First, draw your chosen compounds including any non-bonding electrons and non-zero formal charges Circle and name any pH-sensitive functional groups. Choose an organic solvent that will dissolve both or your compounds and an aqueous solution (i.e. conc. aqueous NaOH, 5% aq. NaHCO3, or 3M HCl) that would successfully separate the two compounds selected.
First, draw your chosen compounds including any non-bonding electrons and non-zero formal charges Circle and name any pH-sensitive functional groups. Choose an organic solvent that will dissolve both or your compounds and an aqueous solution (i.e. conc. aqueous NaOH, 5% aq. NaHCO3, or 3M HCl) that would successfully separate the two compounds selected.
World of Chemistry, 3rd edition
3rd Edition
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Chapter2: Matter
Section: Chapter Questions
Problem 9STP
Related questions
Question
First, draw your chosen compounds including any non-bonding electrons and non-zero formal charges
Circle and name any pH-sensitive functional groups.
Choose an organic solvent that will dissolve both or your compounds and an aqueous solution (i.e. conc. aqueous NaOH, 5% aq. NaHCO3, or 3M HCl) that would successfully separate the two compounds selected.
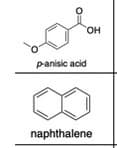
Transcribed Image Text:он
panisic acid
naphthalene
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

Recommended textbooks for you

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning