Q: Why is the amine N atom more nucleophilic than the amide N atom in CH,CONHCH,CH,CH,NHCH,?
A: Nucleophilic tendency of a chemical species is its ability to donate lone pair of electrons to…
Q: Which of the following will give an N-nitroso-N-methyl amine?
A: N-Cyclohexyl-N-methyl amine reaction with HNO2 (NaNO2 + HCl) gives N-nitroso-N-methyl amine.
Q: Show what two compounds you might combine to make the following 3 degreee amine, then what happens…
A: Given that : We have to show what two compounds you might combine to make the following 3 degreee…
Q: Part A Complete the sentences to explain the differences between a salt bridge and a disulfide bond.…
A: Given Part A A Salt Bridge is _________ between ____________ A disulfide Bond is a _________ between…
Q: Show how to convert hexanoic acid to given amine
A:
Q: Construct flow charts (a.k.a. flow diagrams) to show how to separate each of the following pairs of…
A: water insoluble compounds also called as Non polar compounds , which are formed covalent bonding.
Q: Which of the basicity rows of amines given below is correct?
A: Strength of basic nature depends on the tendency of donating the electrons to the electron deficient…
Q: Have a go at naming amines hello CH, H. CI H H• H CH,CH,CH, H. H CH,CH,CH, CH,CH,CH, Notes DO
A: General rules for nomenclature of amine - 'e' ending of alkane of longest chain is change by amine…
Q: Which of the following will give an N-nitroso-N,N-dimethyl amine? „NH2
A: This is the question of Diazotization reaction.
Q: CHC-OCH,CH, D.) сон сон
A: Acid Anhydride is acid derivatives. Which has group = O=C-O-C=O
Q: Reset He CH3COOH CH,CH,CONHCH3 CH;OCH3 CH3CH,NH2 CH3CH,CNH, CH;CNH, CH;COCH3 CH;COOCH,CH3 Carboxylic…
A: Label each molecule with respective category ?
Q: 3. Draw the indicated functional groups below including lone pairs of electrons. You may use R, R1,…
A: Given : Secondary amine, nitro , acid chloride, ester To find : Draw the functional group and show…
Q: Select the 1° amine from the structures below.
A: Given that a question of four cyclic amine compounds. Select the given structure of 1° amine.…
Q: Which of the following has a primary amine (-NH2) as part of its structure? none of the given…
A: Which of the following is correct
Q: Scheme 1 Model A. Methanol: Model B. Methanol amine + red ball- no need of the H yellow red ball +…
A: No Methanol and methanol amine can't overlap each other because for overlapping all substituent must…
Q: An amide has the general structure 2 eq. NH2 NH3 CI + remainir R1- -R2 emaining product For this…
A: Applying concept of reagents and reaction mechanism.
Q: what happens when an amide reacts with water?
A:
Q: Select the reaction that shows the amide ion, NH2, functioning as a pase. O NH2 (g) + C2H2(g) = C2H3…
A: Answer
Q: 1. Underthe conditions used in this experiment, is it possible to stop the reaction at the amide…
A: The production of an acid from nitriles is done by the hydrolysis of that acid. The given reaction…
Q: An aqueous solution of a primary or secondary amine reacts with an acyl chloride to form an amide as…
A:
Q: Complete the below statements with reference to the structures shown in the figure. Drag and drop…
A: Primary amine is in which nh2 is present. Secondary alcohol in which -OH attached with Secondary…
Q: Why would the following reaction first produce an amide and then convert it back to the starting…
A:
Q: he name of the product in question 8 is: A. imine B. oxime C. hydrazone D. None of the above.
A:
Q: Name the following compounds (Amine) 1st pic
A: "To get remaining sub-parts solved please repost the complete question and mention the sub-parts to…
Q: The molecule shown below can best be described as what class of m H H 1 N-H H-C- H OA. Amide B.…
A:
Q: 1. Name this amine properly. Is it a primary, secondary, or tertiary amine? Which of the following…
A:
Q: Rank the following functional groups from least to most reactive: acid anhydride, acyl chloride,…
A:
Q: Thiol, Either, Sulfide, Aromatic, Amine, Imine Please label each one hydrophilic or hydrophobic
A: Thiol - hydrophilic These are polar and hydrophilic, which allows them to interact with aqueous…
Q: Fill in the blanks: The amide substituent of phenol by of the phenoxide through 15 the acidty the…
A: The acidity is defined by the tendency to lose a proton or to accept a pair of electrons. The…
Q: Name Structure Acid/Base/Neutral? | Reacts ...to form? with... (name it) Carboxylic Acid Strong base…
A:
Q: 1. Name this amine properly. CH3 CHb NH CH(CH3), Is it a primary, secondary, or tertiary amine?
A: According to Bartlby guidlines I solve only first question. You post second question separately.
Q: Show what two compounds you might combine to make the following 3° amine, then what happens to that…
A:
Q: Name the following compounds. Where necessary use E/Z notation. 4 N-ehyl-N-methylpropanamide…
A: E/Z notions : To describe the stereochemistry E/Z notion is used.
Q: Define the simplest method to synthesize an amine ?
A: Amines Amines are derivatives of ammonia, in which the Hydrogens are substituted by alkyl or aryl…
Q: Draw the product that will form if the amine in Figure 25 reacts with hydrochloric acid in an…
A:
Q: First, draw your chosen compounds including any non-bonding electrons and non-zero formal charges…
A: The given molecules are p-anisic acid which is polar and naphthalene which is non-polar.
Q: Which amine is less basic? Choose the option that has the factor that makes it less basic. N. .N' 1
A: Amine (1) is less basic due to hybridisation (1). i.e. pyridine is less basic than dimethyl amine.
Q: Choose the amine that would accomplish the reaction shown. ???? pH – 4.8 JDHDMC NH2 NH2 NH2 NH2
A:
Q: Match the following compounds given in Column A and their classes given in Column B. CAPITAL LETTERS…
A:
Q: Which of the following is NOT a primary amine? Select one: a. o N-CH3 b. NH₂ Ý > NH₂ o C. d. -NH₂
A: Amines can be catogarised into Primary Secondary Tertiary
Q: Draw the product that will form when the amine in Figure 25 reacts with hydrochloric acid in an…
A:
Q: Which of these molecules are amides? Check all that apply. DA D. DE
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Use the approximate pkKa's of the functional groups to select all acid-base pairs that give an…
A:
Q: Provide the functional group contained in each lettered compound:
A: Since you have posted multiple questions, we are entitled to answer the first only. Please repost…
Q: H* Amine + Carbonyl group H20 NH2 II IV O II
A: Amine and carbony compound in presence of H+/H2O give the enamine as shown in question .Enamine can…
Q: draw amide functional group with no pka as it exist at ph 1 and ph 10 ( draw twice)
A: We have to keep in mind the value of pH specifically. Lower pH designates the condition is acidic…
Q: Why is the amine N atom more nucleophilic than the amide N atom in CH3CONHCH2CH2CH2NHCH3?
A: Nucleophilicity of a chemical species depends on availability of electrons on it. Factors such as…
Q: From the given structures which is (a) amide that will release a secondary amine upon hydrolysis?…
A: A question about amide, which is to be accomplished.
Q: Question 10 Is the following more likely a nucleophile or a base? O Nucleophile O Base
A: There are the presence of two sterically groups are attached on this structure that's why it is much…
Q: -Which of the above i on amide? 3. Which is a nitrile? e. Which is an anhydride? . Which is an acid…
A: These are all the functional groups.They have their own characteristic properties regardless of the…
show complete reaction mechanism
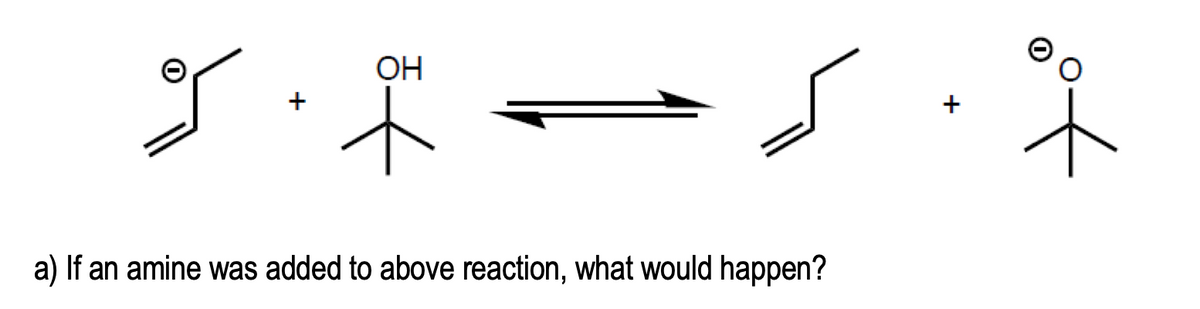
Step by step
Solved in 2 steps with 2 images

- The structure of Amines are classified as primary (1), secondary (2), and tertiary (3) based on the number of R group that concept that seemed difficult to you at first, but then after working on the concept, you were able to master it. Include a description of what made the concept difficult at first, and then discuss what you did in order to better understand the concept.State The Gabriel Synthesis of 1° Amines ?The structure of Amines are classified as primary (1), secondary (2), and tertiary (3) that concept that seemed difficult to you at first, but then after working on the concept, you were able to master it. Include a description of what made the concept difficult at first, and then discuss what you did in order to better understand the concept.
- Be sure to answer all parts. What ammonium salt is formed when this biologically active amine is treated with H2SO4? Be sure to include the charge(s). draw structure ...At pH = 7.0, generally speaking amine groups will be found as which of the following?The structure of Amines are classified as primary (1), secondary (2), and tertiary (3) based on the number of R group including example that concept that seemed difficult to you at first, but then after working on the concept, you were able to master it. Include a description of what made the concept difficult at first, and then discuss what you did in order to better understand the concept.
- From the given structures which is(a) amide that will release a secondary amine upon hydrolysis? (b) product of hydrolysis of MSO (c) a tertiary amide and (d) a diketoneHi, can you draw the structure of trametinib; thanks!Benzylpenicillin shown in Figure 34 was the first antibiotic to be manufactured on large scale. How many ester groups can be formed when this compound reacts with methanol in the presence of strong acid? [The answer is a whole number.] *

