Part A If the student collected 3.40 g of acetone and the theoretical yield is 3.67 g. what is the percent yield? ? percent yield = Submit Previous Answers Request Answer X Incorrect; Try Again
Part A If the student collected 3.40 g of acetone and the theoretical yield is 3.67 g. what is the percent yield? ? percent yield = Submit Previous Answers Request Answer X Incorrect; Try Again
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter34: Particle Size Determination
Section: Chapter Questions
Problem 34.12QAP
Related questions
Question
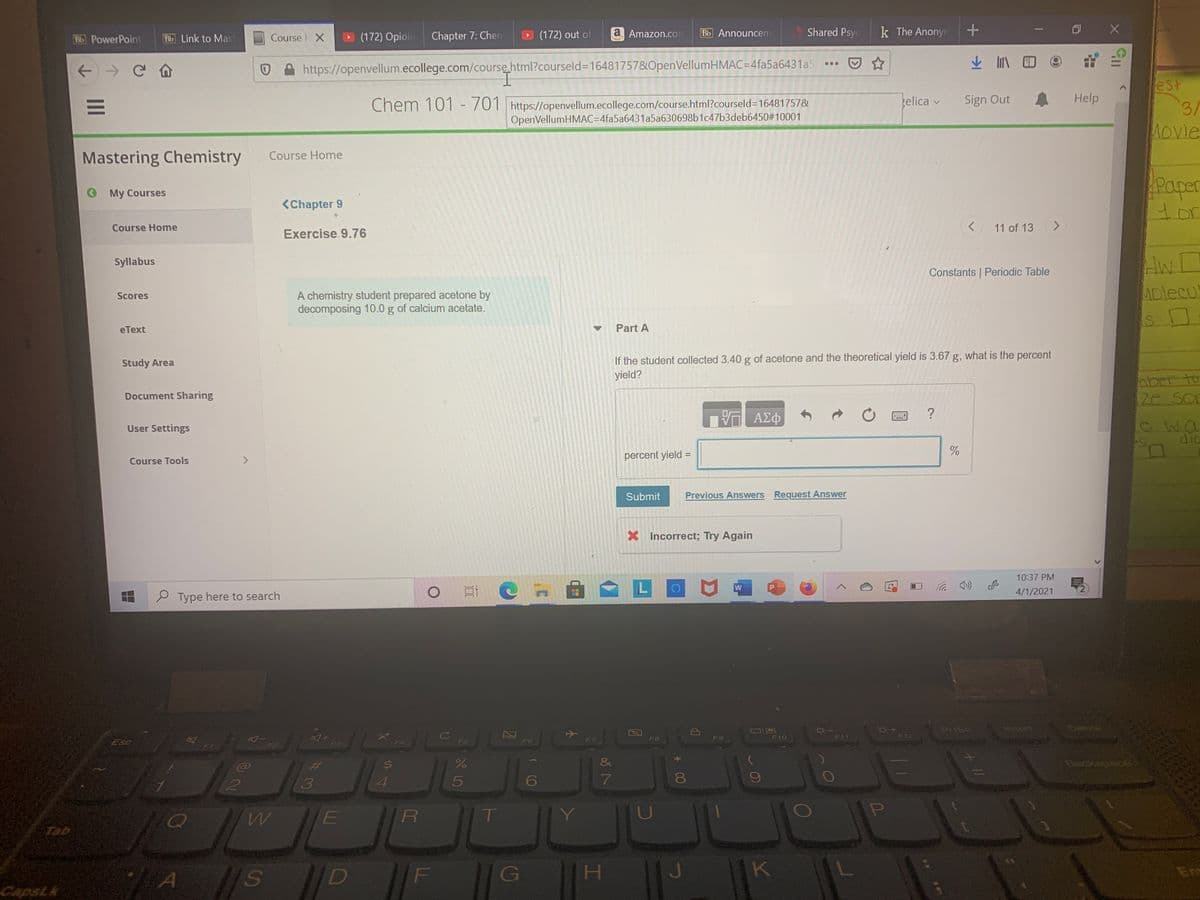
A chemistry student prepared acetone by decomposing 10.0g of calcium acetate.
if the student collected3.40g of acetone and the theoretical yield is 3.67g, what is the percent yield?
percent yield=
Transcribed Image Text:Course X
D
(172) Opioic
Chapter 7: Chen
(172) out of
a Amazon.con
Bb Announcem
Shared Psyc
k The Anonyn
86 PowerPoint
Bb Link to Mas
...
https://openvellum.ecollege.com/course.html?courseld=D16481757&OpenVellumHMAC=4fa5a6431a5
est
3/
Movie
kelica
Sign Out
Help
Chem 101-701 https://openvellum.ecollege.com/course.html?courseld=16481757&
OpenVellumHMAC=4fa5a6431a5a630698b1c47b3deb6450#10001
Mastering Chemistry
Course Home
Paper
My Courses
<Chapter 9
Course Home
11 of 13
<.
Exercise 9.76
Hw
ADleco
Syllabus
Constants | Periodic Table
A chemistry student prepared acetone by
decomposing 10.0 g of calcium acetate.
Scores
eText
Part A
If the student collected 3.40 g of acetone and the theoretical yield is 3.67 g, what is the percent
yield?
Study Area
nber to
ze sor
Document Sharing
ΑΣφ
...
User Settings
dic
%
percent yield =
%3D
Course Tools
Submit
Previous Answers Request Answer
X Incorrect; Try Again
10:37 PM
4/1/2021
P Type here to search
PriSc
Deleie
F9
F10
F11
F12
F8
Esc
FS
F6
JU
%23
&
3
R
Tab
IK
F
S
D
CapsLk
00
24
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning