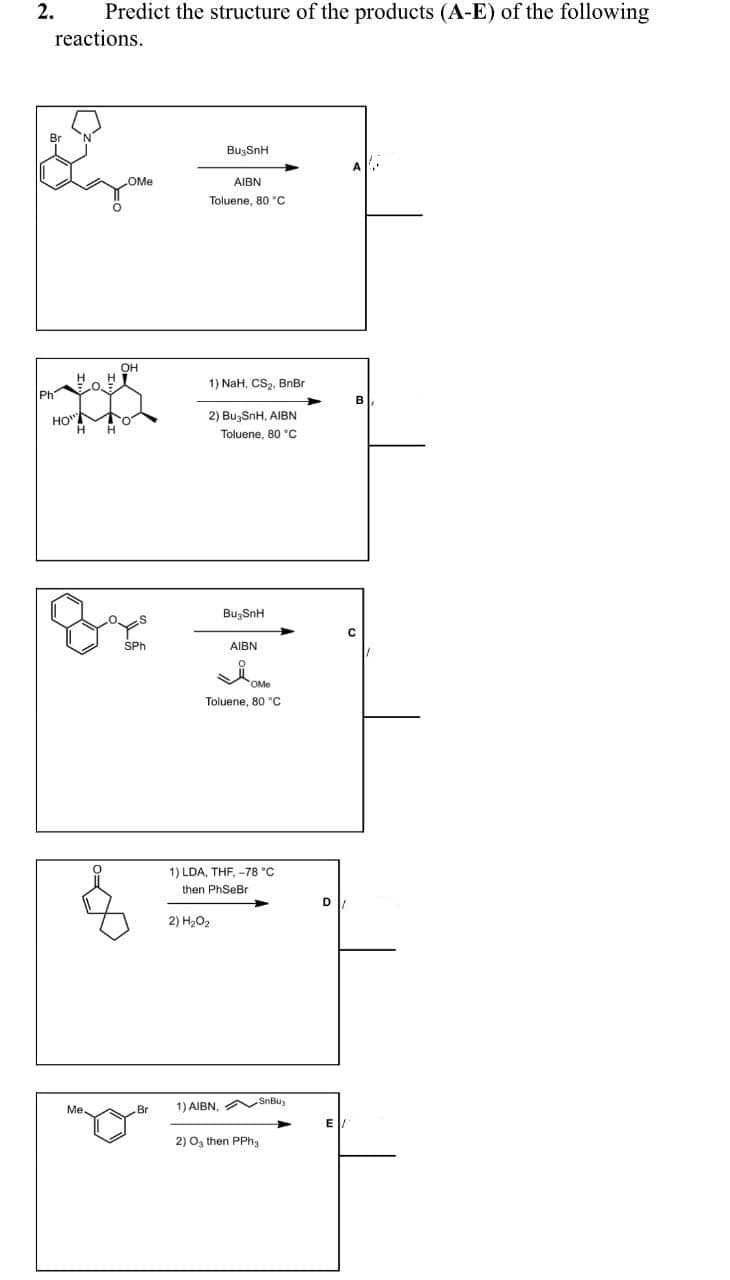
Predict the structure of the products (A-E) of the following reactions. Br BuzSnH OMe AIBN Toluene, 80 "C Он 1) NaH, CSz, BnBr Ph B HO" 2) Bu,SnH, AIBN Toluene, 80 °C BugSnH SPh AIBN OMe Toluene, 80 "C 2.
Q: 1. THE Ph OMe BrMg 2. HCI methyl benzoate Grignard reagent malachite green If 0.043 moles of the Gri...
A: Given :- grignard reagent + methyl benzoate → malachite green Molar mass of grignard reagent = 22...
Q: The volume of gas at 27 C is 400mL.What will be the volume of that gas at 47 C, if the pressure is h...
A:
Q: Show the calculation required to determine the pH of a solution that has H3O+ concentration of 0.015...
A: pH of solution is defined as negative logarithm of hydrogen ions.
Q: Compute for the volume (in mL) of 0.5 M acetic acid and mass (in g) of sodium acetate needed to prep...
A: The concentration of buffer to be prepared is given as 3mM but the volume of buffer is not mentioned...
Q: what is the change in the enthalpy in kj/kg for the complete combustion of 39.0 g of fructose, rough...
A:
Q: Why and how does molecular geometry affect polarity and also solubility? Please explain thoroughly.
A: Molecular geometry determine the polarity and the polarity determines the solubility of a compound. ...
Q: According to the activity series, which of the following metals will react with HCI to produce H2(g)...
A: In the activity series, on moving from above to below in the activity series, there is a lower proba...
Q: In the compound K4[Fe(CN)6], the oxidation state of iron and its coordination number, respectively, ...
A: Note : Since you have posted multiple questions, we are entitled to answer the first only. Please re...
Q: 2. Model C2H6, C2H4, and C2H2. Visualize/sketch molecular orbital(s) and identify if it is o- or t- ...
A:
Q: po arrangement of electrons.. Using terms such as coulombic and exchange energies, determine the dif...
A: Different possible arrangements are shown :
Q: A(g) B(g) C(g) The rate law for the above reaction is: -d[A]/dt = k[A][B] The rate constant is 3.13x...
A:
Q: Based on the thermochemical equation shown below, which statement is corred? C6 H12O6(s) + 602 6 CO2...
A:
Q: What number of atoms of nitrogen are present in 3.54 g of each of the following? a. glycine: C2H5 O2...
A: Given, Calculate the number of atoms of Nitrogen are present in 3.54 grams of each of the following:...
Q: Some chemicals, like hydrogen peroxide, can you prevent photocatalysis? 1. are prone to decompositio...
A: We are authorised to solve only one question at a time. Please post rest of the questions separately...
Q: a. Predict the major and minor products for the following reactions (excluding stereochemistry) b. U...
A: Addition of HBr to alkene follows markonikov's rule.
Q: Predict the products of the following reaction. If no reaction will occur, use the NO REACTION butto...
A: This reaction is proceed in forward direction. Due to ionic form lithium sulfide react with HCL. Ion...
Q: For the following reaction, 5.48 grams of zinc oxide are mixed with excess water . The reaction yiel...
A:
Q: what is the lewis structure of POF3, its geometry, intramolecular forces and covalence
A:
Q: if you start with 50 grams of cobolt-60 how many grams would you have of it left after 5 half lives
A:
Q: For the following reaction, 17.4 grams of chlorine gas are allowed to react with 3.50 grams of water...
A: The correct answer is given below
Q: If qsolution -10 kJ for the following reaction conducted in a coffee cup calorimeter, what is qreact...
A: Please find the below attachments.
Q: 4.52 Plexiglas, a clear plastic used to make many molded articles, is made by polymerization of meth...
A: Polymerization is the process of formation of polymer from the maneric units .
Q: -In the compound Ka[Fe(CN)6], the oxidation state of iron and its coordination number, respectively,...
A:
Q: 3. A 100 mL sample of hard water is titrated with 22.4 mL of the EDTA solution from problem 2. The s...
A: Given information :- additional info: 1)25.95 mL of EDTA are needed to titrate the Ca2+ ion in the ...
Q: Do chelators reduce conductivity? Do water-miscible solvents such as mono ethylene glycol, glycol et...
A: A question based on coordination complexes that is to be accomplished.
Q: Model H2C=C=CH2. Visualize/sketch molecular orbitals, HOMO, HOMO-1 and HOMO-2 and dentify if it is o...
A:
Q: . Choose the correct shape, weak or strong field, and number of unpaired electrons for [Co(NH3)6]** ...
A:
Q: An aqueous solution of calcium hydroxide is standardized by titration with a 0.124M solution of hydr...
A: We have to predict the Molarity of calcium hydroxide.
Q: Rubbing alcohol is commonly used as an antiseptic for small cuts. It is sold as a 70% solution of is...
A: Solution is made up of two components. Solute and solvent. Component which is present in major amoun...
Q: The pH of an acidic solution is 5.47. What is [OH⁻]
A: Hydrogen ion concentration is an important parameter of an aqueous solution. The concentration of th...
Q: How many kilograms of water is needed to prepare 7.12 molal of solution using 23.0 g KNO2? The molar...
A:
Q: Determine the molar solubility (S) of Ag, CO, in a buffered solution with a pH of 3.289 using the sy...
A:
Q: The elements lithium and oxygen react ex- plosively to from lithium oxide (LigO) accord- ing to the ...
A:
Q: Below are the resonance structures for the sigma complex that is formed when the ortho position of c...
A:
Q: When a student mixes 3.9537g of NaHCO3 with excess HCI in a constant pressure calorimeter the temper...
A: To calculate the enthalpy change , we would first calculate heat of solution . Then we would calula...
Q: Determine the molar solubility for Mg(OH)2 (Ksp = 1.0 × 10-31) in an aqueous solution that has a pH ...
A: Given: pH of solution = 8.90 And Ksp of Mg(OH)2 = 1.0 × 10-31 The solubility reaction given is,
Q: Phosphate buffered saline (PBS) is a very common reagent in cell biology labs that do cell culture s...
A: The components of 10X PBS are 10.5 mM KH2PO4, 1.552 M NaCl and 29.7mM Na2HPO4. Volume of 10X PBS req...
Q: a What is the maximum mass of ammonia that can be produced from a mixture of 1.00 x 10 g N2 and 5.60...
A:
Q: Compare and contrast the physical and chemical properties of the following pairs of isomers. Propan...
A: Compounds that contain exactly the same number of atoms but differ in their atomic arrangement is kn...
Q: Calculate the molar solubility of aluminum hydroxide in a solution buffered at pH = 4.50. Al(OH)3 (...
A: Consider the given information is as follows; pH = 4.50Ksp = 1.3×10-33
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the tem...
A: “Since you have asked multiple question, we will solve the first question for you. If you want any s...
Q: Determine if the temperature will increase, decrease, or remain the same for each of the following s...
A: In an exothermic reaction, heat is released to the surrounding from the system. In an endothermic re...
Q: An impure sample of zinc (Zn) is treated with an excess of sulfuric acid (H,SO,) to form zinc sulfat...
A: The given reaction is of zinc with sulphuric acid and we have to write the balanced reaction and fil...
Q: Suppose a 500. mL flask is filled with 1.5 mol of O, and 0.60 mol of NO. The following reaction beco...
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: If 50.0 mL of NaOHNaOH solution is required to react completely with 0.44 gg KHPKHP, what is th...
A: Given :- Volume of NaOH = 50.0 mL Mass of KHP = 0.44 g To calculate :- Molarity of NaOH solution
Q: For these two equations (2) and (3), determine whether they are exothermic or endothermic, and why.
A: The chemical reaction accompanied by or requiring the absorption of heat is known as an endothermic ...
Q: Calculate the initial molarity of iodide for run 1 (addition of beaker 1 to flask 1) given the foll...
A: Dilution equation is used in such cases of reaction to find the final molarity of solution when the ...
Q: Determine the following: Pm, Pv, Pm/v, XA, m, and M if 107.125 grams NaOH is dissolved in 480 mL of ...
A: Given values Mass of NaOH = 107.125 g Volume of water = 480 mL Density of NaOH = 2.13 g/ mL Densi...
Q: Combine the following half-reactions to obtain a balanced overall redox reaction. Cu2++2e−→Cu an...
A:
Q: How do I find the keq of this reaction
A: The answer is as follows:
Step by step
Solved in 4 steps with 3 images

- predict the products: XCL2+ E3PO4 --->Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4AI(C2H5)3 →3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d = 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3): d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257 L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Chemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?Help with the following ochem reaction scheme... Consider 3,4-dimethylpiperidine being subjected to the below reaction scheme: step 1) CH3I (excess) step 2) NaOH, heat step 3) CH3I (excess) step 4) NaOH, heat Provide the bond line structures for the major organic product obtained in each step and then discuss the regiochemistry for step 2.
- When Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.(a) Propose structures for products A and B. (Hint: In many cases, an allylic carbocationis more stable than a bromonium ion.)up an example (not appearing in this ChemActivity) of a pair of molecules that are a)constitutional isomers, b) conformers. c) configurational stereoisomers.All attempts to isolate primary and secondary nitroso compounds result solely in the formation of oximes. Tertiary nitroso compounds, however, are stable. Explain.
- please provide the machanisms of 1a, 1e, 1fArrange the following compounds in the increasing order of reactivity towards Conc.HNO3 & Conc.H₂SO4 1. Benzene 2. Chlorobenzene 3. phenol 4. Toluene 5. Nitrobenzene A 1.2.3.4.5 (B) 5.1,243 5.1.4.2.3 D 5.2.1.4.3Give detailed answer- Provide example of compound that fulfil each criteria below. Explain why-A)1,2-disconnectionB)1,4-disconnectionC)1,5-disconnectionD)1,6-disconnection



