cane sugar _____________________________Write the equation for the fermentation of the simple sugars to alcohol __________6. At the end of the fermentation period, what is formed in the flask containinglimewater? _________________________________________________________________Write the equation for its formation ____________________________________________ C.…What is the chemistry behind fish fermentation process? Provide references and elaborateA. Calculate the concentration of sugar in this solution using the volume percent equation. For the purpose of this exercise, it can be assumed that the weight of the drink mix is 0.00 g. The volume of the solution is 3 and 1/3 cups. Calculate and record the volume percent (g/mL). (Note that 1 cup = 236 mL). Volume %=Volume of Solute / Volume of Solution ×100% I used 1/2 cup of granulated sugar and 1/2 tsp of the drink mix, and 3 cups of water. B. Calculate the concentration of each diluted solution using the equation below and the volumes in Table. C1V1=C2V2 Where: C1=concentration of stock solution V1=volume of stock solution C2=concentration of diluted solution V2=volume of diluted solution (This is all the information that I have)
- 40.875 g of glucose (MM = 180.16 g/mol) are dissolved in 6.0 moles of water (18.02 g/mol). Calculate the mole fraction of water. Be sure to show set up of equation cancellationsCHEMISTRY (Please write the complete solution legibly. No long explanation needed. Answer in 2 decimal places. Box the final answer.) Predict whether each of the following compounds is soluble in water:a. (NH4)2SO4 b. NaCN c. K3PO4d. Cu(C2H3O2)2 e. BaSO4 f. FeCl3What is the boiling point of a 0.50m aqueous solution of aluminum nitrate? (The boiling point elevation constant for water is 0.512C kg solvent/ mol solute) Answer choices are: a.) 100.51 b.) 101.02 c.)102.05 d.)106.60 I know the answer is B but i would like to see step by step how to solve this problem
- What is the concentration of O2 in freshwater? Air temperature= 295.65 K. Air pressure = 1 bar(about = to 1atm). Henry's constant, from the table= 1.3 x 10-3 mol/ (kg x bar). PO2 can be calculated from the fact that oxygen is .21 mole fraction of air. Hint- The partial pressure of a gas in a mix of gasses is found by multiplying the total pressure by the mole fraction of that gas. Find the concentration in g/kg from the answer you just got.1. What is the importance of giving hyperbaric oxygen during treatment of poisoning associated with certain fumes and gases? Provide at least 3 poisoning where hyperbaric oxygen is required. 2. Discuss the importance of maintaining the normal blood pressure during poison management. What substances/poisons can increase blood pressure? Decrease blood pressure? Give 5 examples for each.Chemical wastes (Aqueous) should be stored in wash bottles. True or False? Please explain.

