Part A The following data show the rate constant of a reaction measured at several different temperatures. Use an Arrhenius plot to determine the activation barrier for the reaction. Temperature (K) Rate Constant (s~} Express your answer using three significant figures. 310 0.125 320 0.365 330 0.995 Jemplates Symbols uado redo resət keyboard shortcuts help 340 2.56 Ea = kJ mol- 350 6.23 Submit Request Answer • Part B Use an Arrhenius plot to determine the frequency factor for the reaction. Express your answer using two significant figures. Temp@es Symbols uado redo resət keyboard shortcuts help A =
Part A The following data show the rate constant of a reaction measured at several different temperatures. Use an Arrhenius plot to determine the activation barrier for the reaction. Temperature (K) Rate Constant (s~} Express your answer using three significant figures. 310 0.125 320 0.365 330 0.995 Jemplates Symbols uado redo resət keyboard shortcuts help 340 2.56 Ea = kJ mol- 350 6.23 Submit Request Answer • Part B Use an Arrhenius plot to determine the frequency factor for the reaction. Express your answer using two significant figures. Temp@es Symbols uado redo resət keyboard shortcuts help A =
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter12: Kinetics
Section: Chapter Questions
Problem 65E: The hydrolysis of the sugar sucrose to the sugars glucose and fructose, C12H22O11+H2OC6H12O6+C6H12O6...
Related questions
Question
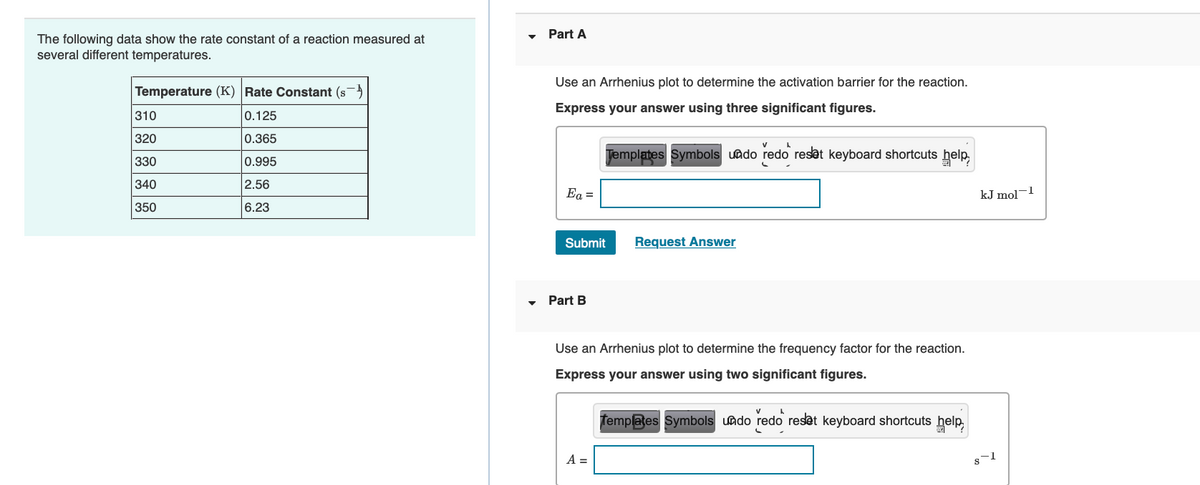
Transcribed Image Text:Part A
The following data show the rate constant of a reaction measured at
several different temperatures.
Use an Arrhenius plot to determine the activation barrier for the reaction.
Temperature (K) Rate Constant (s)
Express your answer using three significant figures.
310
0.125
320
0.365
Jemplates Symbols uado redo reset keyboard shortcuts help,
330
0.995
340
2.56
Ea =
kJ mol-1
350
6.23
Submit
Request Answer
Part B
Use an Arrhenius plot to determine the frequency factor for the reaction.
Express your answer using two significant figures.
Templates Symbols uado redo reset keyboard shortcuts help,
A =
s-1
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning