pH 1X10 7. Consider the titration curve for the titration of 15 ml of Na,CO, as Na,A with 0.T 12 loa se 7 10 AzA Na0 A 4. 10 15 20 25 30 35 Va (mL) Which specie exist at point C? A2 a. pK2 ? b- Which species exist where pOH =
pH 1X10 7. Consider the titration curve for the titration of 15 ml of Na,CO, as Na,A with 0.T 12 loa se 7 10 AzA Na0 A 4. 10 15 20 25 30 35 Va (mL) Which specie exist at point C? A2 a. pK2 ? b- Which species exist where pOH =
Chapter8: Polyfunctional Acids And Bases
Section: Chapter Questions
Problem 10P
Related questions
Question
You need to use the chart to solve A and B. If you can't solve both could you answer part B? Thank you
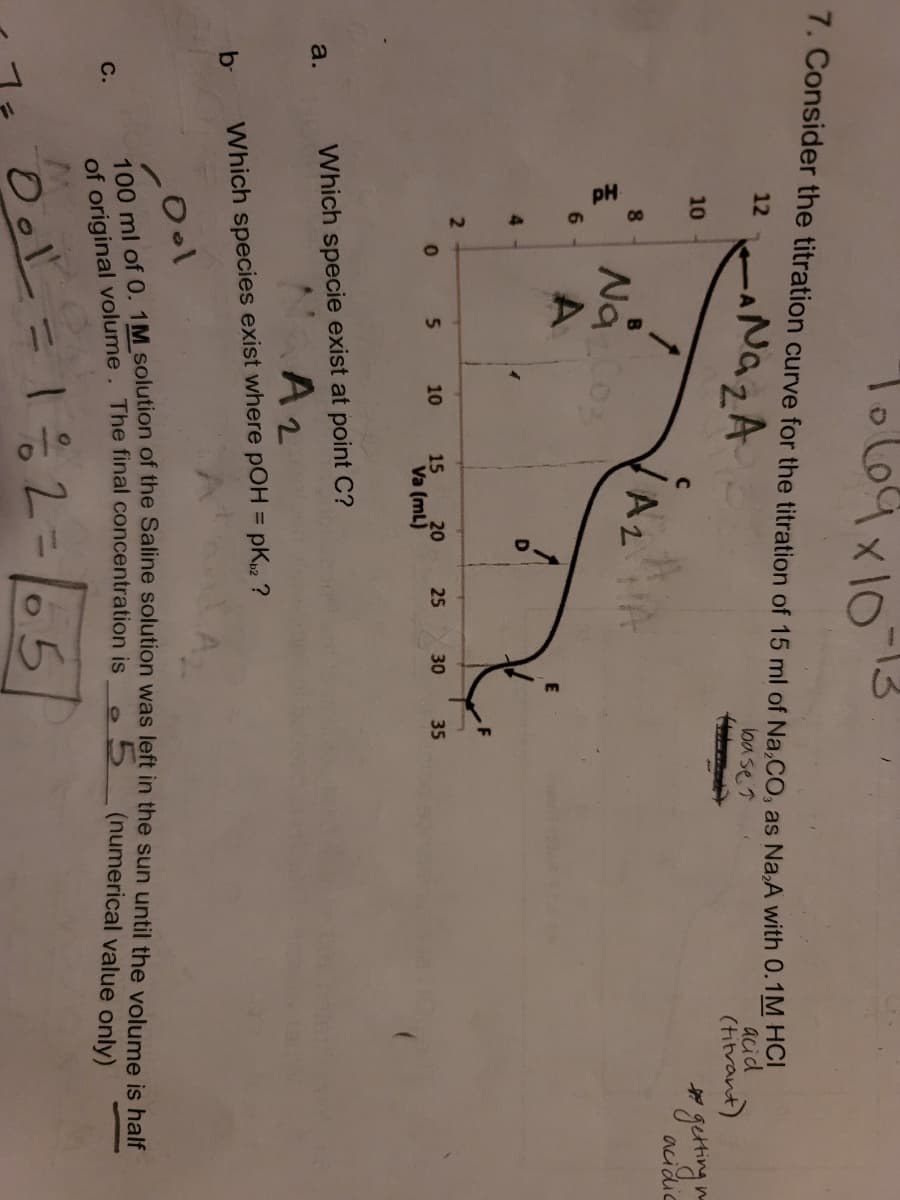
Transcribed Image Text:L69x10
13
7. Consider the titration curve for the titration of 15 ml of Na,CO, as Na,A with 0.TM HO
NazA
12
acid
(hitrant)
loa se 7
10
acidic
8.
Nao
6.
4.
5.
10
15
20
25
30
35
Va (mL)
Which specie exist at point C?
a.
A2
%3D
b-
Which species exist where pOH = pK2 ?
5.
(numerical value only)
С.
of original volume. The final concentration is
Ool
12=65
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning



Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning