Q: draw an atomic/molecular level representation of what you think the solvent (H2O) looks like before ...
A: Water is a polar molecule, which is liquid at standard temperature and pressure. Water molecules can...
Q: a certain temperature, the equilibrium constant K for the following reaction is 8.80 x 10 H,(g) + Cl...
A: The reactions for which equilibrium constant is very small hardly form any product of sizeable quant...
Q: What cyclic product is formed from the dihalide using the masonic ester synthesis?
A: Concept: In the malonic ester synthesis diethyl malonate or another ester of malonic acid is alkyla...
Q: Americium-241 is used in smoke detectors. If 15% of the americium-241 in a smoke detector decays in ...
A:
Q: You add some of the white crystals to a small test tube filled with water. The water in the test tub...
A: Here we have to say the change in Gibbs free energy for above dissolution process of some crystals-
Q: How many grams of H3PO4 are in 975 ml of a 4.00 M solution of H3PO4
A: 1. Given :- Molarity of H3PO4 solution = 4.00 M Volume of H3PO4 solution = 975 mL To be calculate...
Q: solve the problem using SF and PsN. Need help understanding help please
A:
Q: Industrial production of hydrogen gas uses the reaction shown below. If 1.00 metric ton of propane r...
A: Note:-1) 1 metric ton =1000Kg 2)1kg=1000kg 3)Molar mass of propane is 44g/mole 4)% yield = [Actual ...
Q: Which one of the following is an empirical formula? O C2F6 O H2SO2 O C2H402 O P4010
A:
Q: how does sugar smell, what color is sugar, what phase (solid, liquid, or gas) does sugar occur in, a...
A: Pure sugar (chemical name - sucrose ) does not have any smell.Only those substances that are relativ...
Q: How many GRAMS of chromium(II) sulfite are present in 2.73 moles of this compound?
A: Given :- number of moles of chromium(II)sulfite = 2.73 mol To be calculated :- mass of chromium(I...
Q: Four different molecules are shown below, and the highest priority functional group in each molecule...
A: The data given is,
Q: 6. Bond enthalpy Use the given bond enthalpies to estimate the AH for the combustion of 1 mole of me...
A:
Q: Practice Exercise The reaction between aluminum and iron(III) oxide ca temperatures approaching 3000...
A: Given datas ; 2 Al + Fe2O3 ---> Al2O3 + 2 Fe Mass of Al = 124 g Mass of Fe2O3 = 601 g (a.) Al...
Q: trans or cis? include the name
A:
Q: Calculate the mass of tetraborane that contains a billion (1.0x10^9) boron atoms.
A: Given :- number of boron atoms = 1.0 × 109 atoms To be calculated :- mass of tetraborane (in grams)
Q: Given a solution containing 0.010 M CaCl2 and 0.020 M MgCl2, what is the activity of Ca2+?
A: Activity of ion is expressed by the effective concentration of ion. In the solution CaCl2 and MgC...
Q: Which of the following true about reaction mechanisms? O a) The first step is always the slow step. ...
A: We have to find the true options from the given statements about reaction mechanism.
Q: CH3 CH3-CH2-CH–ĆH-0-CH3 (Select all that apply.) ethanal O 3-methylbutanal O propanal methanal O 2,2...
A: Acetals and hemiacetals on hydrolysis gives corresponding aldehyde or ketone and alcohols
Q: please draw the molecule and label it based on the data in the sheet and use the label in the data t...
A: Proton NMR: aromatic region : delta 6.5 to 8.0, Oxygen attached carbon having hydrogens at delta 3.5...
Q: Rank the following compounds from least to most stable. Least stable Most stable
A: The stability of alkenes can be given by resonance or hyperconjugation. Hyperconjugation and resona...
Q: tomic radii decrease from left to right in a period (Na → Ar) on the periodic table Choose the best ...
A: Interpretation: We have to explain why ionic radii increase on moving from left to right.
Q: Determine the number of NH+ and CO, ions required to form a neutral ionic compound. 2- Cations Anion...
A: the number of NH4+ and CO32- ions required to form a neutral compound:
Q: .) Determine the empirical formula for the compound with the molecular formula C14H10O4 Empirical ...
A: Given Molecular formula = C14H10O4 Empirical formula = ?
Q: Which of the following statements is INCORRECT? O Filtration banks on the different sizes of mixture...
A: Filtration , distillation and chromatography are separation techniques.
Q: Which of the following occur if the energy of a photon increases? The speed of the photon increases....
A:
Q: When 4.673 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 16.05 grams...
A: Given: Mass of compound burned = 4.673 g. Mass of CO2 produced = 16.05 g. Mass of H2O produced = 2.6...
Q: In C6H12, how many constitutional isomers have atleast 1 3^o carbon? explain
A: Those carbon which is directly joint with three carbon called 3० carbon
Q: draw an atomic/molecular level representation of what you think the solute (KI) looks like before mi...
A: The solute KI is an ionic compound. An ionic compound is also known as salt.
Q: Draw the structure of the following alkynes: 1. 6-methyl-3-heptyne 2. 3-bromo-3-ethyl-4-octyne 3. 2-...
A: IUPAC rules: First write the parent chain and keep the functional group at right place according to ...
Q: What is the frequency of a photon with a wavelength of 535 nm? O 5.61 x 1014 s 1 O 8.90 x 1014 s1 O ...
A:
Q: A calorimeter contains 30.0 mL of water at 14.0 °C. When 1.70 g of X (a substarice with a molar mass...
A:
Q: Draw structure for heptyl benzoate
A: We have to draw structure for heptyl benzoate .
Q: (2S)-2-amide-3-mercaptopropan-1-olc acid
A: The explanation is given below-
Q: Ascorbic acid (C,H3O6) is also known as Vitamin C. How many molecules of C6H;O6 does a Vitamin C dri...
A:
Q: In each of the following pairs, which has the greater mass? a. 1.0 kg of feathers or 1.0 kg of lead...
A: Note: According to our guidelines we are supposed to answer only first three subpart. Kindly repost...
Q: Consider the following reaction where Kc = 1.20×10-2 at 500 K. PCl5(g) PCl3(g) + Cl2(g) A reaction...
A:
Q: 5. Percent NaCl in the saturated solution: Show the calculation, including units and sig figs: 27.07...
A: “Since you have asked multiple question, we will solve the first question for you. If you want any s...
Q: A helium-filled balloon at 1.00 atm pressure has a volume of 1.340 L. It is placed in a freezer and ...
A: Given data, Pressure in balloon = 1.00 atm Initial volume (V1) = 1.340 L Final volume (V2) = 0.97...
Q: In which set do all elements tend to form anions in binary ioninic compounds? Cs, B, O Ca, Zn, Pb ...
A: in the given option, the set of elements tend to form anions in binary ionic compounds is:
Q: Identify the correct structure below that represents the following molecule: (3E,5E)-6-chloro-3,5-oc...
A: When same periority on same side then Z-configuration and when same periority on opposite side the E...
Q: Calculate the activity coefficient, Y, of Sn²+ when the ionic Ionic Activity strength of the solutio...
A: The explanation is given below-
Q: what are the advantages of mediator-based amperometric enzyme sensors for glucose compred to detecti...
A: Mediators based amperometric enzyme sensors for glucose compred to detecting hydrogen peroxide has m...
Q: Balance the chemical equation below using the smallest possible whole number stoichiometric coeffici...
A: The reaction taking place is given as, => CH3(CH2)5CH3 (l) + O2 (g) → CO2 (g) + H2O (g)
Q: Does a precipitate form when A and B empirical formula of solution A solution B precipitate are mixe...
A: Interpretation: We have to determine whether precipitate will form or not.
Q: The sum of the oxidation numbers of Polyatomic ions is not equal to zero. Some polyatomic ions, thei...
A:
Q: A compound is found to contain 34.60 % chlorine , 46.85 % oxygen , and 18.55 % fluorine by mass. To...
A: Given: Mass percent of chlorine = 34.60 % Mass percent of oxygen = 46.85 % Mass percent of fluorine ...
Q: Draw the compound Cis-hexane-2,4-diol
A:
Q: Please hep with 1-3. thanks 1. In the laboratory, a general chemistry student measured the pH of a 4...
A: Ka is the acid dissociation constant. pH = -log[H+]
Q: 29) For the carbonate ion, CO,2, which of the following is a correct description of its bonding? A) ...
A: Note: According to our guidelines we are supposed to answer only one question. Kindly repost other ...
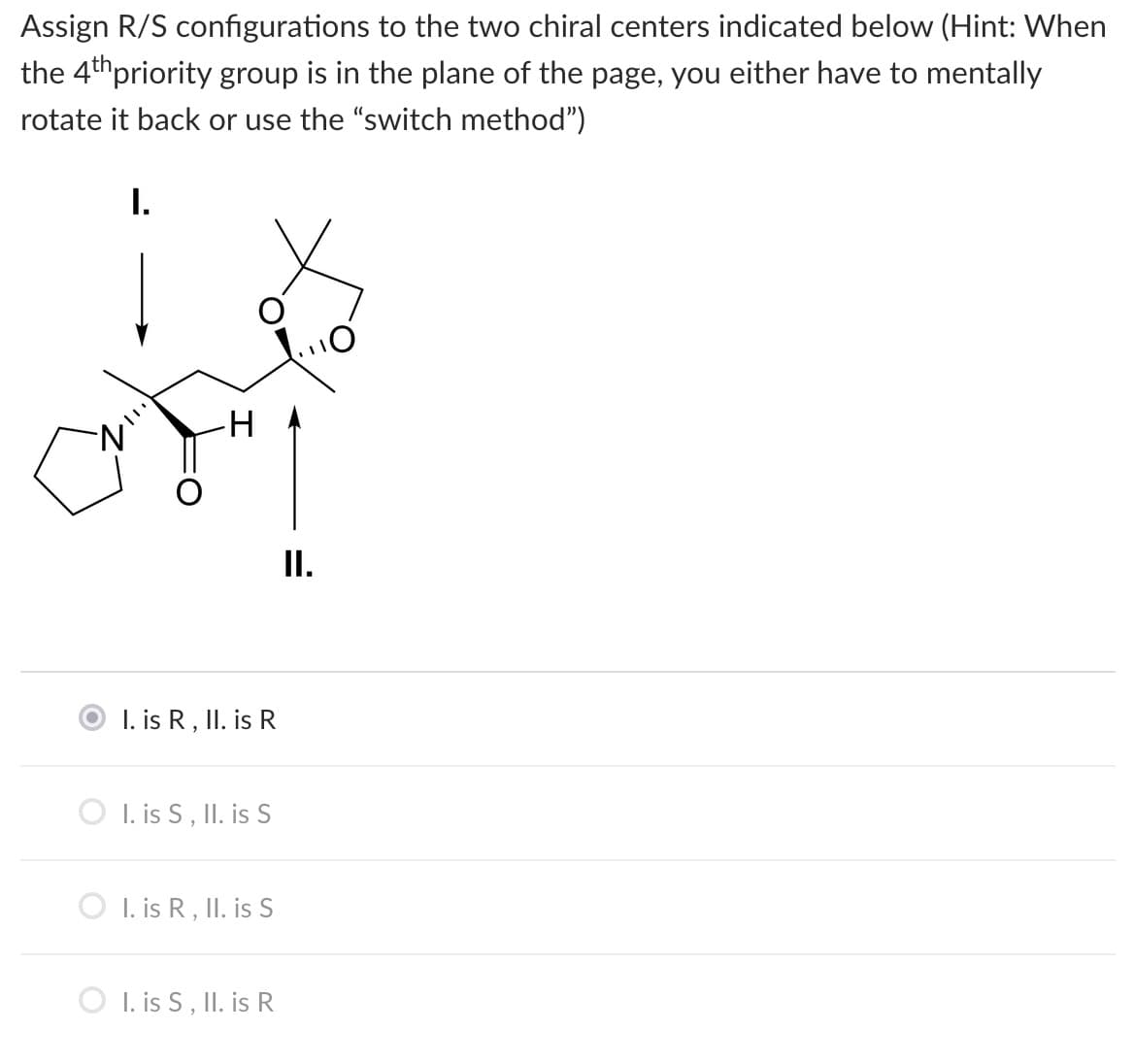
NOTE: I is r, II is R (First option) is the incorrect answer
Step by step
Solved in 2 steps with 1 images

- Determine the absolute configuration of the chiral C in this molecule. (kindly show explanation or illustration. thank you!) Choices: A. R/S not applicableB. SC. RFind all the chiral centers in the following 2 compounds and name the centers as R or S . Show work.1) The most stable chair confirmation of

