Organic compounds that contain large proportions of nitrogen and oxygen tend to be unstable and are easily decomposed. Hexanitroethane, C₂(NO2)6, decomposes according to the equation C2(NO2)6 2NO2 + 4NO+2CO2 The reaction in CCl4 solvent is first order with respect to C₂(NO2)6. At 73.8 °C, k = 0.000330 s¹ and at 103.0 °C, k=0.0189 s ¹. (a) What is the half-life of C₂(NO2)6 at 73.8 °C? i eTextbook and Media (b) What is the half-life of C₂(NO2)6 at 103.0 °C? seconds seconds (c) If 0.100 mol of C₂(NO2)6 is dissolved in CCl4 at 73.8°C to give 1.00 L of solution, what will be the C₂(NO2)6 concentration after 50.0 minutes? i M
Organic compounds that contain large proportions of nitrogen and oxygen tend to be unstable and are easily decomposed. Hexanitroethane, C₂(NO2)6, decomposes according to the equation C2(NO2)6 2NO2 + 4NO+2CO2 The reaction in CCl4 solvent is first order with respect to C₂(NO2)6. At 73.8 °C, k = 0.000330 s¹ and at 103.0 °C, k=0.0189 s ¹. (a) What is the half-life of C₂(NO2)6 at 73.8 °C? i eTextbook and Media (b) What is the half-life of C₂(NO2)6 at 103.0 °C? seconds seconds (c) If 0.100 mol of C₂(NO2)6 is dissolved in CCl4 at 73.8°C to give 1.00 L of solution, what will be the C₂(NO2)6 concentration after 50.0 minutes? i M
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 11.36PAE: The reaction NO(g) + O,(g) — NO,(g) + 0(g) plays a role in the formation of nitrogen dioxide in...
Related questions
Question
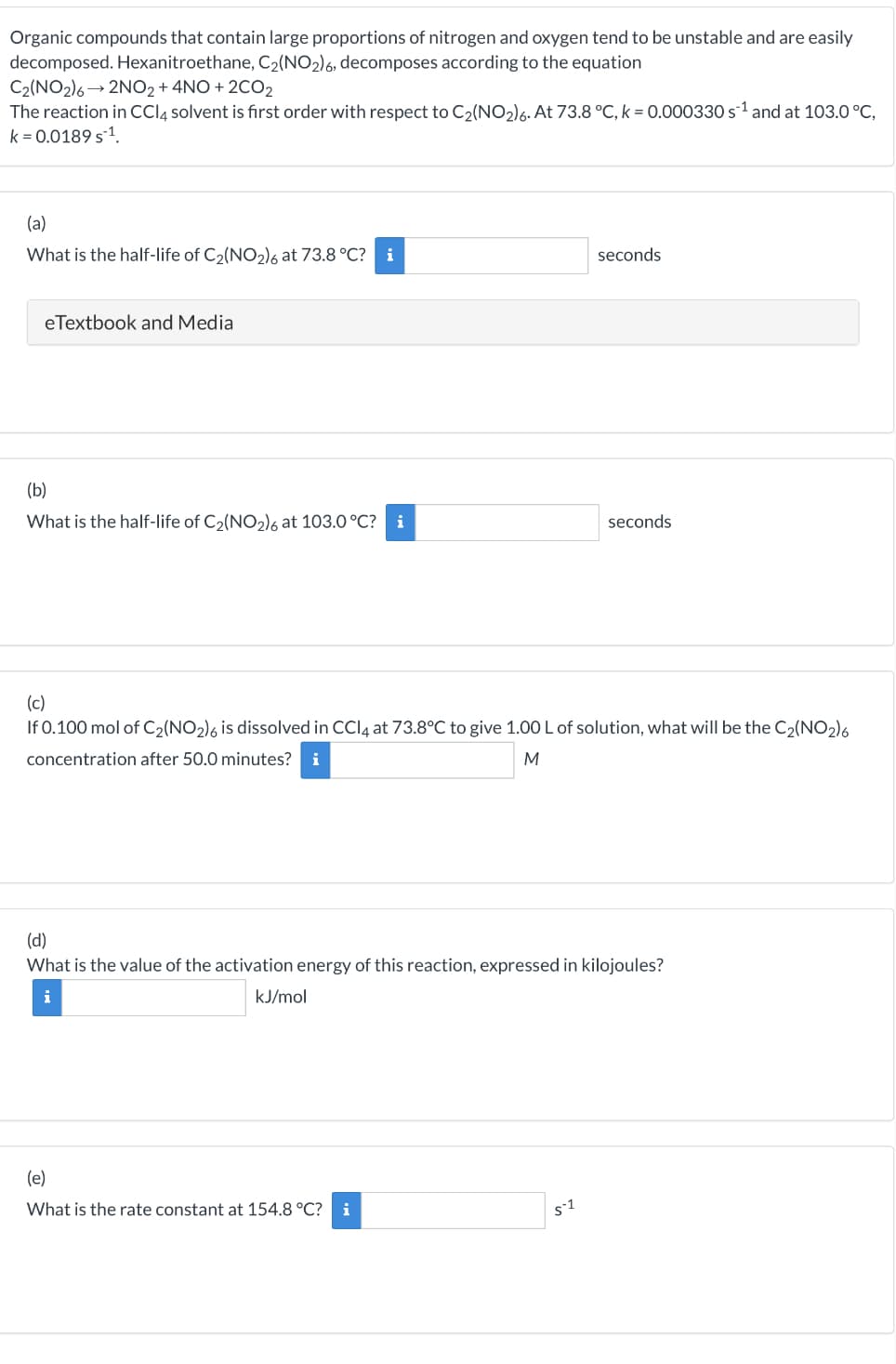
Transcribed Image Text:Organic compounds that contain large proportions of nitrogen and oxygen tend to be unstable and are easily
decomposed. Hexanitroethane, C₂(NO2)6, decomposes according to the equation
C₂(NO2)6→ 2NO2 + 4NO+2CO2
The reaction in CCl4 solvent is first order with respect to C₂(NO₂)6. At 73.8 °C, k = 0.000330 s¹ and at 103.0 °C,
k=0.0189 s ¹.
(a)
What is the half-life of C₂(NO2)6 at 73.8 °C? i
eTextbook and Media
(b)
What is the half-life of C₂(NO₂)6 at 103.0 °C? i
seconds
(c)
If 0.100 mol of C₂(NO2)6 is dissolved in CCl4 at 73.8°C to give 1.00 L of solution, what will be the C₂(NO2)6
concentration after 50.0 minutes? i
M
(e)
What is the rate constant at 154.8 °C? i
seconds
(d)
What is the value of the activation energy of this reaction, expressed in kilojoules?
kJ/mol
51
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 8 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning