Q: Consider the amino acid and its pKa values:Lysine: pKa1= 2.18 pKa2= 8.95, pKaR=10.79 a. Calculate th...
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3.
Q: Can you help with the step by step I'm all over the place. I submitted the front page earlier but i ...
A: Given, Symbol of metal used = Al (Aluminium) Mass of empty test tube = 40.90 g Mass of test tube and...
Q: Match each structure to its mass spectrum.
A: a.
Q: An aqueous potassium chloride solution is prepared by dissolving 76.8g of KCl in 435.0g of water. Wh...
A: Given: Mass of KCl= 76.8g Mass of water= 435.0g Freezing point depression constant of water= 1.86°C/...
Q: Prepare each compound from cyclopentanol. More than one step may be needed.
A: Organic synthesis processes are used to prepare different organic compounds by organic reactions suc...
Q: The phase diagram for CO2 has a triple point at -56.6°C and 5.19 atm, and a critical point at 31.0 °...
A: Phase diagram: A graphical representation of a substance under different condition of pressure and t...
Q: After studying the concept of real gases, Zainab was perplexed to know the following:(a) Why the ter...
A: For real gas
Q: A sample of krypton gas at a pressure of 934 mm Hg and a temperature of 56 °C, occupies a volume of ...
A:
Q: Chemistry Question
A: Part B Addition of hydrogen to a carbon carbon double bond is called hydrogenation. Nickel is most ...
Q: 22. What type(s) of intermolecular forces are exhibited by the molecule below? VDW = van der Waals; ...
A: Dipole-Dipole interactions are interactions in which partially positively charged group of molecule ...
Q: List H1 to H5 as their pKa values from lowest to highest. Explain the reason.
A: From H1 to H5 as their pKa values from lowest to highest have to be listed and the reason has to be ...
Q: g6
A: (a) Calculate the activation energy: 1 J = 0.001 kJ
Q: 6 7. a) Natural gas when allowed to expand from a volume of 5.2 L to a volume of 9.5 L. While the pr...
A: i) The work has been done if the gas expands freely without any external hindrance with proper expla...
Q: Methanol, CH3OH(g), is a possible automobile fuel. The alcohol produces energy in a combustion react...
A:
Q: The number of unshared pairs at atom a is ___The number of unshared pairs at atom b is ___The number...
A: The electronic configuration of Oxygen (O) = 1s2 2s2 2p4 ….(1), so atom (a) have additional one e...
Q: What is the mole fraction of 1.2g of NaHCo3 dissolved in 225g of water?
A: Let the number of moles of sodium bicarbonate and water be x and y respectively. The molar mass of s...
Q: Identify each compound from its molecular formula and its 1H NMR spectrum: C9H12
A: The double bond equivalent for the compound is equal to 1. This suggests the presence of carbonyl gr...
Q: If 9.8g water is used in electrolysis, what is the percent yield if 5.6g of oxygen was collected aft...
A:
Q: Perform each of the following operations, using your calculator where possible:(a) Write the number ...
A: Since we answer only upto 3 sub-parts, we will answer first three. Please resubmit the question by m...
Q: Devise a synthesis of (E)-tetradec-11-enal, a sex pheromone of the spruce budworm, a pest that destr...
A:
Q: The reaction 2NOBr(g)→2NO(g)+Br2(g)is a second-order reaction with a rate constant of 0.80 M−1s−1 at...
A: Since we know that the relationship between the concentration of reactant i.e [A] and time for a sec...
Q: why is n/V squared in the van der Waals equation?
A: Vander Waal’s equation It is corrected form ideal gas equation and is applicable to real gases. Thi...
Q: Determine a molecular formula, e.g. , from the line structure below.Specify elements in the followin...
A:
Q: Give the IUPAC name for each compound.
A: Since we answer only 3 sub-parts so we will answer first 3. Please resubmit the question mentioning ...
Q: A chemist prepares a solution of calcium bromide (CaBr2) by measuring out 3.4 x 102 μmol of calcium ...
A: Since concentration in mmol/L we have to find And concentration = moles of solute / volume of solut...
Q: For which of the following substances is the least energy required to convert one mole of the solid ...
A: The formula to calculate lattice energy is
Q: The water supply for a midwestern city contains the followingimpurities: coarse sand, finely divided...
A: Here are the ways so we can remove these impurities:
Q: Iron has a density of 7.86 g/cm3 and crystallizes in a body-centered cubic structure at room tempera...
A: In body centered cubic unit cell, the number of atom (Z) is 2
Q: Chemistry Question
A: Grignard reagent- Usually it is written as RMgX means alkyl magnesium halide. This is important rea...
Q: A compound decomposes by a first-order process. If 17.0% of the compound decomposes in 60 minutes, t...
A: First order reaction is a reaction that depends only one reactant concentration. Integrated form of ...
Q: Dehydration of 1,2,2-trimethylcyclohexanol with H2SO4 affords 1-tertbutylcyclopentene as a minor pro...
A: a.
Q: Draw a stepwise mechanism for the polymerization of isoprene to guttapercha using (CH3)3CO–OC(CH3)3 ...
A: (CH3)3CO – OC(CH3)3 is basically used to initiate the free radical mechanism reaction as it converts...
Q: Draw the structure of the polymer formed by step-growth polymerization of each monomer or pair of mo...
A: Step-growth polymerization: It is the type of polymerization in which bi or multinational group form...
Q: A first-order reaction has a rate constant of 0.33 min−1. It takes ________ min for the reactant con...
A:
Q: For the following reaction profile, indicate E Reaction coordinate a) the positions of reactants and...
A: a) The initial component of the curve shows the reactant and final shows products hence the positio...
Q: What is the answer to this question? Given answer is acid; 1.86 ml of alkali
A: NOTE : it will be 17.36 mL of 1.075 acid added not 17.36 g Sol : Since the mass of K2CO3 and Na2CO3 ...
Q: In the electrolysis of water, how long will it take to produce 1.000 × 102 L of H2 at STP (273 K and...
A: According to ideal gas law, PV = nRT where P = pressure V = volume n = moles R = gas constant = 0.08...
Q: Draw a second resonance form for the structure shown below.
A: Resonance is a method of representation of bonds in various molecules or ions. Resonance structure i...
Q: Calculate the pressure exerted by 0.5900 mole of in a 1.1000-L container at 25.0°C. (The gas consta...
A: 1.
Q: 12-20d Could you let me know the right answer? Is the starting compound pentanol and is it a Swern o...
A: No the starting compound is not pentanol Since it has OH group not directly attached to the ring h...
Q: Please answer E and F:
A: Part E Let the oxidation number of sulfur be x. the oxidation state of Na and O are +1 and -2 respec...
Q: The mass of an empty 10ml graduated cylinder is 8.6g. What is the mass of a graduated cylinder with ...
A: The given data is: Mass of an empty 10 mL graduated cylinder is 8.6 g Volume of water = 10 mL Densit...
Q: To determine the concentration of citric acid, you will need to titrate this solution with 0.100 M N...
A: Relation between molarity, volume for reaction between two solutions- M1V1=M2V2 Where, M1=Molarit...
Q: Explain the reactivity and orientation effects observed in each heterocycle.a. Pyridine is less reac...
A: Aromatic compounds generally shows electrophilic substitution reactions. In electrophilic substituti...
Q: Consider the following equilibrium: FeO (3) + C0 (2) Fe (9) + CO2 (2) If CO2 is removed from the equ...
A: The given equilibrium reaction is as follows, The Le Chatelier’s principle says that when a system ...
Q: What is the product of the following reaction? NABH4 H. CH3OH
A: The given reaction is a reduction of aldehyde by sodium borohydride ( NaBH4) in presence of methano...
Q: 2
A:
Q: Please answer the question:
A: Faradays first law of electrolysis – During electrolysis of any material, the amount of material pr...
Q: Can I get a step by step walkthrough with this problem? I'm still learning this concept.
A: SOLUTION: Step 1: Since HClO2 has a low Ka value, it is a weak acid. Weak acids partially dissociate...
Q: 8. (a) The enthalpy of the fusion of metal Cesium is 13.5 kJ/mol at the melting point of 28.5 oC. Ca...
A: Since the entropy change for a process happening at a constant temperature of melting point is given...
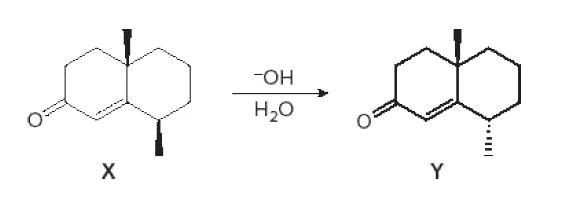
Treatment of α,β-unsaturated carbonyl compound X with base forms the diastereomer Y. Write a stepwise mechanism for this reaction. Explain why one stereogenic center changes configuration but the other does not.
Step by step
Solved in 2 steps with 1 images

- To my understanding that you are supposed to answer 3 questions per asking . Write clear and mine form as questions thank youpart, g,h,iOn a balance mass an amount of potassium hydrogen phthalate (KHP) needed to neutralize approximately 12 ml of 0.1 M NaOH(aq), and completely transfer the KHP to a clean 125-ml Erlenmeyer flask. Record the mass of the KHP at least to The thousandth decimal place. please answere



