Phosphate buffered saline (PBS) is a physiological buffer often used in biology experiments. Because the concentrations are very low and small amounts are difficult to weigh out, it is often prepared as a 10X stock solution meaning that it is prepared at 10 times the working concentration which is 1X. 1X PBS is made up of Na2HPO4 10 mM (MW 141.9 g.mol-1); KH2PO4 8 mM (MW 136.08 g.mol-1), NaCl 137 mM (MW 58.44 g.mol-1) and KCl 74.5 mM (MW 74.5 g.mol-1). You decide to prepare a 1 L 10X solution of PBS. What is the Molar (M) concentration of each of the components in you 10X stock. Write your answers in the table below. How much of each of the components should you weigh out to prepare 1L of the 10X stock solutions. Write your answers in the table below.
Phosphate buffered saline (PBS) is a physiological buffer often used in biology experiments. Because the concentrations are very low and small amounts are difficult to weigh out, it is often prepared as a 10X stock solution meaning that it is prepared at 10 times the working concentration which is 1X. 1X PBS is made up of Na2HPO4 10 mM (MW 141.9 g.mol-1); KH2PO4 8 mM (MW 136.08 g.mol-1), NaCl 137 mM (MW 58.44 g.mol-1) and KCl 74.5 mM (MW 74.5 g.mol-1). You decide to prepare a 1 L 10X solution of PBS. What is the Molar (M) concentration of each of the components in you 10X stock. Write your answers in the table below. How much of each of the components should you weigh out to prepare 1L of the 10X stock solutions. Write your answers in the table below.
Chapter17: Introduction To Iv Therapy
Section: Chapter Questions
Problem 3.6P
Related questions
Question
Phosphate buffered saline (PBS) is a physiological buffer often used in biology experiments. Because the concentrations are very low and small amounts are difficult to weigh out, it is often prepared as a 10X stock solution meaning that it is prepared at 10 times the working concentration which is 1X. 1X PBS is made up of Na2HPO4 10 mM (MW 141.9 g.mol-1); KH2PO4 8 mM (MW 136.08 g.mol-1), NaCl 137 mM (MW 58.44 g.mol-1) and KCl 74.5 mM (MW 74.5 g.mol-1).
- You decide to prepare a 1 L 10X solution of PBS. What is the Molar (M) concentration of each of the components in you 10X stock. Write your answers in the table below.
- How much of each of the components should you weigh out to prepare 1L of the 10X stock solutions. Write your answers in the table below.
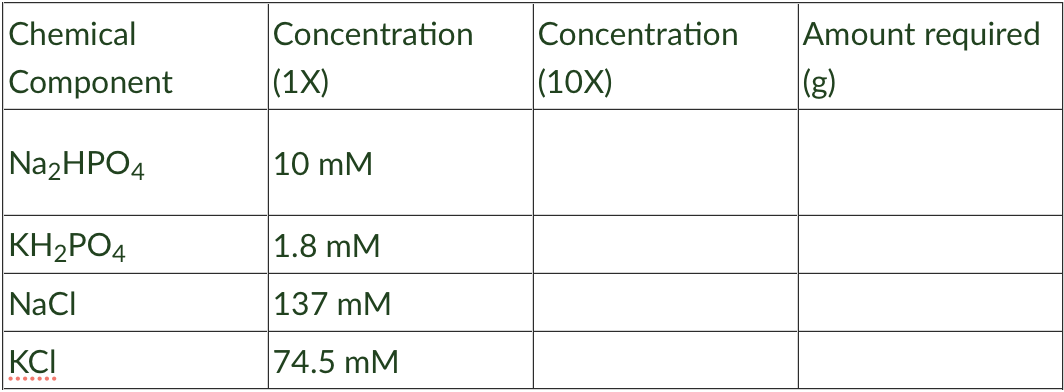
Transcribed Image Text:Chemical
Concentration
Concentration
Amount required
Component
|(1X)
|(10X)
|(g)
Na2HPO4
10 mM
KH2PO4
1.8 mM
NaCI
137 mM
KCI
|74.5 mM
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning



Principles Of Radiographic Imaging: An Art And A …
Health & Nutrition
ISBN:
9781337711067
Author:
Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:
Cengage Learning

