Q: What is the product of the reaction below?
A:
Q: The product obtained when the compound below is treated with H20 under acidic condition and heating…
A:
Q: Write the products of the reaction below and in
A: SN1 reaction- AcOH is a polar protic solvent always favours SN1 reaction. step.1 Loss of leaving…
Q: Draw the major organic product you would expect to isolate from the reaction of 3-ethyl-5-methyl-4-…
A: BH3 is complexed with Tetrahydrofuran(THF) to avoid the dimerization of the borane molecule. This…
Q: Provide the organic products of the following and also show the mechanism involved in (a)? (a)…
A: Wittig reagent (CH2=PPh3) has been used both the reaction .
Q: (a) How will you carry out the following conversions?(i) Acetylene to Acetic acid (ii) Toluene to…
A: In organic chemistry conversions play an important role to convert one functional group into another…
Q: Like other hydroborations, the reaction of alkynes with catecholborane is a syn addition and its…
A: The reaction of alkyne with catecholborane is a syn addition and its regioselectivity is opposite to…
Q: (d) For the following reaction give the structure of the heterocyclic product formed and give a…
A: This reaction is an example of Heck Coupling.
Q: A common alkyne starting material is shown below. Predict the major product for each reaction.…
A: given starting material ALKYNE
Q: Provide the major organic product of the following reaction sequence. 1. NAOCH2CH3 .Co,CH2CH3 2.…
A:
Q: Predict the major product of attached epoxidation.
A: Sharpless epoxidation is an enantioselective reaction that prepares 2,3-epoxy alcohols from primary…
Q: 1) LDA, -78 °С 2) CH3BR (a) 1) LDA, 25 °C 2) CH3B. (b) 1) LDA, -78 °С 2) CH;Br (с) 1) LDA, 25 °C 2)…
A: The reaction of ketonic group with LDA at low temperature i.e. -78°C will result in the formation of…
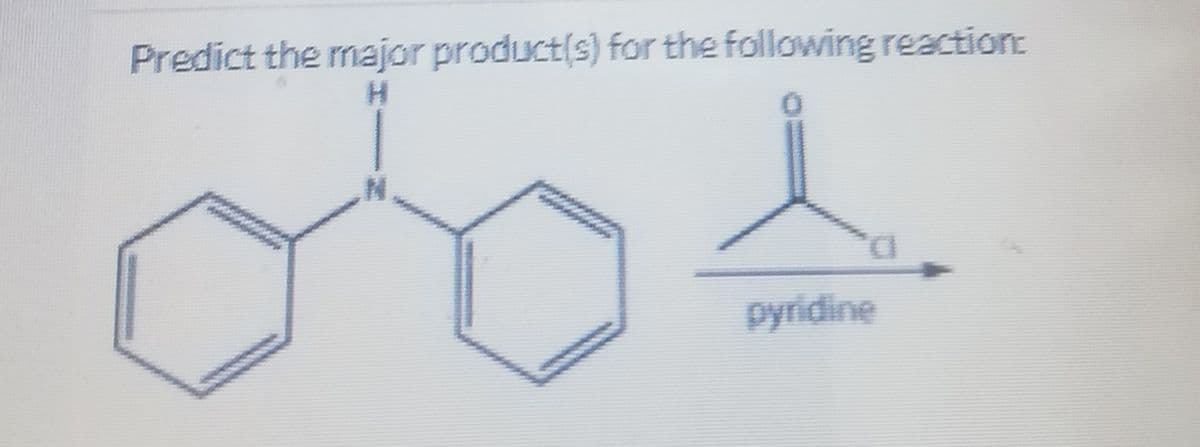
Q: Predict the product of the following Friedel-Crafts acylation reaction. .CI AICI3 OH II II IV Select…
A: Friedel craft acylation is an electrophilic aromatic substitution reaction. This reaction occurs…
Q: Predict the major products of treating the following compounds with hot, concentratedpotassium…
A:
Q: Predict the product and include side product(s):
A: Carboxylic acids in the presence of acidic conditions reacts with alcohols to form esters.
Q: Predict the two most likely mechanisms for the reaction of 2-lodohexane with sodium ethoxide. OA SN2…
A:
Q: Give the expected products of lithium aluminum hydride reduction of the followingcompounds (followed…
A: (a)
Q: Predict the major product(s) you would obtain from sulfonation of 4-bromobenzoic acid.
A:
Q: Predict the products resulting from vigorous oxidation of compound by H2CrO4.
A: Oxidation is the process of addition of oxygen, removal of hydrogen or loss of electrons. Here, the…
Q: Predict the products of reaction of pent-1-yne with the following reagents. Na, liquid ammonia
A: The pent-1-yne is written as: H2H3C-C-C-C≡CH H2
Q: Predict the product(s) of the following reaction. Also identify the nucleophile and electrophile.…
A:
Q: Predict the major monofluorination product from the following explosive reaction: hu F2 ?
A: The explosive reaction for monofluorination of 2,3-dimethylbutane gives 2-fluoro-2,3-dimethyl butane…
Q: Predict the final organic product of the reaction below. Br Ether Solvent Cu +
A:
Q: Two Sonogashira coupling reactions can be used to make unsymmetrical diaryl alkynes by first using…
A: The sequence of reactions starting with trimethylsilylacetylene involves reacting the…
Q: Lindlar Catalyst
A:
Q: Coupling an organic halide with an organometallic reagent or alkene is one of the carbon-carbon…
A: The product of coupling reaction is given below
Q: there are two benzylic bromides that give (z)-4,6-dimethyl-3-phenyl-3-heptene on E2 elimination.…
A: Both the structures have been drawn below.
Q: The synthesis of one of the rings in remdesivir is accomplished using the reaction below. A. Using…
A: Electrophiles are those species which have deficiency of electrons. It attacks on the those species…
Q: Provide the organic products of the following and also show the mechanism involved in (a)? (1) Ph3P…
A: Dear student,the given question is a multiple question, hence I solved first question according to…
Q: HaSou Is03 230°C /24hrs.
A: Pyridine undergoes electrophilic substitution preferably at 3-position(or β-position) as it…
Q: 4. The anisole derivative below undergoes a selective cross coupling reaction on only one of the two…
A:
Q: + Nal II II IV
A:
Q: What will be expected product of nitration of toluene? Explain it with the help of formation of…
A: The question is based on the concept of organic reactions. we have to write the mechanism and…
Q: The structure below represents a tetrahedral alkoxide-ion intermediate formed by addition of a…
A: The starting acid derivative and the ultimate carboxylic acid derivative product is drawn in the…
Q: Predict the major product of the reaction. p-xylene + Na (liquid NH3, CH3CH2OH)
A: When p-xylene is treated with Na in liquid NH3 in the presence of CH3CH2OH(ethanol), it gives…
Q: Select the final product of the following reaction sequence. D. Br 33 OD 1. Mg, ether 2. D₂0
A:
Q: In the early age of the development of Organometallilc Chemistry, the preparation of Organometallic…
A:
Q: (a) Suggest the mechanism of the following reactions and comment on your choice of SN1 or SN2. ()…
A: Detail mechanism is shown below
Q: Predict the products of the following reactions. An excess of acid is available in each case.…
A: Given, anisole (methoxybenzene) + HBr
Q: Predict the major product formed in each of the following reactions. in each case only one…
A: Organic reactions are those in which organic reactant react to form organic products.
Q: . The following diagram represents a synthesis which begins with bromoethane. (i) (iii) C2H6O (C)…
A: A chemical reaction mechanism represents the way to form and break the chemical bonds between…
Q: Which of the following does not correctly describe SN2 reactions of alkyl halides? Explain your…
A:
Q: Provide all resonance structures of the sigma complex when aniline reacts with an electrophile to…
A: Reaction is taking place between aniline and Br2 (Br electrophile)
Q: ONa NH2 2) Hг0
A: Given,
Q: Show how you would prepare the following from the indicated starting materials. More than one step…
A:
Q: Give the product(s) formed when the following species react together: CH3CH2OH and-NH2
A: A substance that can give H+ ion is called acid and a substance that accepts H+ ion is called base.
Q: Predict the Product. Provide the stable organic product(s) for the reactions below. 1. сн,MgBr (xв)…
A: MeMgBr in excess converts ester or lactone to secondary alcohol NaBH4 reduces aldehyde to primary…
Q: NaBH, NaBH, OH PCC CH,CI,
A:
Q: Predict the product of the reaction of p-methylbenzoic acid with each of the following: (a) LiAlH4,…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Predict the Minor Product 2,5-dimethyloct-3-ene + HCl --->Predict the products resulting from vigorous oxidation of compound by H2CrO4.Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…
- Predict the major organic product of the following reaction for d or eOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Answer the following question pertaining to the reaction of maleic anhydride and 1,3-butadiene: Why does 1,3-butadiene react more rapidly with maleic anhydride than with another molecule of itself?
- E. TESTS FOR PHENOLIC COMPOUNDSPhenolic compounds such as phenol, salicylic acid, etc., give characteristiccolors with FeCl3 and Millon’s Reagent.1. Ferric Chloride TestAdd 5 drops of FeCl3 solution to 4 different test tubes containing 1 mL each ofdilute solutions of the following compounds and note the color obtained.a. Phenol _______________________________________________________________b. Salicyclic acid ___________________________________________________________c. Resorcinol _______________________________________________________________d. Picric acid _______________________________________________________________2. Millon’s TestPrepare another set of 4 test tubes containing 1 mL each of the solutions listedbelow. Add 3 drops of Millon’s reagent to each and place all the test tubes in aboiling water bath. Note the color formed.a. Phenol _______________________________________________________________b. Salicylic acid ___________________________________________________________c.…What happens when (write reactions involved) iv and vWhich or which of the statements given below is correct. I) Maleic anhydride is a carboxylic acid derivative and its reaction with water is a reduction reaction. II) Fumaric acid and maleic acid are stereoisomers of each other III) Since fumaric acid has a more stable structure than maleic acid, its boiling point is higher. A. Solo I B. I and III C. II and III D. I, II, III E. Solo III
- 1. Write equations, using displayed formulae, to show the conversion of but-1-eneinto:a) polybut-1-eneb) 1,2-dibromobutane 2. a) Explain why alkenes tend to react with electrophiles.b) Show the mechanisms for the following reactions:i) ethene with bromineii) but-2-ene with hydrogen bromidec) Explain how bromine can behave as an electrophile. 3. Reaction of but-1-ene with HBr gives two products in unequal amounts. In eachcase, identify the two products, state which is the major product, explain why it isthe major product and give the mechanism for its formation.Predict the major organic product of OH and Br with NaOHDraw the structure of the compound you would expect from SN2 reaction of the molecule below with NaCN.



