Chapter8: Sampling, Standardization, And Calibration
Section: Chapter Questions
Problem 8.5QAP
Related questions
Question
100%
Plot the graph please
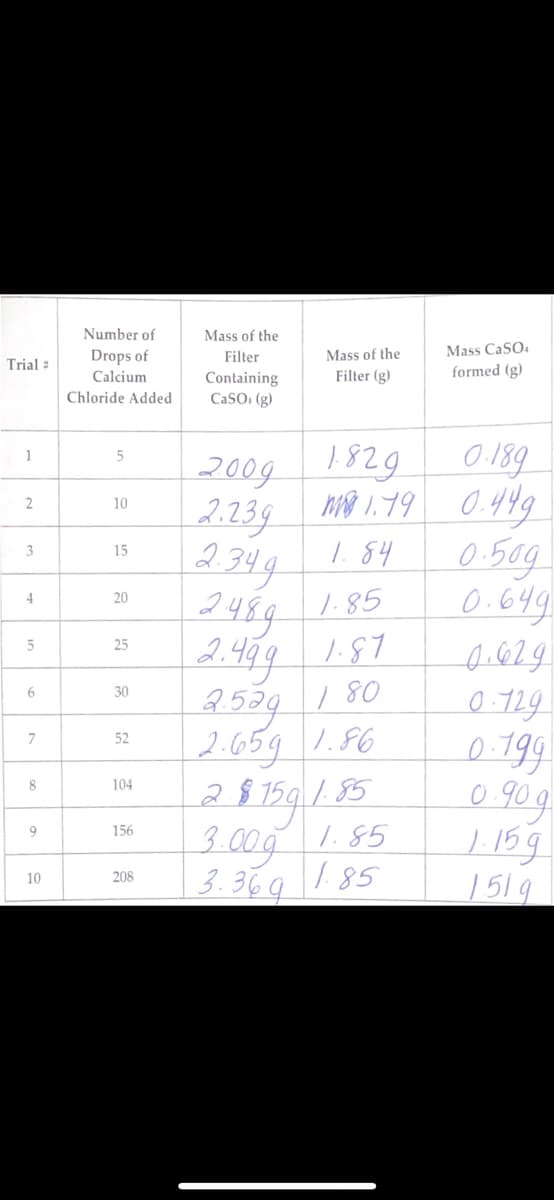
Transcribed Image Text:Trial :
1
2
3
4
5
6
7
8
o
9
10
Number of
Drops of
Calcium
Chloride Added
5
10
15
20
25
30
52
104
156
208
Mass of the
Filter
Containing
CaSO, (g)
Mass of the
Filter (g)
0.189
2009 1829
2.239 1.19 0.449
то
1.84
2.349
2489
1.85
2.499 1.87
1.80
2.539
2.659 1.86
Mass CaSO4
formed (g)
28759 1.85
3.00g
3.369 1.85
1.85
0.50g
0.649
0.629
0.729
0.199
0.90g
1.159
151g
Transcribed Image Text:0.9
(3) р
0.8
0.7
0.4
0.3
0.2
0.1
Calcium Sulfate Precipitated versus Number of Drops of Calcium Chloride Stock Solution added
MgSO, (aq) + CaCl, (aq) → CaSO, (s) + MgCl, (aq)
10 20 30 40
50 60 Overal page number 15 130 140 150 160
170
180 190 200 210
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



