Part A Calculate the volume of the gas, in liters, if 1.75 mol has a pressure of 1.26 atm at a temperature of -8 °C. Express the volume in liters to three significant digits. V| ΑΣΦ h CE ? V L Submit Request Answer Review Part B Calculate the absolute temperature of the gas at which 3.53x10-3 mol occupies 498 mL at 760 torr Express the temperature in kelvins to three significant digits. VE ΑΣΦ T = K Submit Request Answer Part C Calculate the pressure, in atmospheres, if 2.75x10-3 mol occupies 415 mL at 137 °C. Express the pressure in atmospheres to three significant digits. VE ΑΣΦ ? P = atm
Part A Calculate the volume of the gas, in liters, if 1.75 mol has a pressure of 1.26 atm at a temperature of -8 °C. Express the volume in liters to three significant digits. V| ΑΣΦ h CE ? V L Submit Request Answer Review Part B Calculate the absolute temperature of the gas at which 3.53x10-3 mol occupies 498 mL at 760 torr Express the temperature in kelvins to three significant digits. VE ΑΣΦ T = K Submit Request Answer Part C Calculate the pressure, in atmospheres, if 2.75x10-3 mol occupies 415 mL at 137 °C. Express the pressure in atmospheres to three significant digits. VE ΑΣΦ ? P = atm
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter5: Gases, Liquids, And Solids
Section: Chapter Questions
Problem 5.55P
Related questions
Question
Please answer all parts or leave for someone else.
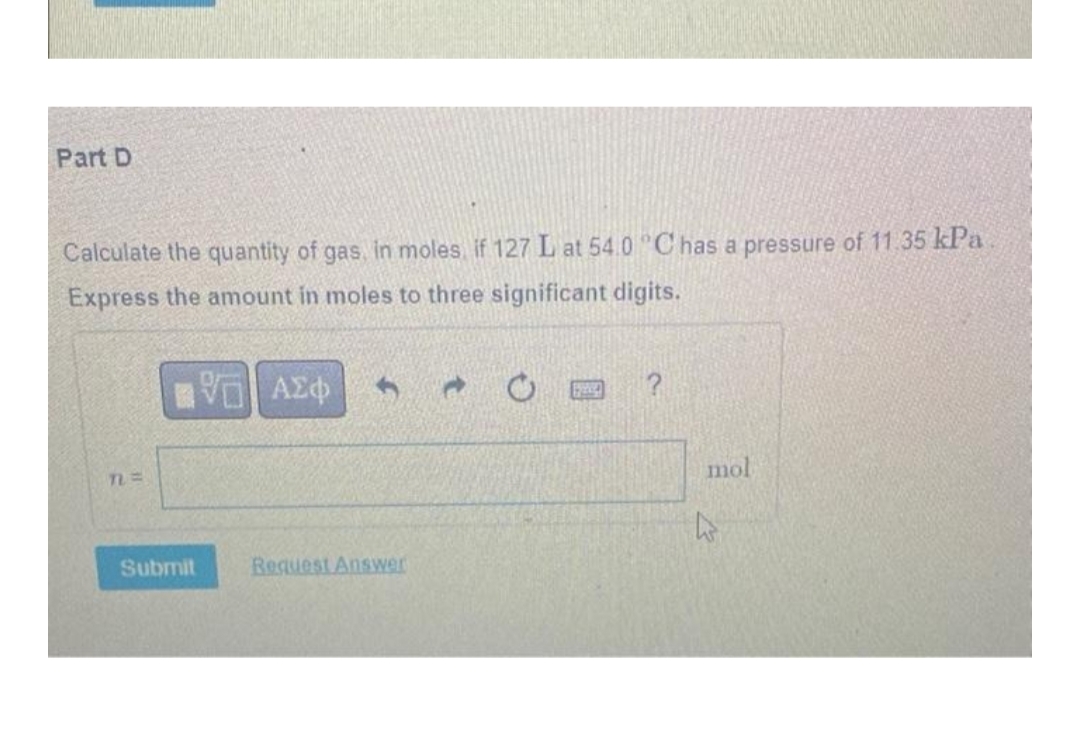
Transcribed Image Text:Part D
Calculate the quantity of gas, in moles, if 127 L at 54.0 °C has a pressure of 11.35 kPa
Express the amount in moles to three significant digits.
|| ΑΣΦ 3
TL=
mol
Request Answer
Submit
4
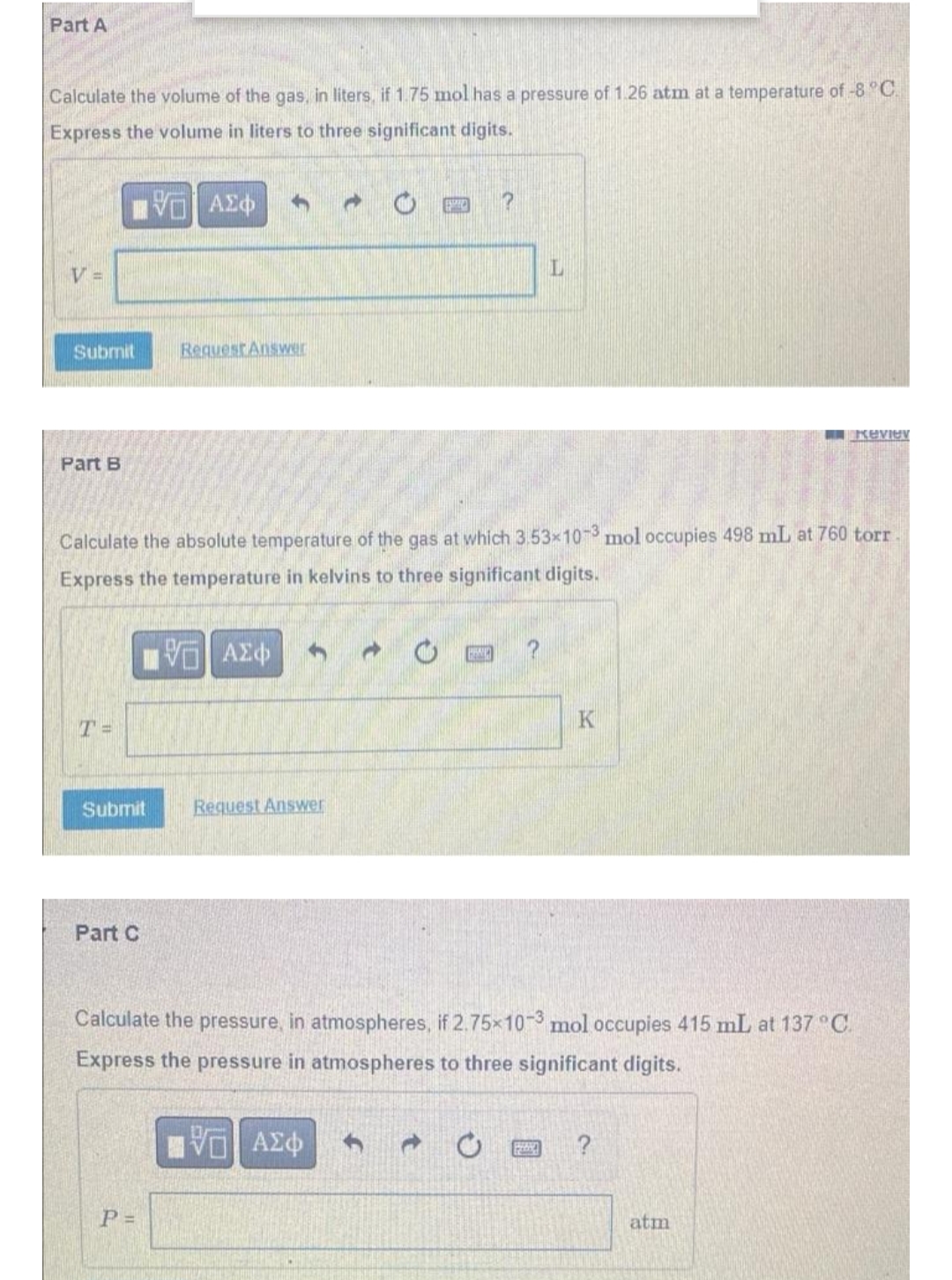
Transcribed Image Text:Part A
Calculate the volume of the gas, in liters, if 1.75 mol has a pressure of 1.26 atm at a temperature of -8 °C.
Express the volume in liters to three significant digits.
VD ΑΣΦ h
CE
?
V
L
Submit
Request Answer
Review
Part B
Calculate the absolute temperature of the gas at which 3.53x10-3 mol occupies 498 mL at 760 torr
Express the temperature in kelvins to three significant digits.
VE ΑΣΦ
T =
K
Submit
Request Answer
Part C
Calculate the pressure, in atmospheres, if 2.75x10-3 mol occupies 415 mL at 137 °C.
Express the pressure in atmospheres to three significant digits.
VE ΑΣΦ
?
P =
atm
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning