Q: 4. Which of the following arises from π to π* transitions? Select all that apply.
A: π to π* transition is observed in UV spectrum. Therefore only option 5th is correct.
Q: Q-3. Provide a structure consistent with the set of spectra provided. - Interpret as many MS…
A: The provided mass number(147) is odd means odd number of Nitrogen may be present. In the mass…
Q: 4.8 0 0.8 0.6 0.4 0.2 20 4000 40 3000 INFRARED SPECTRUM 2000 Wavenumber (cm-1) 42 MASS SPECTRUM 60…
A: From the above given data,we can assume that the compound is bromoethene ( CH2=CHBr ).
Q: 2.5 3 7 8 9 10 12 14 16 20 24 100 80 60 40 20 4000 3500 3000 2600 2200 2000 1800 1600 1400 1200 1000…
A: In Stretching vibrations , the distance between two atoms are increases or decreases but atoms…
Q: Help
A:
Q: Identify some characteristic group frequencies in the spectrum of an organic molecule shown below…
A: Infrared spectroscopy is used to identify the different types of functional groups present in the…
Q: Using a 60 MHz spectrometer, a chemist observes the following peak: doublet, J = 7 Hz, at 4.00 ppm…
A: Given: At 60MHz, Peak is doublet. Coupling constant J = 7Hz Chemical shift = 4.00 ppm To find:…
Q: 4. For the compounds below, fill in the charts of expected integration, multiplicity, and chemical…
A: The same kind of hydrogens will give give same peak and the hydrogens in different environment will…
Q: The functional group region of an infrared spectrum is greater than or equal to 1500 cm-1. True…
A:
Q: 10. The mass spectrum of a C, H, O containing compound is shown below. Use the Rule of 13 to…
A:
Q: 2. The data below correspond to a molecule with the formula C10H12O Use the data to solve the…
A: It's a multiple part question type. The above mentioned question is explained below:
Q: 100 HS-NU-SS06 80 40 20 10 20 30 40 50 60 70 80 90 100 110 120 m/z Relative Intensity
A: The natural abundance of 13C is 1.1%. The number of carbon atoms present in the given molecule can…
Q: A proton has resonance at 90Hz downfield from TMS when the field strength is 1.41 T(14,100 Gauss)…
A: Chemical shift=90HzInitial Field strength=1.41TOscillator frequency=60MHzFinal field…
Q: What information is obtained from the mass spectrum and IRspectrum of an unknown compound X? Assume…
A: Base peak – It is the most intense or tallest peak. Molecular ion peak – It is the heaviest ion peak…
Q: Determine the structure based on the following spectrum: 100 MS-NW-0660 80 60 40 20 0 mt 10 20 30 40…
A: Normal/ straight chain alkanes mass spectra generally contains lots of common peaks but it must have…
Q: Identify the structure of compound A (molecular formula C9H10O) fromthe 1H NMR and IR spectra given
A:
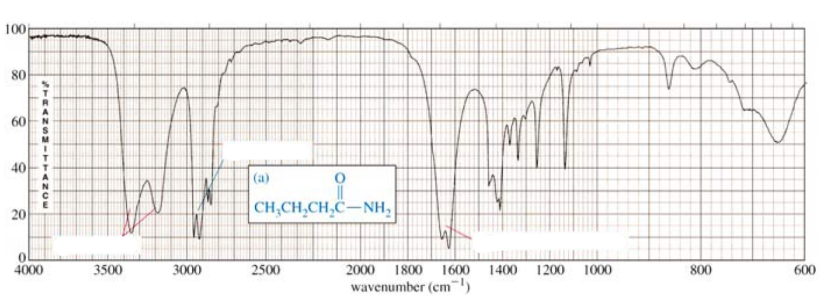
Q: 100 80 60 40 (a) 20 CH;CH,CH,C-NH, 4000 3500 3000 2500 2000 1800 1600 1400 1200 1000 800 600…
A:
Q: Benzoic acid Benzoic acid Infrared Spectrum 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0.0 3500 3000…
A: FTIR values of benzoic acid:
Q: 1. The infrared spectrum of ethyl acetate is shown below. Classify the functional groups based on…
A: Infrared spectrum helps in identifying all the functional groups in the sample based on their…
Q: 2- The chemical shift value of o-nitro toluene is higher than that of m-nitro toluene.
A: 2) In 1HNMR spectroscopy , the substituents, such as -NO2 can act as an electron withdrawing group…
Q: 100 80 60 40 CH-C-(CH2),CH3 20 (а) 2-heptanone 1718 4000 3500 3000 2500 2000 1800 wavenumber (cm)…
A:
Q: Benzoic acid Benzoic acid Infrared Spectrum 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0.0 3500 3000…
A: Functional group present for each band in the FTIR spectra of benzoic acid - The presence of strong…
Q: Following are infrared spectra of 2-methyl-1-butanol and tert-butyl methyl ether. Assign each…
A:
Q: Provide a structure consistent with the set of spectra Q-5. provided. - Interpret the key MS…
A: IR spectrum: A peak at 2960 cm–1 indicating the presence of C–H stretching absorption for alkyl…
Q: m/z Intensity M* 120 100 d = 0.91 ; triplet, 3 H 121 5.5 d = 1.12; doublet, 3H 122 32 d = 1.73;…
A: MASS SPECTROSCOPY Mass spectroscopy identifies the mass of a given compound by calculating the mass…
Q: Where are you likely to find a new peak related to the product of this reaction? „Br Na* NH2 3500…
A: NaNH2 is a strong base and it converts alkyl halides to alkenes.
Q: MOLECULE #3 INFRARED SPECTRUM 4000 1000 MAVENERI ELEMENTAL ANALYSIS: C, 77.75; H, 7.46; 0, 14.80…
A: Spectroscopy deals with the interaction of electromagnetic radiations with matter. Every compound…
Q: 4000 100 80 20 3284 % of base peak IR Spectrum (KBr disc) 3000 Japan 13C NMR Spectrum 2000 1658 1600…
A: Molecular formula C10H13NO2
Q: What if the functional group and wave number for each functional group In the IR SPECTRUM attached…
A: 4-methoxybenaldehyde IR frequency range 3000-3100 cm-1 peaks is represent as sp2 =C-H stretch And…
Q: 2-Octanone INFRARED SPECTRUM 0.8 0.6 0.4 0.2 3000 2000 1000 Wavenumber (cm-1) ST Chemistry WehRook…
A: IR spectroscopy is the technique which is used to determine the functional groups present in the…
Q: 100 80 60 40 20 % of base peak 40 45 59 V (cm¹) M+ = 60 (<1%) 120 80 160 m/e 800 Mass Spectrum 1200…
A:
Q: IR Spectrum (liquid fim) 2984 1741 1243 4000 3000 2000 1600 1200 800 V (cm") 100 Mass Spectrum 43 80…
A: 1H NMR: Number of signals -------> Number of sets of protons in different electronic environment…
Q: For preblems you ere net expected to come up with an exaet strueture. Use the provided data to…
A:
Q: Out of the indicated hydrogen molecules, which one would you expect to have the greatest downfield…
A: The hydrogen atom attached to the electron withdrawing atom or group is said to be deshielded and…
Q: How to get coupling constants from this spectra. Values at the top are in ppm. The instrument is 300…
A: Frequency of instrument = 300 MHz Coupling constant (J) = ?
Q: You're in lab and there's a bottle of solvent that has a faded label. You want to dispose of it…
A: IR spectrum is a very good tool to detect which functional group a compound have.
Q: Following are infrared spectra of methylenecyclopentane and 2,3-dimethyl-2-butene. Assign each…
A: First IR-spectra is due to methylene cyclopentane. IR absorption at around: 3080-3100 cm-1 is due…
Q: Find the Correlation Chart for Functional Group Assignments in Infrared Spectra Compound:…
A:
Q: 1. Identify the correct structure for the given IR spectrum. 100T 50 2000 500 1500 1000 4000 3000…
A: IR spectrum is mainly used to identify the functional group present in the compound. It measures the…
Q: chloromethane Difluoromethane methylene chloride methyl chloride compounds difluoroamine MW (g/mol)…
A: Molecular peak : in mass spectrum , the peak which represents the molecular ion , is called…
Q: -아 (a) CH;CH,CH,C-NH, 4000 3500 3000 2500 2000 1800 1600 1400 1200 1000 800 600 wavenumber (cm)
A:
Q: 100 80 60 1-butanol CH3(CH2);OH %3D 40 20 4000 3500 3000 2500 2000 1800 1600 1400 1200 1000 800 600…
A: The IR spectra help to recognize the functional groups present in the molecules. These groups can be…
Q: Compound 2 IR Spectra: INFRARED SPECTRUM 0.8- 0.4 2000 Wavenumber (cm-1) 3000 1000 Interpretation of…
A: The IR spectroscopy only tells about the functional group present in the Molecule . SO For complete…
Q: Molecule #15 INFRARED SPECTRUM VENERat Elemental Analysis: C, 57.84; H, 3.64; 0, 38.52 Molecule #15…
A: From the data given by elemental analysis, the percentage of the elements are, Carbon- 57.84%…
Q: Mass Spectrum 100- 1H NMR Spectrum 6H M (74) = 100% M+1 (75) = 4.52 M+2 (76) = 0.28 doublet 80- 2H…
A:
Q: A compound with molecular formula CgH100 gives the following IR spectrum. 100 80 60 1650 40 20 1720…
A:
Q: 100 80 1660 60 H H. 40 3023 H;C (CH,),CH3 20 (c) cis-2-octene 4000 3500 3000 2500 2000 1800 1600…
A: Fourier-transform infrared spectroscopy is a technique used to obtain an infrared spectrum of…
Q: Use the 1H NMR and IR spectra given below to identify the structures of two isomers (A and B) having…
A: Given,
Q: PPM Key Peaks m/z = 27, 43, 107, 109, 150, 152 50 100 150 m/z Br Br-
A: The question is based on the concept of Spectroscopy. we have to analyse the spectra provided and…
Q: 12. An Organic Compound with the Following Spectra (you need to determine what atoms are present)…
A: The structure I have elucidated as per the given experimental data is butanal. C4H8O Degree of…
5. Identify
Step by step
Solved in 2 steps with 1 images

- Why is it reasonable that the Ksp of NiCl2 should be so high? given: NiCl2~~>Ni2+(aq)+2Cl-(aq) ksp=497.504 ksp=[Ni2+][Cl-]^2 [Ni2+]=4.992 [Cl-]=9.983A photon of wavenumber 100 cm-1 has a wavelength ofCalculate the (a) Linearity constant (r), (b) y-intercept, (c) Slope, and (d) Protein content of an unknown sample having an absorbance of 0.325.
- Principle of fluorescence spectroscopyProvide the significance of the signals 113 and 141 m/z, and what is the indication of these signal regarding the fragment?Calculate the wavenumber (cm^-1) and excitation energy (kj/mol) for each of the following max wavelengths: a.) 327.6 nm b.) 452.98 nm c.) 579.1 nm
- a. If two signals differ by 1.5 ppm in a 300 MHz spectrometer, by how much do they differ in a 500 MHz spectrometer? b. If two signals differ by 90 Hz in a 300 MHz spectrometer, by how much do they differ in a 500 MHz spectrometer?Calculate the resolution required to resolve the following peaks; i) CH3N+ (MW = 28.0187) and N2+ (MW = 28.0061) ii) C2H4+ (MW = 28.0313) and CO+ (MW = 27.9949) iii) C3H7N3+ (MW = 85.0641) and C5H9O+ (MW = 85.0653) iv) 116Sn+ (At W = 115.90219) and 232Th2+ (At W = 232.03800)The magnetogyric ratio of 19F is 2.5177 x 108 T-1 s-1. Calculate the frequency of the nuclear transition in a field of 7.500 T.
- How to get coupling constants from this spectra. Values at the top are in ppm. The instrument is 300 MHz.Compute for the concentration of an alcoholic solution of Methylene blue with an absorbance reading of 0.6681. A standard calibration was done which yielded the following result: Conc ABS 3.000 ppm 0.2099 7.000 ppm 0.4421 15.000 ppm 0.7892 20.000 ppm 0.9931 Choices: a. 12.559ppm c. 14.559ppm b. 13.559ppm d. 11.559ppmWhat is the main particularity of a MS signal for a molecule having: An enriched level of 13C atoms (up to 50/50 12C/13C)?

