photoelectron spectra of two different elements are shown below. 60 50 40 30 Binding Energy (MJ/mol) 20 10 O Identify which element each spectrum corresponds to and explain your reasoning. electrons in that group) O Identify which orbital(s) the leftmost peak in each spectrum corresponds to. Explain why the navy peak has a larger binding energy than the magenta peal (Hint: the height of each peak is proportional to the number O Identify which orbital(s) the rightmost peak in each spectrum corresponds to Explain why the rightmost navy peak has a slightly smaller binding energy th the leftmost magenta peak. the Aufbau principle.) (Hint: think about what makes an exception Relative Number of Electrons
photoelectron spectra of two different elements are shown below. 60 50 40 30 Binding Energy (MJ/mol) 20 10 O Identify which element each spectrum corresponds to and explain your reasoning. electrons in that group) O Identify which orbital(s) the leftmost peak in each spectrum corresponds to. Explain why the navy peak has a larger binding energy than the magenta peal (Hint: the height of each peak is proportional to the number O Identify which orbital(s) the rightmost peak in each spectrum corresponds to Explain why the rightmost navy peak has a slightly smaller binding energy th the leftmost magenta peak. the Aufbau principle.) (Hint: think about what makes an exception Relative Number of Electrons
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter7: The Structure Of Atoms And Periodic Trends
Section: Chapter Questions
Problem 83SCQ
Related questions
Question
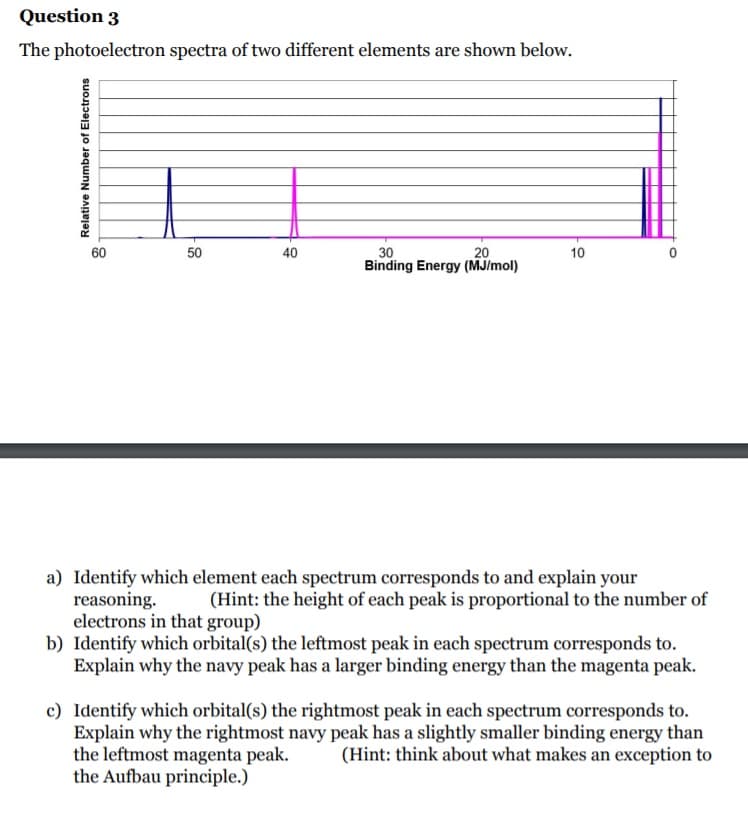
Transcribed Image Text:Question 3
The photoelectron spectra of two different elements are shown below.
60
50
30
Binding Energy (MJ/mol)
20
40
10
a) Identify which element each spectrum corresponds to and explain your
reasoning.
electrons in that group)
b) Identify which orbital(s) the leftmost peak in each spectrum corresponds to.
Explain why the navy peak has a larger binding energy than the magenta peak.
(Hint: the height of each peak is proportional to the number of
c) Identify which orbital(s) the rightmost peak in each spectrum corresponds to.
Explain why the rightmost navy peak has a slightly smaller binding energy than
the leftmost magenta peak.
the Aufbau principle.)
(Hint: think about what makes an exception to
Relative Number of Electrons
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning
