Q: 3A(g) + B(g) + 2C(g) → D(g) + 2E(g) Rate of formation of D= k [A] [B]? Doubling the concentration of...
A:
Q: 1. AL + HНе 30 15P + on -> 63 2Cu + H 2 on + 63 Zn. 30 65 3. 30Ca 65 29SC 2.
A: Since you have asked multiparts questions, we will solve the first three sub parts questions for you...
Q: using magnesium hydroxide as an example, provide definitions for solubility product and saturation r...
A: The solubility product constant is the equilibrium constant for the dissolution of a solid into an a...
Q: Synthesis Here are the targets. Starting compounds will be provided. 0. NHCH, starting Compound: sta...
A:
Q: Calculations and Analysis: 1. If vinegar is 5% acetic acid and the density of vinegar is 1.05 g/mL, ...
A: Since you have posted multiple questions, as per our company guidelines we are supposed to answer on...
Q: In an aqueous chloride solution cobalt(II) exists in equilibrium with the complex ion CoCl4². co2+(a...
A: Given reaction is : CoCl42- (aq) <-----------> Co2+ (aq) + 4Cl- (aq)
Q: Suggest a synthesis for the molecule shown below. Only starting materials containing six (6) carbons...
A: We can get the final product by taking terminal alkyne as a starting material and we can expect alke...
Q: 1. For the following elements, determine the four quantum numbers Element 1 mi ms V Gd Ni
A:
Q: What volume of 0.73 M NaCl solution contains 145.9 grams of NaCI?
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: equation, use the coefficient in the box to determine what color to Use TOF 1 = Red 2 = Pink 3 = Ora...
A:
Q: ) Calculate the pH, [H+] and (OH-] of a solution with a pOH of 5.36 e) What is the pH of a solution ...
A: Since you have posted question with multiple subparts as per guidelines we can answer three per sess...
Q: Which of the following is a buffer solution? blood O vingear water O saliva
A: Solution of weak acid and it's conjugate base is called buffer solution.
Q: What is the average molar mass of a gas containing the following by mass % composition: CO2 = 10 O...
A:
Q: Aqueous sulfuric acid (H, SO reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium sul...
A:
Q: Complete the following table. Round each of your answers to 3 significant digits. energy content whe...
A: The conversion of quantity from one unit to another unit is known as unit conversion. During the uni...
Q: Water is a substance that exists in all the three states of matter. What do you call when a substanc...
A: Physical change - A change in the appearance, shape and size of a substance without a change in its...
Q: 3 Write the ionization equilibrium for lysine and calculate its pI.
A: Lysine is an essential amino acid which has characteristics amine group as well as carboxylic acid ...
Q: . How are gas Pressure and Volume related? Give an example using math and do a calculation to prove ...
A: Ideal gases are those gases which obey ideal gas equation. The gases that do not obey ideal gas equa...
Q: In an experiment to determine the content of acetylsalicylic acid in an aspirin tablet, the result s...
A: Answer: Given- the calculated mass of acetylsalicylic acid was much higher than the indicated amount...
Q: A student ran the following reaction in the laboratory at 70 K: H2(g) + I2(g) 2HI(g) When she introd...
A:
Q: Wine goes bad soon after opening because the ethanol (CH,CH,OH) dissolved in it reacts with oxygen (...
A: Given moles:: moles of oxygen = 1.1 mol moles of water = ?
Q: A(n) 8.40-mol sample of carbon monoxide was stored in a 30.0-L container at 52.0°C. What is the pres...
A: Given : n = 8.40 mole V = 30.0 L T = 52 .0°c T= 52.0 + 273.15 k T = 325.15 k R = 0.082 atm...
Q: For numbers 33 - 38, consider the following: Element G is from Group 17 (Halogen Group) and element ...
A:
Q: A gas mixture has a total pressure of 0.51 atm and consists of He and Ne. If the partial pressure of...
A: Ptotal = 0.51 P He = 0.23
Q: Ammonium perchlorate (NH,CIO,) is a powerful solid rocket fuel, used in the Space Shuttle boosters. ...
A: Ammonium perchlorate decomposes into nitrogen, chlorine, oxygen, and water. The equation for the bal...
Q: If I have 4.0 moles of a gas at a pressure of 5.6 atm and a volume of 12 liters, what is the tempera...
A: Ideal gas equation: The temperature, pressure, volume, and mole of the gas are related by the ideal ...
Q: Calculate the pH of the following acid or base: 1. 3.6 x 10-7 M HNO3 2. 2.0 x 10-4 M NaOH
A:
Q: A sample of water from the North Sea was analysed by ion-chromatography and the sulfate concentratio...
A:
Q: Draw the structure of a named di-saccharide molecule, giving its chemical formula.
A: A disaccharide is a carbohydrate made up of two monosaccharides by glycosidic bond . Example : sucro...
Q: Predict the products of the reaction below. That is, complete the right-hand side of the chemical eq...
A:
Q: Ammonium perchlorate (NH,clo,) is the solid rocket fuel used by the U.S. Space Shuttle. It reacts wi...
A: Given-> Weight of NH4ClO4 = 8.0 gm
Q: What is the pH of a 0.85 M H2SO4? Show your ICE table and calculations. Ka1 for H2SO4 is a very...
A: The sulfuric acid completely disassociates in the solution, hence we can assert that the concentrati...
Q: Solve the following with the correct chemical notation and significant figures. 1.24x10^2 + 2.5x10^3...
A:
Q: 12. P + }H 28 14Si si + 13. 14N + 17 80 14. 23Pu + H • on + 944
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for...
Q: failure Mode why bellows corrosion.causes of corrosion bellows and their solutions
A: Corrosion is defined as 'an irreversible interfacial reaction of a material with its environment whi...
Q: 8. Determine the enthalpy of the following reaction given the data: 2N2() + 502g) → 2N½Osg) H22) + 1...
A: Given-> 2N2(g) + 5O2(g) ---> 2N2O5(g)
Q: What mass (in grams) of magnesium oxide can be produced from igniting 1.5 g of Mg in Oxygen? (MM of ...
A: Mole can be defined as a standard unit for measuring large quantities of very small entities such as...
Q: The equilibrium constant, Kc, for the following reaction is 1.29x10-2 at 6000 K. cocI2(g) co(g) + Cl...
A:
Q: 2CH2CI2(g) =CH4(g) + CCI4(g) If 0.552 moles of CH2CI2(g), 0.514 moles of CH4, and 0.444 moles of CCI...
A: 2CH2Cl2(g) --> CH4(g) + CCl4(g) At equilibrium- Moles of CH2Cl2 = 0.552 moles Moles of CH4 = 0.51...
Q: Calculate the pressure, volume, or temperature of a gas under certain conditions of change. Solve th...
A: Given, Initially Volume of a gas (V1) = 1.7 L moles of a gas (n1) = 0.2 mol Later: (after adding 0....
Q: 4.91 mL of NaOH solution was required to reach the endpoint when titrated against 0.1005 g of potass...
A: Here 4.91 ml of NaOH solution was required to react with 0.1005 g of KHP dissolved in 50ml water.We ...
Q: The rate constant for the breakdown of an isotope of iodine is first order and has a half-life of 32...
A:
Q: 4..Provide an alternate synthesis to synthesize each of the products 1-phenyl-1-propanol and 2-pheny...
A: Alcohols we can prepare from alkene by acid catalysed hydration. Using grignard reagent from carbon...
Q: pressure
A:
Q: Write them on a separate sheet of paper. An aerosol disinfectant can has a pressure of 5.0 atm at 25...
A:
Q: Pure ozone decomposes slowly to oxygen, 203(g)302(g).203(g)-302(g). Determine the order and rate con...
A: Solution . Given that Pure ozone decomposes slowly to oxygen . Dete...
Q: Dinitrogen tetraoxide is a colorless gas at room temperature. It can dissociate into nitrogen dioxid...
A: First we calculate each value using the concentration values in table. Then check all the values to...
Q: For each of the following, identify the formulas of the major entities present when the substance is...
A: Water(H2O) is a polar molecule capable of forming hydrogen bonding. Hence polar molecules, and pola...
Q: Select the molecules that can hydrogen bond with water Multiple answers: Multiple answers are accept...
A:
Q: For a 0.0005M dilute aqueous solution of alcium sulfate at 0oC, calculate the following: a.) ionic ...
A:
Give the IUPAC name.
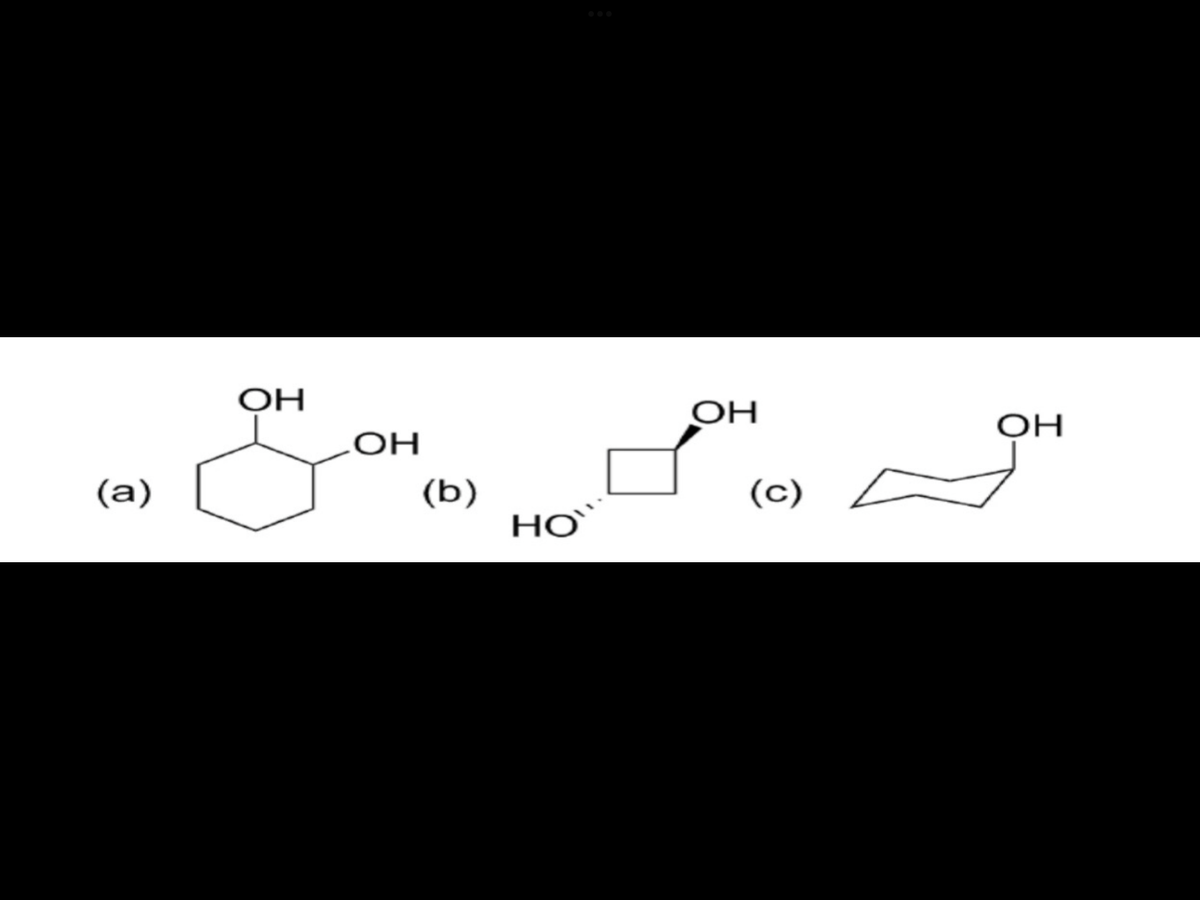
Step by step
Solved in 2 steps with 1 images


