Q: Phosgene (COCI,) reacts with formaldehyde in the following reaction to produce dichloromethane and c...
A: A question based on kinetics of reaction in solutions that is to be accomplished.
Q: formula NISO,•XH2O? 4
A:
Q: 2. Consider the electrochemical cell where one of the half cells is comprised of a Pt electrode in a...
A:
Q: Calculate the pH of a solution that is 0.200 M in NH3 and 0.300 M in NH4Cl. Calculate the pH change ...
A: Kb = 1.75 x 10-5 Concentration of NH3 = 0.200M Concentration of NH4Cl = 0.300M Volume of buffer solu...
Q: reaction compound concentration expected change in concentration vessel CH,CH, NH, 0.79 M f increase...
A: A question based on equilibrium concept that is to be accomplished.
Q: O CHEMICAL REACTIONS Interconverting number of atoms and mass of compound Calculate the number of ma...
A: Mass of forsterite (Mg2SiO4) = 80.0 g Number of magnesium atoms = ?
Q: 3. A šólútióň iš made by dissolving5.02 g ofT KNO3 in 95.0 g of water. Calculate the following a. Mo...
A: Given Data's are : Mass of KNO3 (Solute) = 5.02 g Mass of H2O (Solvent) = 95.0 g calculation of a. ...
Q: e theoretical yield a
A:
Q: IO3. Ksp= 1.57x10^-9 How many millimoles of Ba(NO3)2 are needed to completely react with NaIO3? 5...
A:
Q: A. What is the pH after the addition of 180.0 mg of HBr? Express your answer using two decimal place...
A: Mass of HBr = 180mg Mol. Mass of HBr = 81g/mol Mass of NaOH = 50mg Mol. Mass of NaOH = 40g/mol Assu...
Q: In what phase is the substance at 50 °C and 1 atm pressure? P (atm) 1 . 10 20 30 40 50 60 70 T(deg. ...
A: Solution Given that what phase is the substance at 50 °C and 1 atm pressure = ...
Q: Question 3 Consider the following two molecules. The hydroxyl group in the blue circle is able to fo...
A: To find which hydrogen is more deshielded proton.,
Q: A water has pH = 8.8 and an alkalinity of 3.2 meq/L. Estimate the concentrations of: a. H₂CO3* b. HC...
A: Answer:
Q: Compound Draw in skeletal form (1 as the highest BP and 4 as the lowest SKELATAL/BOND-LINE FORM BP) ...
A: The boiling points of alkanes increase with increasing number of carbons. the boiling point of an a...
Q: Summarize your understanding about the different sources of energy by filling out the table below. U...
A: We are assigned different sources of energy. We have to find their source, examples, and their advan...
Q: b) If that heat came from cooling a 1.361 kg lump of iron, what was the initial temperature of the i...
A:
Q: If you performed this experiment using MgCl2 as the solute would the molar concentration of MgCl2 th...
A: Solution Given that Explain this experiment using MgCl2 a...
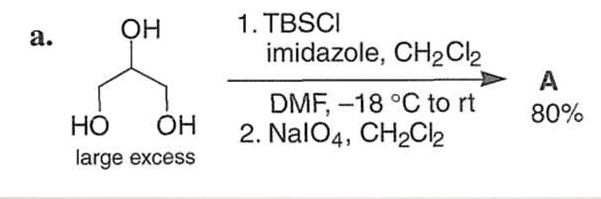
Q: b. HO 1. TsCl, py A> "CH3 2. LIET3BH, THF
A:
Q: Sulfur dioxide and oxygen react to form sulfur trioxide, like this: 250,(g)+0,(g) → 2so,(g) Suppose ...
A: Equilibrium reaction : 2SO2(g) + O2(g) ↔2SO3(g) Perturbation :...
Q: pls explain and answer what is the most stable conformational isomer of butane in the pic
A:
Q: addition of HBr
A:
Q: What is AStot (in )for 4 moles of a substance at 93°C cooling to 32°C at constant pressure? Helpful ...
A: Here we have to calculate ∆S total to convert 4 moles of a substance from 93°C to 32°C by cooling at...
Q: Measurements show that the energy of a mixture of gaseous reactants increases by 175. kJ during a ce...
A:
Q: Determine the number of C atoms in 0.01 g of Caffeine C8H10N4O2
A:
Q: CF2Cl2 CFCl3 C2F3Cl3 CF3Cl How many grams of Cl are in 32.2g of each sample of chlorofluorocarbons (...
A: Molar mass of CF2Cl2 = 120.91 g/mol Molar mass of CFCl3 = 137.36 g/mol Molar mass of C2F3Cl3 = 187.3...
Q: A sample of polystyrene, which has a specific heat capacity of 1.880 J-g .°c', is put into a calorim...
A: Given data,Specific heat capacity of polystyrene=1.880J.g-1.oC-1Mass of water=250.0gInitial temperat...
Q: Which of the following represents the electron configuration of S? A) 1s°2s 2p°3s?3p® B) 1s²2s²2p°3s...
A:
Q: HCI heat CH,MgBr H,0 9) ether h) CH,OH 1. хв СH 2. Ag,0 3. heat
A:
Q: Batteries are rated in terms of ampere-hours (A - h). For example, a battery that can produce a curr...
A: Energy store in battery can be ) calculate by using battery capacity (CA) and voltage of battery. ...
Q: Calculate the mass of hydrazine (N_2H_4) that contains a billion (1.000 x 10^9) nitrogen atoms. Be s...
A: Given, Number of atoms of N in N2H4 = 1.000 x 109 atoms of N Calculate the mass of N2H4 that conta...
Q: Acetylene (C,H, gas is often used in welding torches because of the very high heat produced when it ...
A:
Q: Order the following compounds with respect to SN1 reactivity: Br CH2BR CH CH3 Assuming that the nitr...
A:
Q: Example (2): Calculate the molar fraction of nitrogen in a solution consisting of 14 grams of oxygen...
A:
Q: CE264 students prepared 60g of cement and 60mL of kerosene. They put the kerosene inside the le chat...
A:
Q: Find the solubility of CuI in 0.55 M KCN solution. The Kp of Cul is 1.1 x 10-12 and the Kf for the C...
A:
Q: What is the percent rate if a quantity is 1/1000 of the base?
A:
Q: What are the responsibles ways on using acids and bases?
A:
Q: The required amount of heat of the liquid substance for its one mole to vaporize.
A:
Q: 6. Calculate the pH of the resulting solution when 20.0 ml of 0.25 M HCI is added with 20.1 ml of 0,...
A: Using formula mixing of solutions:- Mmix.=M1V1+M2V2/V1+V2 After calculating [H+]=Mmix. Then use form...
Q: A vitamin syrup for infants contains 27.44 mg of zinc sulfate monohydrate (ZnSO4.H2O) and 100.00 mg ...
A:
Q: PLEASE ANSWER THHS ASAP Find the geometry, hybridization, polarity, and intermolecular forces in eac...
A: Given : In CBr4 central atom is carbon atom while Bromine is surrounding atom. In SPF central atom...
Q: ♥ Draw Lewis structures for each of the following. Use the DEN and the number of REDs around the cen...
A: We have been asked to draw the lewis structure of the given compounds along with the valence electro...
Q: 2) Calculate AS (for the system) when the state of 3.00 mol of perfect monoatomic gas atoms is chang...
A:
Q: for the fischer projection, how do we number the configurations (2R,3S) or (2S,3R)? CHO OH но CH2OH ...
A:
Q: CO,H H2N- -H H FOH CH3 CO,H H2N. H H2N- H. A) Н. LOH B) H. CO,H COH ČH3 ČH3
A: Fischer projection formula is represent the eclipsed conformation of a molecule. The conversion of F...
Q: A nonelectrolyte is an ionic compound that doesn’t dissolve in water. an ionic compound that diss...
A:
Q: Given the reaction inside the container, CO2 (g) + H2O (l) --> H2CO3 (aq). What will happen if the p...
A: We know that When there is an increase in pressure, the equilibrium will shift towards the side of t...
Q: molecular weight
A:
Q: Find the geometry, hybridization, polarity, and intermolecular forces in each molecule for CH2O ( o ...
A: Intermolecular forces are the molecules that exist between two or more molecules.
Q: How many M-M bonds do you predict for each of the following molecules or ions? b. [(n°-CsHs)Mn(CO):]...
A:
Step by step
Solved in 2 steps with 1 images

- The following evidence was obtained from an experiment to determine the solubility of calcium chloride at room temperature. A sample of saturated calcium chloride solution was evaporated to dryness, and the mass of solid residue was measured.EvidenceVolume of solution (mL) = 15.0Mass of empty beaker (g) = 90.54Mass of beaker and residue (g) = 101.36The solubility of calcium chloride is g/100 mLHexanoic acid was added to an immiscible biphasic solvent sysem, water and CCl4 at 20.0OC and the equilibrium concentrations of hexanoic acid were determined to be 3.66 g/L in H2O and 67.0 g/L in CCl4. Caluclate the distrubution coeffiecent (D1) of hexanoic acid in CCl4 with respect to water.Calculate the osmolarity in mOsm/L of 500 mL of a solution containing 5% w/v dextrose, 0.78% w/v sodium chloride, and 27mEq of potassium acetate. dextrose: MW 180, # particles on dissociation 1 sodium chloride: MW 58.5, # particles on dissociation 2, valence 1 potassium acetate: MW 98, # particles on dissociation 2, valence 1
- A solution is prepared by dissolving 40.00 g of NaCl (f.w. = 58.44 g mol–1), a non-volatile solute, in enough water (m.w. = 18.02 g mol–1) to result in exactly 1 L of solution at 25 °C. Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Find the freezing point of the solution(in C to 2 decimal places)A solution is prepared by dissolving 40.00 g of NaCl (f.w. = 58.44 g mol–1), a non-volatile solute, in enough water (m.w. = 18.02 g mol–1) to result in exactly 1 L of solution at 25 °C. Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Find the osmotic pressure in atm to three decimal placesA solution is prepared by dissolving 40.00 g of NaCl (f.w. = 58.44 g mol–1), a non-volatile solute, in enough water (m.w. = 18.02 g mol–1) to result in exactly 1 L of solution at 25 °C. Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Determine the boiling point of the solution(in C to 2 decimal places)
- A solution is prepared by dissolving 40.00 g of NaCl (f.w. = 58.44 g mol–1), a non-volatile solute, in enough water (m.w. = 18.02 g mol–1) to result in exactly 1 L of solution at 25 °C. Assume the density of the solution is that of pure water (dsolution = 1.000 g mL–1). The ebullioscopic constant (Kb) for water is 0.513 °C m–1. The cryoscopic constant (Kf) for water is 1.86 °C m–1. The vapor pressure of pure water is 0.0313 atm. Determine the following: Boiling point of solution (in °C to two decimal places) Freezing point of solution (in °C to two decimal places) Vapor pressure of the solution (in atm to three decimal places) Osmotic pressure (in atm to three decimal places)For 13 and 14. Find the freezing pt. and boiling pt. of a solution containing 40 g sucrose (C₁₂H₂₂O₁₁) dissolved in 200g water. The Kb and Kf values for water are 0.52°C/molal and 1.86°C/molal respectively. 1. Tf solution A. -1.3 deg C B. -1.09 deg C C. -1.0 deg C D. -1.1 deg C 2. Tb solution A. 100.4°C B. 100.3°C C. 100.06°C D. 101°COxygen is soluble in water, much to the benefit of aquatic life forms. At 25 °C, the solubility of oxygen is about 7×10–6 g/cm3, and the pressure of O2 is about 20% of the atmospheric pressure. Could the oxygen dissolved in water affect your experimental results significantly?
- What is the boiling point rise in oF of a 15% solution of nitric acid in water boiling at 200oF? Need asapHexanoic acid was added to an immiscible biphasic solvent system, water and CCl4 at 20.0OC and the equilibrium concentrations of hexanoic acid were determined to be 3.66 g/L in H2O and 67.0 g/L in CCl4. Caluclate the distrubution coeffiecent (D2) of hexanoic acid in water with respect to CCl4.Example: Calculation the weight of barium iodate Ba(IO3)2 if it dissolved in 500mL of distilled water (D.W) at 25 °C if M.wt of ppt = 487 g/mole and %3D Ksp= 1.57x10.





