pressure Units of pressure: • 1 atm = 101325 Pa (pascal) • 1 atm = 760 torr (mmHg) sold iquid vapour temperature Figure 6: The Normal Melting and Boiling Points in the Phase Diagram
pressure Units of pressure: • 1 atm = 101325 Pa (pascal) • 1 atm = 760 torr (mmHg) sold iquid vapour temperature Figure 6: The Normal Melting and Boiling Points in the Phase Diagram
Chapter10: Liquids And Solids
Section: Chapter Questions
Problem 93E: Carbon tetrachloride, CCl4, has a vapor pressure of 213 torr at 40.C and 836 torr at 80.C. What is...
Related questions
Question
How will you differentiate the two important points in the phase diagram?
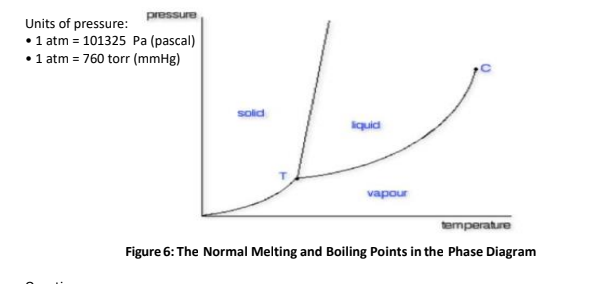
Transcribed Image Text:Units of pressure:
•1 atm = 101325 Pa (pascal)
•1 atm = 760 torr (mmHg)
anssaid
solid
iquid
vapour
temperature
Figure 6: The Normal Melting and Boiling Points in the Phase Diagram
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning