Optimum temp of amylase was identified by incubation of the amylase together with starch at various temps for 10 mins. and stopped with HCI. The volume of added DNS is negligle. Volume Volume Volume Concentration of Temperature (°C) of of stock of stock starch distilled Absorbance amylase starch (mg/mL) water (uL) (mL) (mL) 1 1 10 9 0.0053 10 1 1 10 9 0.1309 20 1 1 10 9 0.1185 30 1 1 10 9 0.0531 40 1 1 10 0.0206 Using this graphically determine the optimum temp of amylase?
Optimum temp of amylase was identified by incubation of the amylase together with starch at various temps for 10 mins. and stopped with HCI. The volume of added DNS is negligle. Volume Volume Volume Concentration of Temperature (°C) of of stock of stock starch distilled Absorbance amylase starch (mg/mL) water (uL) (mL) (mL) 1 1 10 9 0.0053 10 1 1 10 9 0.1309 20 1 1 10 9 0.1185 30 1 1 10 9 0.0531 40 1 1 10 0.0206 Using this graphically determine the optimum temp of amylase?
Chapter13: Dimensional Analysis/units Conversion
Section: Chapter Questions
Problem 1.3P
Related questions
Question
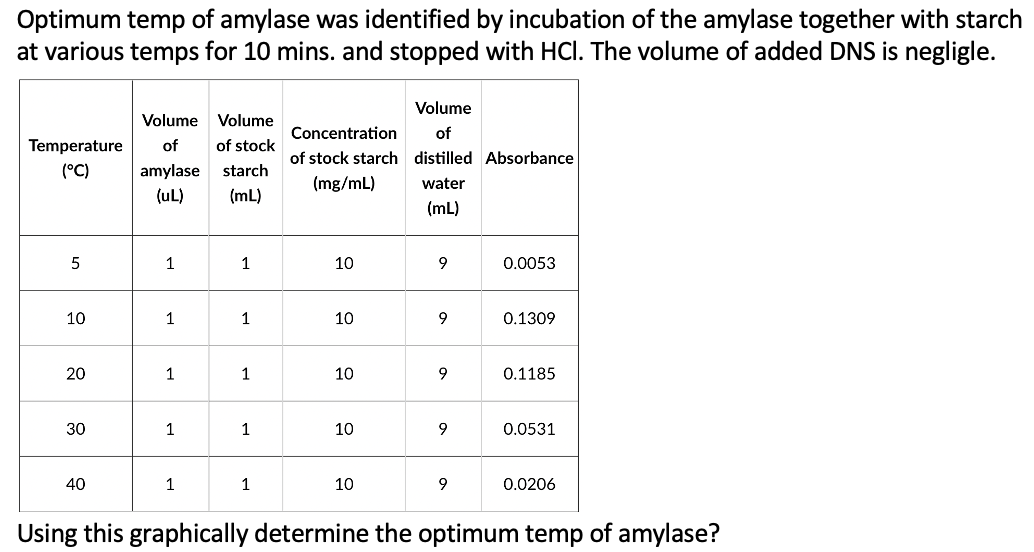
Transcribed Image Text:Optimum temp of amylase was identified by incubation of the amylase together with starch
at various temps for 10 mins. and stopped with HCI. The volume of added DNS is negligle.
Volume
Volume
Volume
Concentration
of
Temperature
(°C)
of
of stock
of stock starch distilled Absorbance
amylase
starch
(mg/mL)
water
(uL)
(mL)
(mL)
1
1
10
9
0.0053
10
1
1
10
9
0.1309
20
1
1
10
9
0.1185
30
1
1
10
9
0.0531
40
1
1
10
0.0206
Using this graphically determine the optimum temp of amylase?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning


Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning