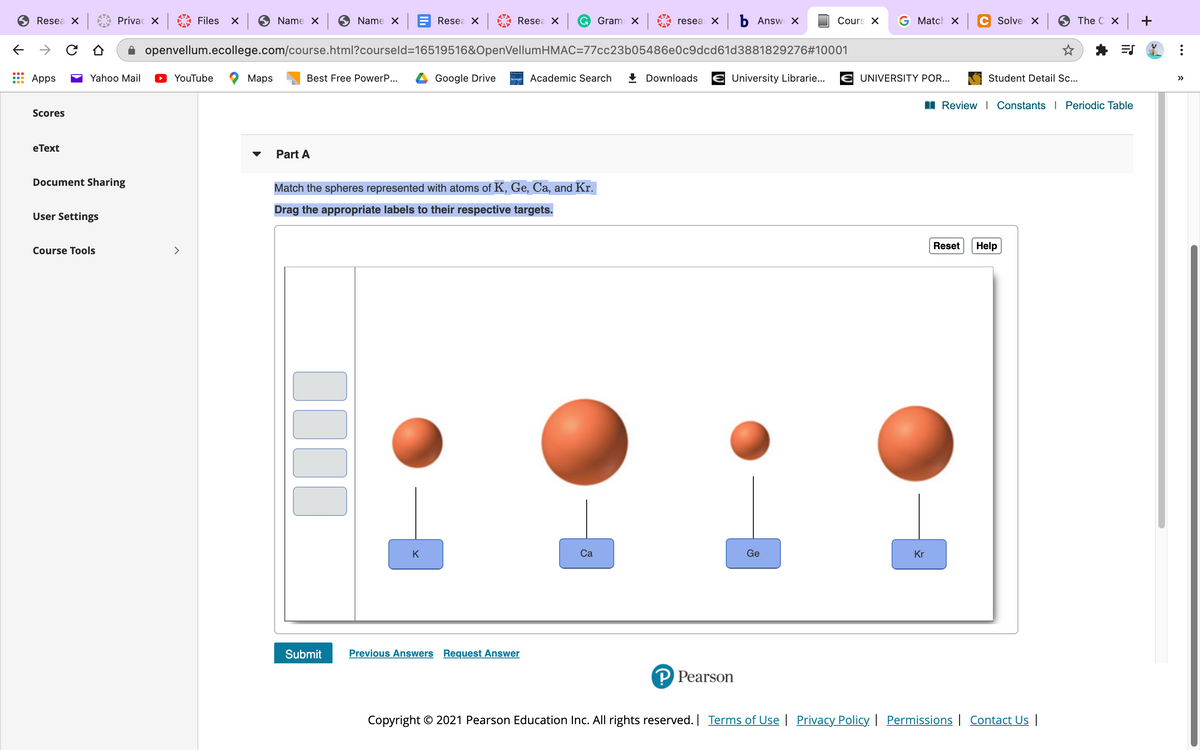
Part A Match the spheres represented with atoms of K, Ge, Ca, and Kr. Drag the appropriate labels to their respective targets. Reset Help K Ca Ge Kr
Part A Match the spheres represented with atoms of K, Ge, Ca, and Kr. Drag the appropriate labels to their respective targets. Reset Help K Ca Ge Kr
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter12: Thermodynamic Processes And Thermochemistry
Section: Chapter Questions
Problem 81AP
Related questions
Question
Match the spheres represented with atoms of K, Ge, Ca, and Kr.
Drag the appropriate labels to their respective targets.
Transcribed Image Text:Resea X
Privac X
Files
Name X
Name X
Resea X
Resea X
G Grami X
resea X b Answ X
Cours X
G Match x
Solve X
The C X
+
openvellum.ecollege.com/course.html?courseld=16519516&OpenVellumHMAC=77cc23b05486e0c9dcd61d3881829276#10001
Apps
Yahoo Mail
YouTube
Мaps
Best Free PowerP...
Google Drive
Academic Search
+ Downloads
University Librarie...
E UNIVERSITY POR...
Student Detail .c..
>>
Review I Constants I Periodic Table
Scores
eТext
Part A
Document Sharing
Match the spheres represented with atoms of K, Ge, Ca, and Kr.
Drag the appropriate labels to their respective targets.
User Settings
Course Tools
<>
Reset
Help
K
Ca
Ge
Kr
Submit
Previous Answers Request Answer
P Pearson
Copyright © 2021 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy Policy. | Permissions | Contact Us |
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning