Q: The cis and trans isomers of 2,3-dimethyloxirane both react with −OH to give butane-2,3-diol. One st...
A: In the given reaction, both cis and trans isomers of 2, 3-dimethyloxirane react with -OH (a strong n...
Q: Explain why, when a small amount of glycerol is added to the reaction mixture of toluene-2,6-diisocy...
A: When a small amount of glycerol is added to the reaction mixture of toluene-2,6-diisocyanate and eth...
Q: Draw a stepwise mechanism for the following reaction.
A: The reaction with NBS in presence of hv is a free radical reaction. NBS creates a free radical in co...
Q: Draw the repeating unit of the step-growth polymer that is formed from the following pairs of monome...
A:
Q: Calculate the solubility of Co(OH)3 (s), Ksp = 2.5 x 10-43, in a solution at pH= 4.0
A: The equilibrium is,
Q: Use resonance structures to explain the stabilization of the intermediate formed in the cationic pol...
A: Since the intermediate formed in the cationic polymerization of styrene, is the carbocation. And sin...
Q: An equilibrium mixture of PCI, (g), PCI, (g), and Cl,(g) has partial pressures of 217.0 Torr, 13.2 T...
A: The given balanced chemical reaction at equilibrium is The Kp expression and Kp value for above rea...
Q: Draw a stepwise, detailed mechanism for the following reaction.
A: Since the reactant is tertiary alkyl halide. Hence it will form a carbocation in intermediate Hence...
Q: Draw a stepwise mechanism for the following reaction.
A: The two products are formed by 1,2 addition and 1-4 addition of DCl to the alkene.
Q: Predict the product of the following reactions. [H,SO,] Xs MEOH -Н-о но Он [H,SO4] b) -но
A: a) Please find below the product along with the reaction mechanism.
Q: Table 2. Gibbs Free Energies of formation (kJ), ∆G°f, for Ions in 1M Solution and Ionic Solids Ca...
A:
Q: Consider the following redox equation.Mn(OH)2(s) + MnO4–(aq) → MnO42–(aq) (basic solution)When the e...
A:
Q: Which of the following is false about ethers? Group of answer choices will not burn can be ...
A: Ethers are quite stable compounds and they possess low chemical reactivity. They are extremely flamm...
Q: Which bond in each pair has the higher bond dissociation energy?
A: 1) Since we know that the atomic size increases on moving top to bottom in a group of periodic table...
Q: Pentalene, azulene, and heptalene are conjugated hydrocarbons that do not contain a benzene ring. Wh...
A: The shown above do not contain benzene ring but are conjugated. The structure of the pentalene is pl...
Q: Rank the compounds in each group in order of increasing strength of intermolecular forces.
A:
Q: Draw the products of each elimination reaction.
A: a) Please find below the products formed by elimination reaction. The 2nd minor product which is a...
Q: Calculate the expected Ecell of a 0.002M Cu^2+ solution paired with a Cu^2+(0.1M)/Cu half-cell . Her...
A: The given cell represents a concentration cell. A concentration cell consists of same species at ano...
Q: Consider a mixture of 75.00 g water at room temperature 23.0 0C and 25.0 g water at 95.0 0C. Cal...
A:
Q: Calculate the pH of a 3.5 M solution and the equilibrium concentrations of the species H3PO4, H2PO4-...
A: Ka1 value of H3PO4 is 7.5×10-3. Ka2 value of H3PO4 is 6.2×10-8. Ka3 value of H3PO4 is 4.8×10-13. H3P...
Q: 1. Given the following problem, why would you NOT want to use the formula V1 / T1 = V2 / T2 to so...
A: Hey, since there are multiple questions posted, we will answer the first question. If you want any s...
Q: The four solutions were made and then mixed in the following order. 1. Starting with 0.100M Bacl2 10...
A: Morality: Morality is calculated by moles of solute divided by volume of the solution. ...
Q: For the chemical equations shown below, label each reactant as either acid or base, and each product...
A: Bronsted-Lowry acid is species that loses proton. Conjugate base is formed when acid loses a proton....
Q: Devise a synthesis of X from the given starting materials. You may use any organic or inorganic reag...
A:
Q: #4 Find the pH of a buffer that consists of 0.60 M HCOOH and 0.22 M HCOONa. (pKa of HCOOH = 3.74) pH...
A: The given problem is of buffer solution between an acid and its salt. Concentration of HCOOH = 0.6...
Q: A student weighed out 0.150 g of protein powder and dissolved it in 100 mL of water (Solution 1). Th...
A:
Q: Heptane will react with the following: Group of answer choices hydrochloric acid water no...
A: Heptane does not react with mentioned compound . 1)Concentrated sulphuric acid - No reaction , becau...
Q: Please help me, double check your answers as previous tutors got it wrong.
A: pH is the potential of the hydrogen or the power of the hydrogen. It basically explains how acidic o...
Q: State the three postulates (principle statements) of Kinetic Molecular Theory.
A: EXPLANATION: Postulates of Kinetic molecular theory of gases are: 1)The molecules in a gas are small...
Q: The mercury (I) ion (Hg22 +, known as the mercury ion) is a diatomic ion with a 2+ charge. Mercury i...
A: The dissociation reaction is given as Hg2(IO3)2 (s) ------> Hg22+ (aq) + 2 IO3- (aq) Since KIO3 ...
Q: Please answer Part 2. Thank you.
A:
Q: For the reaction shown, find the limiting reactant for each of the following initial amounts of reac...
A:
Q: Please show work
A: Pre requisite: Molar mass of Chlorine = 35.5 g/mol Molar mass of Zinc = 65.4 g/mol Molar mass of Oxy...
Q: Describe principles of separation by capillary isotachophoresis (CI) using a diagram
A: Isotachophoresis is a technique used for selective separation and concentration of ionic analytes. I...
Q: Draw the products of these reactions
A: The reaction of epoxides with LiAlH4 and H2O results in the opening of epoxide ring. This forms alco...
Q: Using curved notation, write the stepwise mechanism for the condensation reaction between 1,10-decan...
A: The condensation reaction between 1,10-decanedioicdiacid chloride and 1,6-hexanediamine to gives Nyl...
Q: To what volume should you dilute 50 mL of a 13 M stock HNO3 solution to obtain a 0.105 M HNO3 soluti...
A: The volume of diluted solution can be calculated using the equation (1). In this equation, M1 and V1...
Q: Rank the following isomers in order of increasing boiling point.
A: Boiling point depends on 1. Surface Area: Low Surface Area means Low Boiling Point 2. Dipole Moment:...
Q: If you were assigned casein protein powder in lab, how much powder (in g) would you need to add to t...
A: Amount of casein protein powder required to make a solution of 1 mL = 1.42 mg Therefore, amount of c...
Q: If squalene is synthesized in a medium containing acetate with a 14C-labeled carbonyl carbon, which ...
A:
Q: If a balloon filled with air has a volume of 15L at 5°c, what is the volume at 75°c?
A: The given conditions are as follows: Initial Volume (V1) = 15 L Final Volume (V2) = V2 Initial Tempe...
Q: Ammonia can also be synthesized by the reaction: 3H2 (g) + N2(g)→2NH3(g) What is the theoretical yie...
A: To calculate the theoritical yield , 1) Write the balanced chemical equation. 2) Identify the limiti...
Q: Draw the products of each reaction.
A: The epoxide is the three-membered ring structure in which on one of the vertices, we have oxygen and...
Q: A 0.395-M aqueous solution of a weak acid has a pH of 3.67. Calculate Ka for the acid. Ka =
A: pH = -log[H+] where [H+] = concentration of H+ ions => 3.67 = -log[H+] => [H+] = 10-3.67 = 2.1...
Q: Fluticasone, the chapter-opening molecule, can be prepared by the following reaction. Draw a stepwis...
A:
Q: ho <CHE154 S20 Ch19 Sec8-10 Ay Chapter 19 Multiple Choice Question 48 Review I Consta Part A What is...
A: The relationship between equilibrium constant K and ΔG0rxn is given by ΔG0rxn = -RTln(K) Hence if ...
Q: 2.) Answer the following questions pertaining to the dissociation of acetic acid (CH3COOH) given bel...
A: The equilibrium constant for acid dissociation is denoted as Ka. It represents the ratio of the equi...
Q: Draw a stepwise, detailed mechanism for the following reaction.
A: The CH3NH2 attacks on the Cl and removes it in step 1. And then in step 2, due to presence of intern...
Show how the reactant can be converted to the product in two steps.
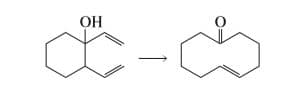
Step by step
Solved in 3 steps with 4 images


