of 11 Name the compound. (2S,3S)-2-bromo-3-isopropoxy-4-methylpentane (2S,3R)-2-bromo-3-isopropoxy-4-methylpentane (2R,3S)-2-bromo-3-isopropoxy-4-methylpentane (2R 3R)-2-bromo-3-isopropoxy-4-methylpentane O Rotate Z Q Zoom In Q Zoom Out A Label Atoms
of 11 Name the compound. (2S,3S)-2-bromo-3-isopropoxy-4-methylpentane (2S,3R)-2-bromo-3-isopropoxy-4-methylpentane (2R,3S)-2-bromo-3-isopropoxy-4-methylpentane (2R 3R)-2-bromo-3-isopropoxy-4-methylpentane O Rotate Z Q Zoom In Q Zoom Out A Label Atoms
Chapter22: Bulk Electrolysis: Electrogravimetry And Coulometry
Section: Chapter Questions
Problem 22.6QAP
Related questions
Question

Transcribed Image Text:help me please
Click on the 3D molecular structure and drag to rotate it, or use the controls provided. Analyse the structure and sume the
compound. You might find it helpful to use your model kit to make a model based on each same, then compare it with the 3D
structure to determine configurations
..
line ihe cos
utaܛܕ 4 ܫܥ Q!?X3>3hxnwp
ܕ 4 ܢܡ2323©
9
BANKE
O
، ، ܪ [jo܀
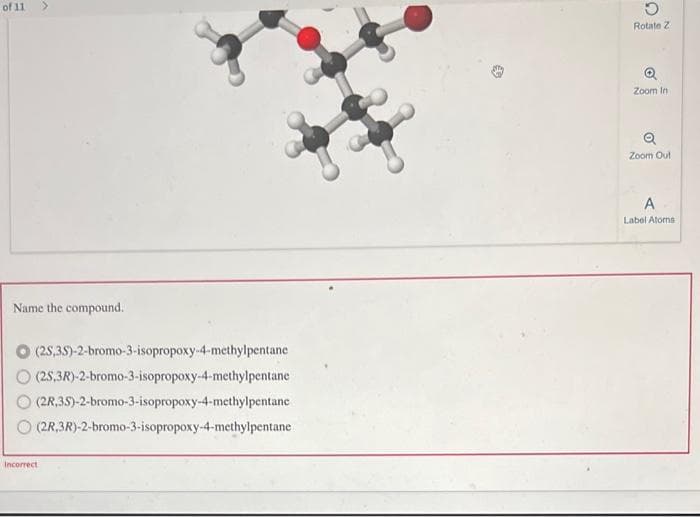
Transcribed Image Text:of 11
Name the compound.
(2S,3S)-2-bromo-3-isopropoxy-4-methylpentane
(2S,3R)-2-bromo-3-isopropoxy-4-methylpentane
(2R,3S)-2-bromo-3-isopropoxy-4-methylpentane
(2R,3R)-2-bromo-3-isopropoxy-4-methylpentane
Incorrect
Rotate Z
Zoom In
Q
Zoom Out
A
Label Atoms
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

