One mole of diatomic ideal gas undergoes a reversible carnot cycle shown in the figure. Processes 1 2 and 3 4 are reversible isothermal process while processes 2 → 3 and 4 1 are reversible adiabatic process. What are the en- tropy changes for each process in the cycle? 120 70 3 0.0127 0.02 Volume (m') Pressure (kPa)
One mole of diatomic ideal gas undergoes a reversible carnot cycle shown in the figure. Processes 1 2 and 3 4 are reversible isothermal process while processes 2 → 3 and 4 1 are reversible adiabatic process. What are the en- tropy changes for each process in the cycle? 120 70 3 0.0127 0.02 Volume (m') Pressure (kPa)
Principles of Physics: A Calculus-Based Text
5th Edition
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Raymond A. Serway, John W. Jewett
Chapter17: Energy In Thermal Processes: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 36P
Related questions
Question
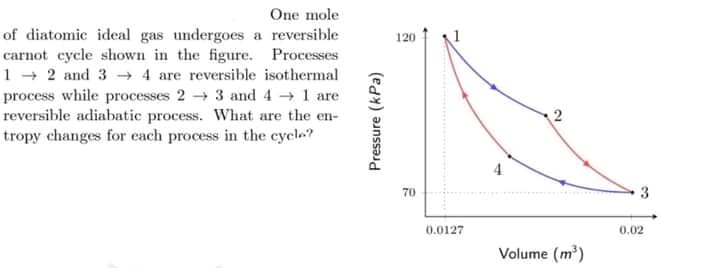
Transcribed Image Text:One mole
of diatomic ideal gas undergoes a reversible
carnot cycle shown in the figure. Processes
1 + 2 and 3 -→ 4 are reversible isothermal
process while processes 2 3 and 4 → 1 are
reversible adiabatic process. What are the en-
tropy changes for each process in the cycle?
120
70
3
0.0127
0.02
Volume (m)
Pressure (kPa)
2.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781305952300
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781305952300
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

Physics for Scientists and Engineers with Modern …
Physics
ISBN:
9781337553292
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning


Physics for Scientists and Engineers
Physics
ISBN:
9781337553278
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning