Q: What is the hybridization of Kr in KrF3* ?
A:
Q: Write a balanced chemical equation based on the following description: sulfur trioxide gas and water…
A: Write balanced chemical reaction for the given ?
Q: Unshared, or lone, electron pairs play an Important role in determining the chemical and physical…
A: Here, we have to find the number of unshared pairs on the designated atoms of the given compounds.
Q: 2. A student named a compound as 6-methyl-6-ethylhept-1-en-5-one. Even though one can draw a correct…
A: Please find your solution below : According to IUPAC naming conventions, any compound is named…
Q: (f) a Newman projection of the following molecule looking down the C2-C3 bond in the anti-…
A: Newman projection visualizes the conformation of a chemical bond from front to back, with the front…
Q: Match the following aqueous solutions with the appropriate letter from the column on the right.…
A: Question is asked about the freezing point of the compound. Some compound are given , we have to…
Q: Using the pk, table, estimate pK, values for the most acidic group on the compounds below, and draw…
A:
Q: The boiling point of water is about 200°C higher than one would predict from the boiling points of…
A: The boiling point of any aqueous solution or liquid depends on the intermolecular force between…
Q: What is the molar mass of 27.8 mg of a compound that is made up of 3.80 x 1020 molecules? 44.1 g/mol…
A:
Q: OCN+OCI→CO¾¯ + N₂ + Cl¯ Match the following for the above reaction. Drag and drop options on the…
A: The given redox reaction is: OCN- + OCl- → CO32- + N2 + Cl-
Q: All machines reduce the amount of force required to complete a task by doing what? O a b Oc Od…
A: Energy can be defined as the property that can be transferred to a physical system.
Q: Identify the functional groups in the following molecules. (Use names from the table below. List…
A:
Q: po For the reaction 2H1 (aq) + H₂O₂ (aq) → 12 (aq) + 2H₂O The kinetics of the reaction can be…
A:
Q: A calibration curve of ZnCl2 conductivity (y-axis) vs concentration (mass percent, x-axis) is made.…
A: In the linear regression plot, the X-axis represents the mass percent of ZnCl2. The Y-axis…
Q: Let the rate of the reaction 2 NH3N2 + 3 H2 be expressed as -Δ[NH3]/Δt. Which of the following is…
A: 2 NH3 -------> N2 + 3 H2 Rate is expressed in terms of -Δ[NH3]/Δt.
Q: All of the following alkyl bromides react by SN2 substitution when treated with sodium cyanide.…
A: Inversion of configuration takes place during SN2 reaction if the C atom at which the substitution…
Q: What precipitate is most likely formed from a solution containing: Ba2+, Na+, OH, CI" and CO3²- O…
A:
Q: Which is the most stable free radical? A B C D E
A:
Q: For the standardization procedure, 0.2023 g of anhydrous sodium carbonate was dissolved in 100 mt…
A: Concentration of HCl can be found out from the moles of sodium carbonate. Moles of borate ion can be…
Q: What is the stereochemical relationship between the following pairs of molecules? Possible answers:…
A: Given : structure of molecules
Q: Which reacts the most rapidly in a nucleophilic substiution reaction? a. 2-Iodo-2-methylpropane…
A: The substitution of an incoming nucleophile in an organic compound is called nucleophile…
Q: A zinc pot (coefficient of linear expansion : ) is filled with mercury at 100 °C, then having a 10.0…
A:
Q: Which of the following represents a neutralization reaction? O 2 NaOH(aq) + CuCl2(aq) → 2 NaCl(aq) +…
A: There are different types of chemical reactions neutralization reaction is one of them.…
Q: Which two Fischer projections represent a pair of enantiomers? CH3 CH3 CH3 HOH H- H- CI но- C₂H5 A O…
A:
Q: How many stereoisomers (including itself) does the following molecule have? Br que OH 04 016 02 03…
A:
Q: The Ka of a weak acid is 3 x 10^-12 What is the pKa of this acid?
A: Ka which is dissociation constant of acid is given 3*10^-12 Pka = ?
Q: A chemist puts 0.085 mol of N and 0.038 mol of O₂ in a 1.00L container. What are the concentrations…
A:
Q: αcarboxyl and α-amine are 2.2 and 9.1 perspectively. Calculate isoelectric point of tyrosine. i want…
A:
Q: 7. Give the IUPAC names for the following compounds: Br I II OH CI III Br
A:
Q: calculate the theoretical amount (in mL) of 0.100 M HCl required to precipitate Pb (MM=207.2) as…
A:
Q: When there is Treatment of Compound E with sodium amide and then methyl iodide it does not produce…
A: NaNH2 or NH2- is a strong base that reacts with very weak acid to form NH3(aq)
Q: The half-life of Palladium-100 is 4 days. After 20 days a sample of Palladium 100 has been reduced…
A:
Q: D D Question 42 The ability of potassium nitrate to mix with water is most likely due to: O Lewis…
A: #Q.42: Potassium nitrate, KNO3 is an ionic compound dissolved in water to give the cation, K+(aq),…
Q: Provide the correct systematic name for the compound shown here. H3C NH₂ CH3
A:
Q: Steel wool consists mostly of iron. The combustion of iron produces iron(III) oxide, Fe2O3. Write…
A:
Q: 2. Using the solubility table, predict whether the following compounds will be soluble or insoluble…
A:
Q: Arrange the following compound in the order of increasing acidity (least acidic first) I)…
A: Acidic character of the given organic compounds can be explained by the presence of replaceable…
Q: DELLISE 3433.73 Copyright © 1994 1H 13 Proton NHA 180 160 Carbon 13 NMR 18 951 140 HAVEHUMBERS 120…
A: Find the structure of organic compound using ir and nmr spectra
Q: to HO From Malonic Acid
A: Malonic acid is an example of active methylene compound.
Q: Arrange the following carbocations in order of their decreasing stability (most stable first). va A…
A: Carbocation is a electron deficient species.
Q: III. I. II. N i 1.CH2 CHCCH3 2. H₂O/H₂0 CH₂CH₂CCH3 O CH₂CH₂CCH3 QH CCH3 CH=CH₂ IV. V. i CH₂CH₂CCH3…
A:
Q: Which of the following statements is true for non-ideal gasses? O The higher the temperature, the…
A: In this type of question you have to first neglect the false statement , Please find the below…
Q: both for species that act as both donors and acceptors neither for species that act neither as…
A: The donor in hydrogen bond is a strong electronegative atom for example N, O, or F which is…
Q: When a solute is added to a solvent, the resulting solutions's boiling point freezing point and its…
A:
Q: Which of the following represents the correct Cahn-Ingold-Prelog priority ranking of C5H11…
A: Ans: d. A<D<B<C Here all the groups are carbon -hydrogen containing ,hence 3° carbon will…
Q: Which of the following molecular formulas is reasonable for a stable compound? a. C7H10NCI O b.…
A:
Q: Acid-catalyzed dehydration of 3,3-dimethyl-2-butanol gives three alkenes: 2,3-dimethyl-2-butene,…
A: -> Reactant -> 3,3-dimethyl-2-butanol -> Products-> 2,3-dimethyl-2-butene ,…
Q: Please use 3 significant figures or 3 decimal places. What is the pH of a buffer is prepared, 1.00…
A: Aqueous Solution consists of a mixture of a weak acid and its conjugate base or weak base and its…
Q: Use the information provided to determine AH°rxn for the following reaction: AH°f (kJ/mol) CH4(g) +4…
A:
Q: What is the percent recovery if you started with 9.3 mL of benzaldehyde and ended with 4.1 g of…
A:
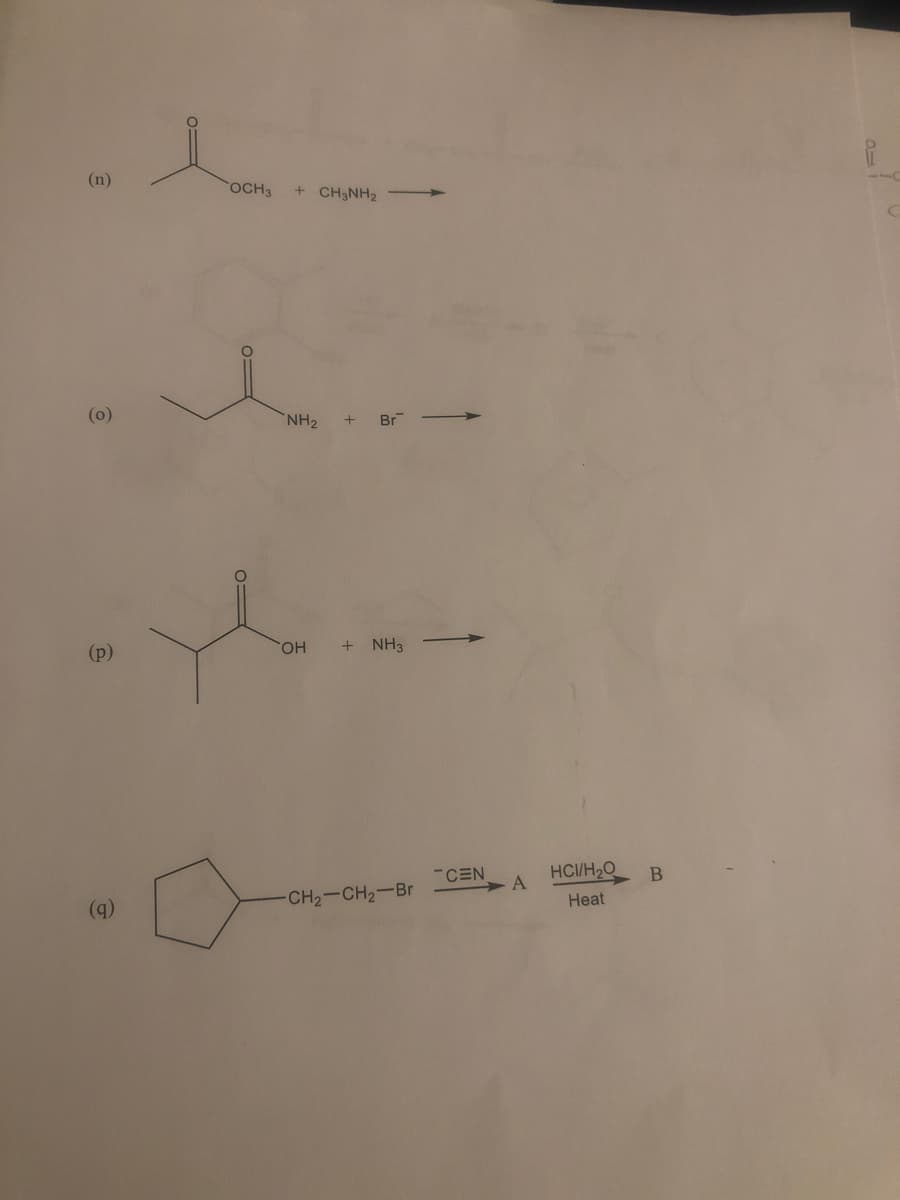
Step by step
Solved in 3 steps with 1 images

- Rank the following groups in order of decreasing priority. −F, −NH2, −CH3, −OHProvide a systematic name (IN SMALL LETTERS except for E/Z and R/S, NO SPACES) for each of the given compounds. Show all work.Arrange each set in order of decreasing priority using Cahn-Ingold-Prelog priority rules (1 for highest priority). d) _____-Cl, _____-OH, _____-H, _____-NH2 e) _____–CO2CH3, _____-CO2H, _____-OH, _____-OCH3
- With link additivity values estimate DH ° 298 for CH3CH2OH (g) → CH3OCH3 (g).Please help me I don't understand this, its a simple mecahnism but im lost.I put in the answer for compound A, (dimethyl‐lambda4‐sulfanylidene)cyclopropane, and the product 9‐oxadispiro[2.0.44.13]nonane, in the boxes, but it says it's incorrect. What am I not getting?Thank you for your help!



