Q: Which of the following contains a sulfur atom as part of the side chain? A) Glycine B) Proline C)…
A: ->We can understand it by drawing their structure
Q: When the nuclide bismuth-214 undergoes beta decay: The name of the product nuclide is The symbol for…
A:
Q: What is the absolute configuration of the molecule on the photo?
A: RS nomenclature: The RS nomenclature is an important nomenclature system for denoting the…
Q: This Diekmann cyclization proceeds through the expected five-membered ring transition state. Draw…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What products would be obtained fo the reactions below 1CHO, heat OCH, HSO +HNO CH,CH, (draw all…
A: We have to give the products of the given reactions. a) Here, in presence of the H2CrO4, heat, the…
Q: (1) The radioactive isotope strontium-81 is used in medical imaging as indicated on the table above.…
A: While writing nuclear reaction the sum of mass number on reactant side is equal to the sum of mass…
Q: How many molecules are in 3.1x10^-3 g of C9H8O4?
A:
Q: Write a balanced nuclear equation for the following: The nuclide boron-8 decays to beryllium-8 by…
A: Given :- boron-8 decays to beryllium-8 by positron emission To write :- balanced nuclear equation
Q: 2. There is 1250. cm3 of chlorine gas at 25°C and 90,000 Pascals, what volume will the same gas…
A:
Q: When the compounds analyzed by Robrob were passed on to the next test, another scientist, Kikoko,…
A: Given: Concentration of 20 % (w/w) solid active compound (M1) = ? Volume of 20 % (w/w) solid active…
Q: Consider the following electron configurations to answer the questions that follow: (i) 152 252 2p6…
A:
Q: WEGT (Hank-FM) operates at a radio frequency of 95.3 MHz 1. What is the wavelength of this radiation…
A:
Q: Consider the following decay series: d b. Bi Po C 214 210 Po Pb 218 a 214 214 Pb 82 84 82 83 84…
A:
Q: What amount of heat (in kJ) is required to convert 19.3 g of an unknown liquid (MM = 67.44 g/mol) at…
A: Here , heat is used in three steps , 1) in rising of temperature of liquid till boiling point. 2)…
Q: Which of the following describes the reaction: 218 Po →°,e+ 218 At 84 85 O bombardment reaction O…
A:
Q: For each chemical reaction listed in the first column of the table below, predict the sign (positive…
A:
Q: A Na,CO3 standard solution is prepared by transferring 2.4817 grams of primary standard-grade sodium…
A: Answer: This question is based on stoichiometric calculation where moles sodium carbonate will be…
Q: A student performs a calorimetry experiment. The student combines 100. g of 0.40 M HC1(aq) with 100.…
A: Heat absorbed by the particular species = mass × heat capacity × temperature change
Q: or the following reaction, AP(C6H14)/At was found to 6H14(9) → C6H6(g) + 4H2(g) etermine AP(H2)/At…
A: Which one of the following is correct
Q: Consider the following reaction: H2O2(aq) H20() + ½ O2(g) and the kinetics plots below to answer the…
A: The plots given are,
Q: KMNO4 NazCr20, [1] NACN p. d. HO, f. H2SO4, H2O Br (2] H2O, ¯OH
A: The reactions given are,
Q: Cl, 2 2C1- (6-12a) + CO CIĊO (6-12b) + Cle CIĄCO + Cl- (6-12c)
A:
Q: Write a balanced nuclear equation for the following: The nuclide boron-8 decays to beryllium-8 by…
A:
Q: I am not understand these problems. Can someone help?
A:
Q: Under normal room conditions (standard pressure at about 20°C), water will not boil, so we do not…
A: Given: Under standard pressure of 1 atm and room temperature of 20 oC, water does not boil. The…
Q: Some Radioactive Isotopes Useful in Medical Imaging Mode of Isotope Decay Half-life Use in Medical…
A: Both the parts have been solved in the following step.
Q: Name the major product of the reaction between (S)-2-bromohexane and KCH3COO
A: (S)-2-bromohexane is an alkyl halide and the acetate ion acts as a nucleophile. Nucleophilic…
Q: 900 800 - Carbon disulfide 700 600 Methano 500 Ethanol 400 Heptand 300- 200 100 - 20 30 40 50 60 70…
A: Vapour pressure is the pressure exerted by vapour over the surface of liquid at the condition of…
Q: A chemist fills a reaction vessel with 2.90 atm nitrogen monoxide (NO) gas, 6.95 atm chlorine (Cl,)…
A:
Q: What is the mass percent of an aqueous sodium hydroxide solution in which the mole fraction of NaOH…
A:
Q: Which of the following describes the reaction: 210 Bi + He + 206 TI 83 81 O alpha decay reaction O…
A: Which of the following describe the reaction--->
Q: 4. CH3CH2 CH2CH2CH2CH3 C=C H CH2CH3 CH2CH3 H3C CH3-CH-CEC-CH2-CH-CH3 CH3-CH2 CH2-CH3 5.
A:
Q: 3) Below are two possible means of synthesizing the following compound, cyclohexyl ethyl ether.…
A: The possible reactions given are,
Q: The equilibrium constant, K, for the following reaction is 1.77x10 3 at 568 K. NH4CI(s) NH3(g) +…
A: Kc is equilibrium constant and Kc is equal to ratio of concentration of product and concentration of…
Q: condensat ion HO-CH2-CH3 HO-C-CH2-CH3
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: How much heat is required to warm 2.07 kg of sand from 32.6 to 48.9 °C. The specific heat of sand is…
A:
Q: is the diphenylmethane generated from friedel-crafts reaction with DCM solid or liquid?
A: Reaction is as below- Diphenyl methane is obtained when excess benzene is treated with DCM in…
Q: 1. Beginning with the correct organic halide and benzophenone (C6HS C(0)C6H5) show how one would…
A: Detail mechanistic pathway is given below
Q: For each of the following, write a balanced chemical equation for each double displacement reaction.…
A: Since you have asked multiparts questions, we will solve the first three sub parts questions for…
Q: Question 1 Identify the correct statements consistent with the following scenario: 1. Heat is lost…
A: The correct statements have been marked in the following step.
Q: Which salts will be more soluble in an acidic solution than in pure water? Sn(OH)2 AgBr KClO4 CaCO3…
A: The fact to these is knowing what anions react with H^+ (the acid) and if any weak acids or…
Q: PQ-28. How are these molecules related? H. OH HO Br CH3 Br constitutional (D) isomers (A) identical…
A: Detail description is given below
Q: A student performs a calorimetry experiment. The student combines 100. g of 0.40 M HC1(aq) with 100.…
A: When the temperature of solution increases this means heat is released during the reaction and it is…
Q: 3. Write the equilibrium expression for the reaction, Zn 2(aq) + NH, (aq) → Zn (NH,), (aq) NH.CO,NH,…
A: At equilibrium, the rate of the forward and the reverse reaction is the same and the concentration…
Q: When the nuclide bismuth-214 undergoes beta decay: The name of the product nuclide is | The symbol…
A: Given :- nuclide bismuth-214 undergoes beta decay To determine :- name and symbol of product…
Q: What is the practical importance of extraction in relation to chemistry especially when it comes to…
A: Extraction is important for Isolating important materials from plants .
Q: What pressure is exerted by 25.0 g of neon gas at temperature of 25 °C if the original temperature…
A: Given-> Weight of Ne = 25.0 gm T2 = 25°C = 25 + 273 = 298 K T1 = 50.0°C = 50.0 + 273 = 323 K P1 =…
Q: Explain why each name is incorrect, and then write a correct name a. 1-Methylpropene b.…
A: Name of the organic compounds are done according to VSEPR theory.
Q: Consider the following half reactions in the attached image and their corresponding standard…
A: Given:
Q: Calculate the mass of O, that is dis
A:
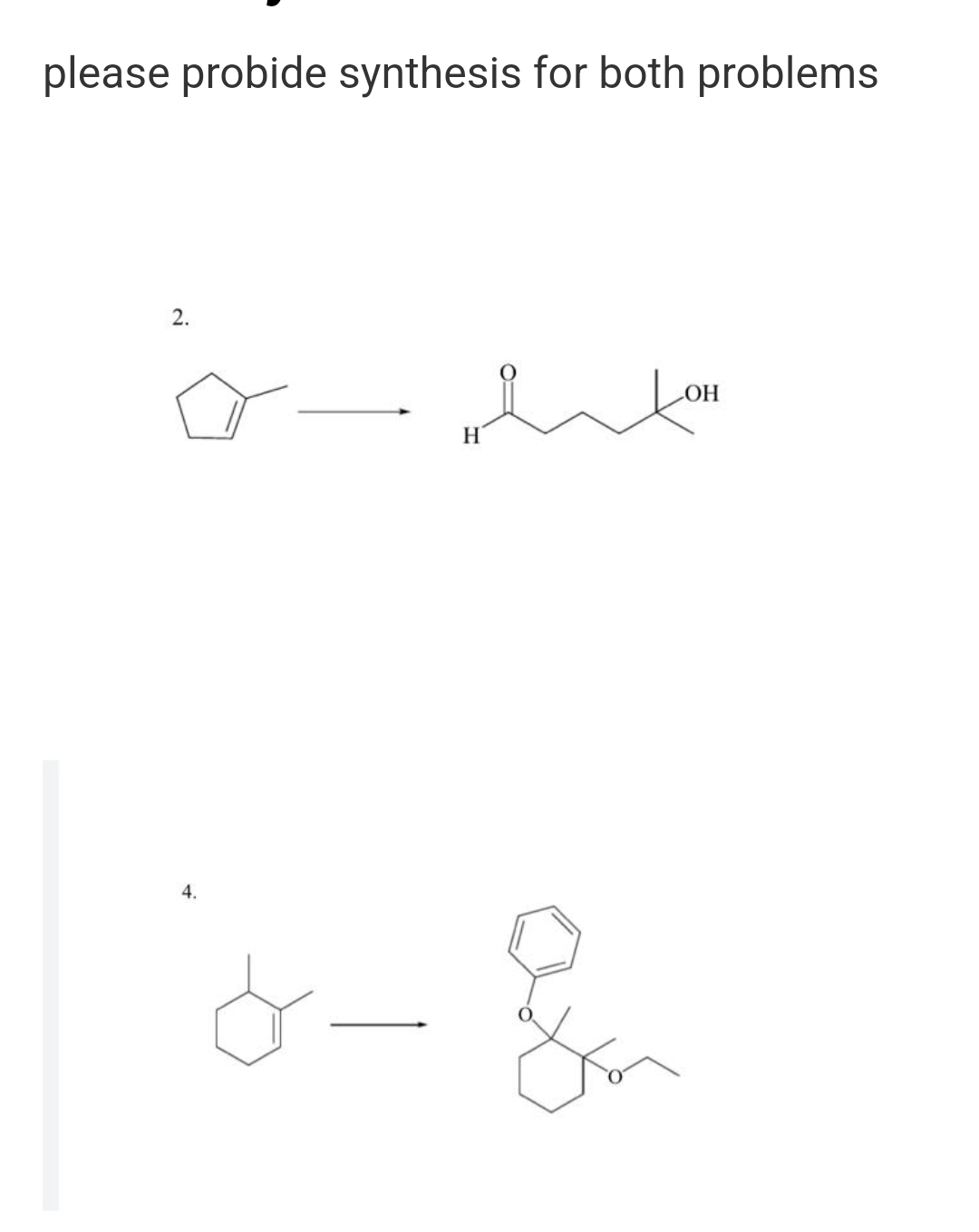
Step by step
Solved in 2 steps with 1 images

- Ochem... Predict the major product(s) for the following reaction sequence and provide the stepwise mechanism for both steps 1 and 2 (see attached image)(Organometallic) Give me 5 example of Associative (A) from these table , Because I want to understand please!ochem help Predict the major product(s) for the following reaction sequence and provide the stepwise mechanism for both steps 1 and 2. see attached image
- Answer is Reaction 7? Can you explain to me, please?Give reactent, products of all reactions subparts below. With appropriate wedge dash when possible.1. what reaction and meachanism is this ? 2. Explain mechanism what happenning in each step on this picture 3. why is it says "stop" when NaOEt added ? does reaction stop there ? why does it stop ? ? what principle ?

