Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter6: Equilibria In Single-component Systems
Section: Chapter Questions
Problem 6.50E
Related questions
Question
100%
Please Answer all the following items will give thumbs up and good rating.
Matching type
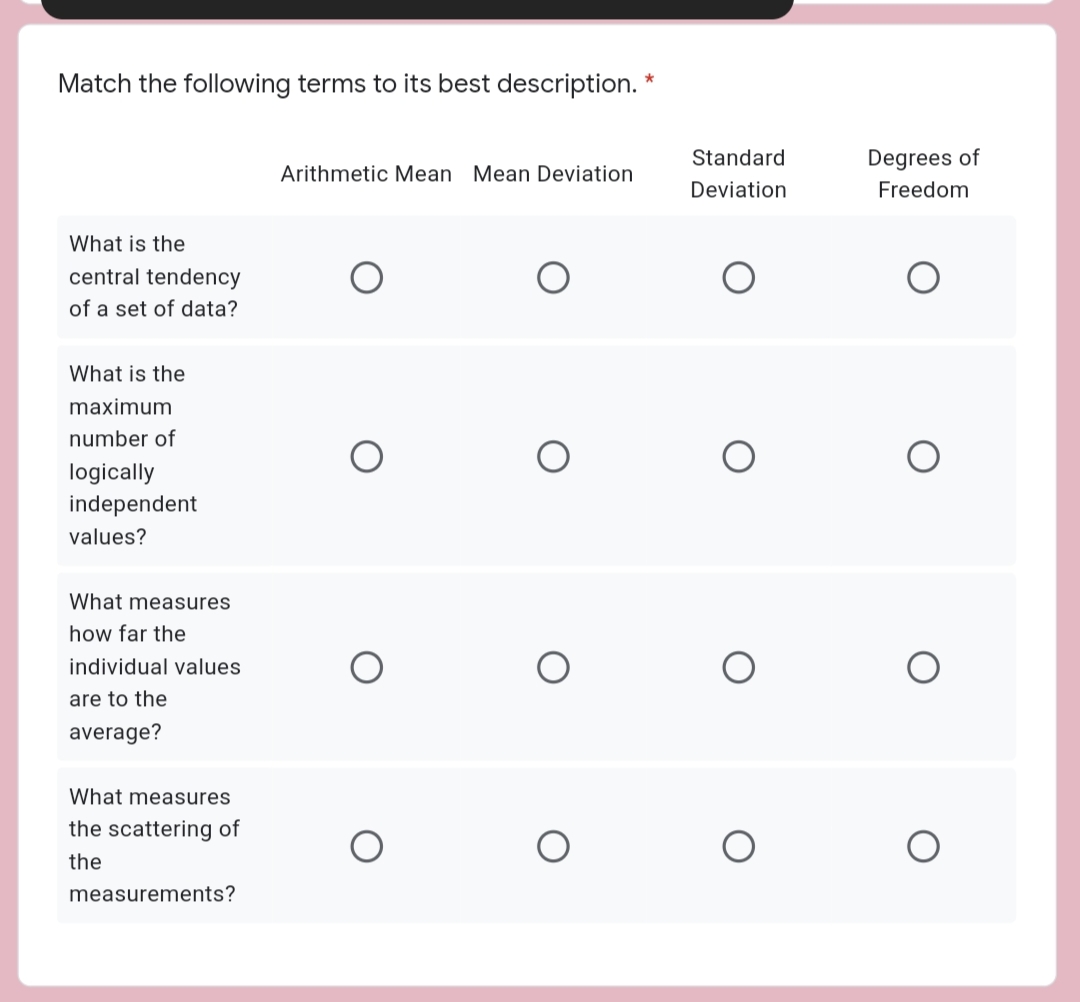
Transcribed Image Text:Match the following terms to its best description. *
Standard
Degrees of
Arithmetic Mean Mean Deviation
Deviation
Freedom
What is the
central tendency
of a set of data?
What is the
maximum
number of
logically
independent
values?
What measures
how far the
individual values
are to the
average?
What measures
the scattering of
the
measurements?
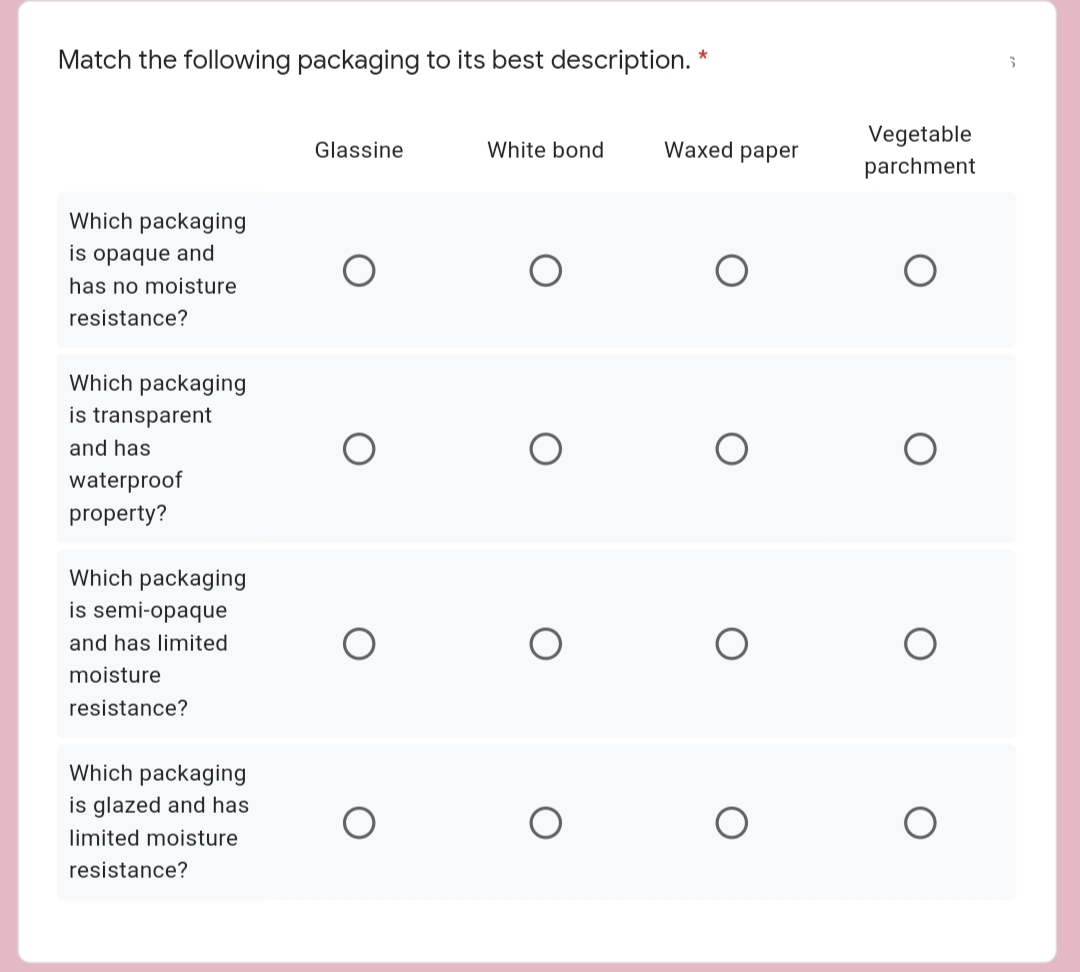
Transcribed Image Text:Match the following packaging to its best description. *
Vegetable
Glassine
White bond
Waxed paper
parchment
Which packaging
is opaque and
has no moisture
resistance?
Which packaging
is transparent
and has
waterproof
property?
Which packaging
is semi-opaque
and has limited
moisture
resistance?
Which packaging
is glazed and has
limited moisture
resistance?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,