number of valence electron: Periodic Table Jacksonville University -X / mod /flcn/view.php?id=9635823 HW 3 A Activities and Due Dates HW3 Cive Uph Resources Group the electronic configurations of neutral elements in sets according to those you would expect to show similari chemical properties. Set A Set B Answer Bank 1s22s22p 1s2s22p 3s23p64s23d104p Is2s22p 3s23p 1s2s22p6 3s23p Determine the chemical symbols for the neutral elements corresponding to the electronic configurations. Use proper formatting; letter case matters. 1s22s22p3 1s22s22p3s23p64s23d104p 1s22s22p 3s23p9: 1s22s22p3s23p terms of use about us help privacy policy contact us careers eN P hp
number of valence electron: Periodic Table Jacksonville University -X / mod /flcn/view.php?id=9635823 HW 3 A Activities and Due Dates HW3 Cive Uph Resources Group the electronic configurations of neutral elements in sets according to those you would expect to show similari chemical properties. Set A Set B Answer Bank 1s22s22p 1s2s22p 3s23p64s23d104p Is2s22p 3s23p 1s2s22p6 3s23p Determine the chemical symbols for the neutral elements corresponding to the electronic configurations. Use proper formatting; letter case matters. 1s22s22p3 1s22s22p3s23p64s23d104p 1s22s22p 3s23p9: 1s22s22p3s23p terms of use about us help privacy policy contact us careers eN P hp
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter11: Atomic Theory :the Quantum Model Of The Atom
Section: Chapter Questions
Problem 103E
Related questions
Question

Transcribed Image Text:number of valence electron:
Periodic Table
Jacksonville University -X
/ mod /flcn/view.php?id=9635823
HW 3
A
Activities and Due Dates
HW3
Cive Uph
Resources
Group the electronic configurations of neutral elements in sets according to those you would expect to show similari
chemical properties.
Set A
Set B
Answer Bank
1s22s22p
1s2s22p 3s23p64s23d104p
Is2s22p 3s23p
1s2s22p6 3s23p
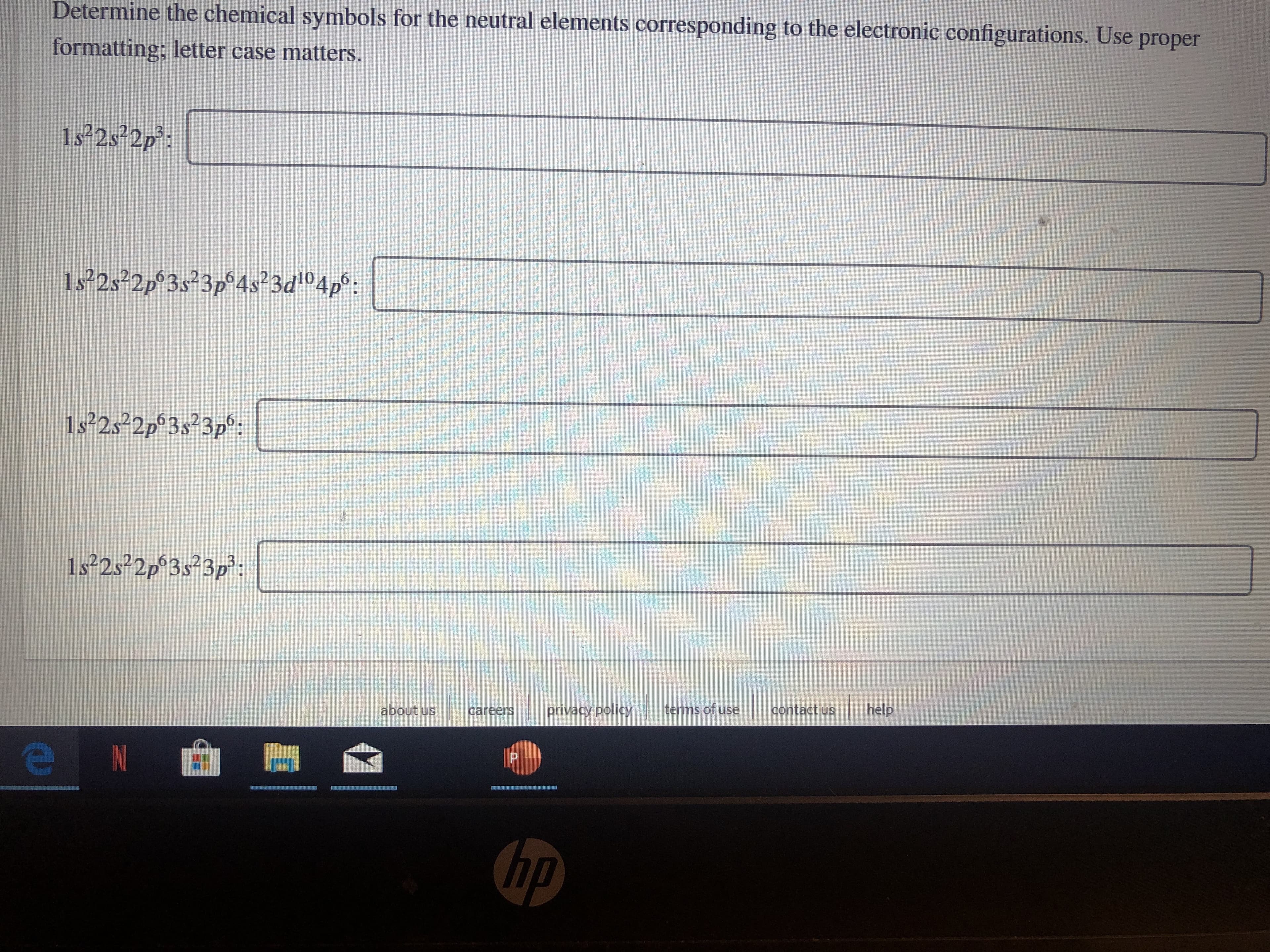
Transcribed Image Text:Determine the chemical symbols for the neutral elements corresponding to the electronic configurations. Use proper
formatting; letter case matters.
1s22s22p3
1s22s22p3s23p64s23d104p
1s22s22p 3s23p9:
1s22s22p3s23p
terms of use
about us
help
privacy policy
contact us
careers
eN
P
hp
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning