Q: NO21 15. Formula: No: 2. 16. Name:
A: Note - Since you have asked multiple questions, we will solve the first question for you. If you…
Q: Thought Questions NOTE: Answer the following questions before the lab. The grade of these questions…
A: A question based on qualitative analysis, which is to be accomplished.
Q: 1-5. How do you make 0.5 L of pH 7, 0.1 M phosphate buffer (pKa=7.21) with NaH₂PO4 (MW 119.98) and…
A: pKa = 7.21 0.5 L of 0.1 M phosphate buffer = 0.5 L * 0.1 mole / L = 0.05 mole Now let mole of…
Q: Which of the equations is NOT CORRECT? A B D y2 - gR w? - GM R2 %3D y2 -GM y² _ GM w? -
A: D is not correct
Q: What is the [AsOg] for a solution labeled 0.10 M arsenic acid (H3ASO4)? [Ka1 = 6 x 103, Ka2 = 1 ×…
A:
Q: Pre-lab question #6-1: Calculate the percent (%) C2042- in H2C204. 53.0 57.1 65.7 97.8
A: Mass percent: Mass percent of ion = atomic mass of the ionMolar mass of the compound×100
Q: 7. Na2Cr207 HO, H2SO4 8. Но HIO4 OH
A: #7: H2CrO4(Na2CrO4+H2SO4) is a strong oxidizing agent and oxidizes primary alcohol to acid and…
Q: (a) How many grams of nickel are contained in 10.0 g of a 10.2 wt% solution of nickel sulfate…
A: Given : mass of solution = 10.0 g The mass of NiSO4.6H2O present in the solution is given by…
Q: Cesium chloride is used to prepare dense solutions required for isolating cellular components with a…
A: a) Cesium chloride is used to prepare dense solutions required for isolating cellular components…
Q: A lead compound would be most promising if it had: A.KM = 4.7 × 10^5 M. B.K(I) = 1.5 × 10^-8 M.…
A: The Lead Compound would be most promising if the K(I) value is the least among all given values . As…
Q: Br Method 1: 1) t-BUOK 2) H30 3) PCC 4) MeMgBr 5) H20 6) PCC Method 2: 1) NaOH 2) PCC 3) MeMgBr 4)…
A:
Q: 10-27. A 10.231-g sample of window cleaner containing ammonia was diluted with 39.466 g of water.…
A:
Q: A 95.0 wt% solution of ethanol (CH3CH2OH, FM 46.07) in water has a density of 0.804 g/mL. (a) Find…
A: Density is defined as the mass per unit volume. Its units are g/mL or kg/L. Molarity is a way of…
Q: Calculation The Ksp for Al(OH)3=2.0X10-32at 25.0 °C. How many grams of aluminum hydroxide will…
A: Grams of aluminium hydroxide
Q: What is the Ksp of Zn3(PO4)2 (MW = 320.8 g/mol) if its solubility in water at 25 °C is 4.9 x 10-5…
A:
Q: Post-lab Question #1-1: Using the Ka for HC2H3O2° (from Appendix F: Ka = 1.8 x 10-5), calculate the…
A: Ka=1.8×10^-5 Kb of C2H3O2^- =?
Q: P * OWLV2 | Online t X C. nment/takeCovalentActivity.do?locator%3Dassignment-take Gmail O YouTube…
A:
Q: You want to prepare 500.0 mL of 1.000 M KNO, at 20 C, but the lab (and water) temperature is 28 "C…
A: We have to find out how many grams of KNO3 should be dissolved at 28℃ temperature to get the below…
Q: STARTING AMOUNT X How many moles of Na,SO, are in a 0.140 m aqueous solution that was prepared with…
A: Molality of solution is defined as number of moles of solute dissolved per kilogram of solvent.
Q: The concentration of C20H42 (FM 282.55) in winter rainwater in Figure 1-3 is 0.2 ppb. Assuming that…
A: Molar concentration is the concentration of a solution expressed as moles/liter and molarity…
Q: Product characterization: 0.91 g of the crude product 0.77 g of the final product M.p. (crude):…
A: Given: Mass of Crude product = 0.77 g Mass of Final product = 0.91 g
Q: ) Complete Question#9 1 CCl Heat + CH3CC 2- CH2 1. 03 2.Zn/H30+ CH3
A: Fridel-crafts acylation:
Q: If the stock of H2S04 contain the following information %=35 Sp.gr.= 1.8 The concentration .M.wt. 98…
A:
Q: 2. How many grams of Cu(C2H302)2 · H20 (FW = 199.65 g/mol) are needed to prepare 500.0 mL of %3D a…
A:
Q: Pre Lab Quiz Lab 3 1. A hydrated FeBr2 salt weighing 1.782 g is heated in a crucible until reaching…
A: The answers are as follows:
Q: So, Keq SOO 2.5x10- [2x] 0.0200.050 2.5x10-9-2x² √2.5x10-9 22x10¹5-x Final answer - The…
A:
Q: A 500.00 mg vitamin C (MW 176.12 g/mol) tablet was ground, acidified and dissolved in H2O to make a…
A: here we are required to find the % vitamin C in the tablet.
Q: e) .SH COCI2 HO excess PhMgBr d) Ph NOH f) 1) nBuLi 2) НСНО 3) с. Н2SO4 Ph
A: Detail mechanistic pathway is given below to find out the product
Q: 101 Chem101 Question 2 of 5 In an aqueous solution at 25°C, if [H,O*] = 2.4 x 10-4 M, then [OH ] is:…
A:
Q: 1.) Me CuLi 2)LDA 3.) a) b.) 1.) c.) 1.) O. 2) 2) O. 2) 3.) 3.) 3.) R. d) 1.) e) 1.) R R. 2) 2) 3.)…
A: Gilman's reagent Me2CuLi acts as a nucleophile.
Q: Supply the missing compounds A through G. В A ОСН3 2. Н.О, НCІ 1. C 2. NHẠCI 1. DIBAH NaOH, A NaBH4…
A:
Q: Cesium chloride is used to prepare dense solutions required for isolating cellular components with a…
A: Given: Density of solution = 1.43 g/mL. And weight percent of CsCl = 40.0 %.
Q: Formula: C10H15NO MCOLET 208 PTR 16 17 19 21 2 ECRONS NEAT 35 28 27 28 28 3 100 10 11 12 13 14 15…
A:
Q: 1) PB13 2) NaCN O Option 1 Option 2 CN CN Option 3 O Option 4
A: 1 is SN2 reaction without rearrangement and 2 is also SN2 reaction . Hence two times stereo changes…
Q: 5. If Ksp = 8.1 x 1019 of an MX3 type salt, what is its solubility? %3D
A: Given : salt type : MX3 Ksp = 8.1 × 10-19 We need to find solubility. Note : There are multiple…
Q: 17. Calculate the percent composition of C6H12O6 Group of answer choices a) 40% C , 53% O , 7 % H…
A: Explanation to the correct answer is given below Standard values Molar mass of carbon = 12 g/mol…
Q: Type: 17) _C4H8 +O2 → - - Туре: 18) KCI +_ Mg(OH)2 → - Туре: 19) Zn + Au(NO2)2 → - Туре:
A: Note: According to our guidelines we are supposed to answer only first three subpart. Kindly repost…
Q: 2-20. A solution was prepared by dissolving 6.34 g of KCl- MgCl2 6H20 (277.85 g/mol) in sufficient…
A: Since you have posted a question with multiple subparts, we will solve the first three subparts for…
Q: Pre-lab question #6-2: Calculate the percent (%) C2042- in Na2C204. 53.0 57.1 65.7 97.8
A: Mass percentage is the ratio of mass of a atom or group to the total mass of the molecule which is…
Q: Post-lab question #1: Ag2CrO4 (s) → 2 Ag* (aq) + CrO42- (aq) If the Ksp for Ag2CrO4 is 1.1 x 10-10.…
A: For Ag2CrO4 : Ksp = 1.1 ×10-10 Volume of solution = 10 mL Molar mass of Ag2CrO4 = 331.73 g mol-1
Q: Experiment 10-Polarity and Solubility name section date Pre-Laboratory Assignment 1. Define the…
A: a) Immiscible: Liquids are classified into miscible and immiscible. Immiscible liquids are to…
Q: Pre-lab question #1: The Ksp for BaCO3 is 5.1 x 10°. How many grams of BaCO3 will dissolve in 1000…
A:
Q: What is the coefficient of H20 when the following equation is properly balanced with the smallest…
A: In a balanced chemical reaction, the number of atoms of each element on the reactant side is equal…
Q: Procaine hydrochloride (MW = 273; L iso value = 3.37). Calculate its NaCl Equivalent
A:
Q: Prepare 100ppm of Cd (NO3).4 H2O in a 500ml
A: Given information, Concentration (in ppm) = 100 ppm Volume of solution = 500 mL
Q: 1. LDA, - 78 °C 2. Cals-CHz-Br в D
A:
Q: 103 + AsO33- l2 12 + AsOs2-
A:
Q: What is the mass percent of carbon in isopropyl alcohol, C3H8O? Group of answer choices 40.0% 60.0%…
A: The mass percent of carbon in isopropyl alcohol has to be given.
Q: -3 Calculate the number of moles of CI ions in 1.75 L of 1.0x10 M ZnCl2
A: ZnCl2 is an ionic compound. Hence it will dissociate into ions completely in solution. The…
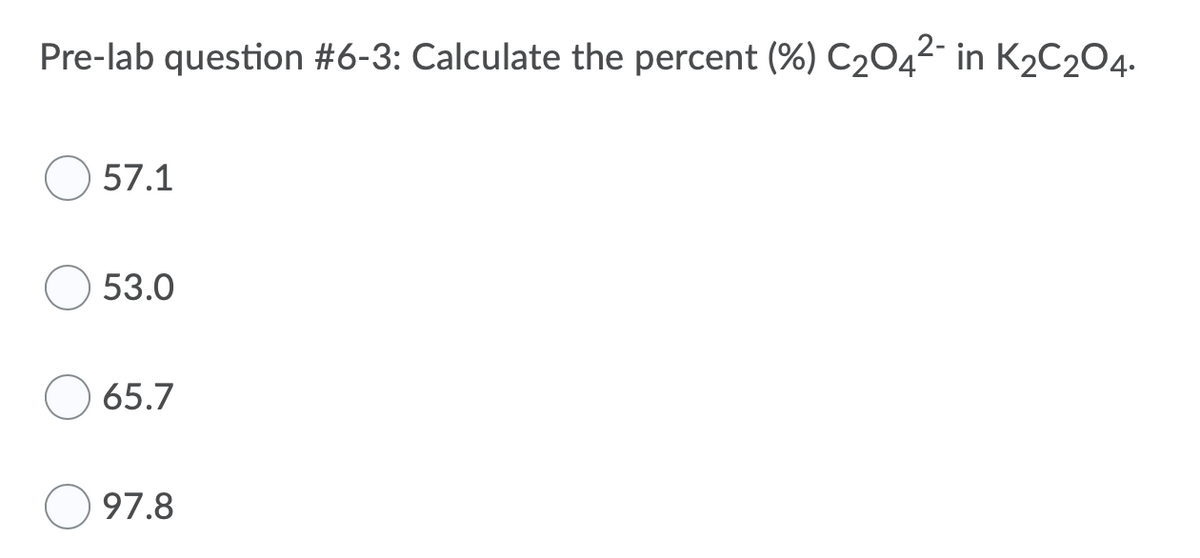
Step by step
Solved in 2 steps with 1 images

- What is the percent magnesium hydroxide, if a sample of magnesia magma weighing 5.2430g when dissolved in 25 mL of 0.9915 N H2SO4 required 9.85 mL of 1.4102 N NaOH to titrate the excess acid? Round to 2 decimal point; Answer should be with the correct unit.How many milliliters of 0.10 M Pb(NO 3 )2 should be used to make ni(en)(h2o)4so4 10% mole excess?A solution of sulfuric acid was made by dissolving 20.0g of 98% sulfuric acid in a 250mL of water. (H2SO4 = 98.078 g/mol; H2O = 18.015 g/mol)Additional information for 35-37 What if the solution has a total volume of 262ml? 1. Express the concentration of this solution in % by weight.a. 7.3%b. 4.4%c. 8.0%d. 7.8% 2. Express the concentration in molaritya. 0.10 Mb. 2.00 Mc. 0.76 Md. 0.93 M 3. Express the concentration in normalitya. 2.0 Nb. 1.5 Nc. 1.0 Nd. 0.76 N 4. Express the concentration in molalitya. 0.80 mb. 0.77 mc. 1.00 md. 1.50 m
- What volume (in mL) of 30.0% hydrogen peroxide solution is required to provide the mass required in question 8 above? Mass=10.2g of a 30% hydrogen peroxide soltuion. Density of 30.0% H2O2 = 1.11 g/mL Answer with 3 sig figs.Dapsone tablets, labelled 50 mg has an average tablet weight of 0.6509 g. 2.0g of powdered tablets reacted (see reaction below) with 26.05 mL of 0.0506N sodium nitrite (NaNO2) solution. Determine the amount of Dapsone as a percentage of the Label Strength (or Label Claim). Report your answer to 1 decimal place. Given: Dapsone molar mass, 248.302 g/mol Dapsone structure Reaction: Sodium nitrite (NaNO2) reacts with hydrochloric acid (HCl) to generate nitrous acid (HNO2). NaNO2 + HCl ® HNO2 + NaCl Nitrous acid (HNO2) then reacts with each amino group on Dapsone, and an indicator indicates the endpoint. R-NH2 (full structure shown above) + HNO2 + HCl ® R-N2Cl + 2H2ONaOH Concentration (M): 0.09669M Trial 1: Inital Volume of NaOH: 0.55mL Final Volume of NaOH: 28.25 mL Trail 2: Intial Volume of NaOH: 18.1 mL Final Volume of NaOH: 46.1 mL Concentration of HCl Solution Subtract the initial buret reading from the end point reading. Convert the readings to liters by dividing by 1000. Solve this equation for the concentration of HCl: (CHCl) (0.01000 liters ) = (CNaOH) (volume of NaOH in liters). CNaOH is the normality of the NaOH you recorded from the bottle. Average the two HCl concentrations.
- SOLVE STEP BY STEPDO NOT USE CHATGPTMAKE THE RESOLUTION QUICKLY PLEASE 18. How many mL of hydrochloric acid are required to measure to prepare 1 L of a 0.25 M solution if the density of HCl is 1.17 g/mL and its purity is 36%? (PA of H is 1,008 amu, Cl is 35,451 amu)What volume in liters of 0.500 M H2SO4 is required to neutralize a solution prepared by dissolving 28.5 g of KOH in 250 mL of water? Group of answer choices 0.508 1.02 0.254 0.127Calculate the ksp for this hypothetical an ionic compound M2X3 (MM=288 g) if its solubility is 3.6 x 10-17 g/L.
- 6-40 What is the molarity of each solution? (a) 47 g of KCI dissolved in enough water to give 375 mL of solution (b) 82.6 g of sucrose, C12H22O11, dissolved in enough water to give 725 mL of solution (c) 9.3 g of ammonium sulfate, (NH4)2SO4 dissolved in enough water to give 2.35 L of solutionA 500 mL solution of 0.250 M NH, is required. What is the volume of 28wt \% of NH, (density = 0.899g / m * L ) required to make this solution? (a) 14.8 mL (b) 2.56 mL (c) 28.3 mL (d) 8.46 mL4. Assuming no calculation errors, why would a percent recovery > 100% or a percent recovery < 100% be obtained? Give at least one reason for each

