Problem #1 Consider the following balanced equation: AgNO3 + NaCl → NaNO3 + AgCl Assume that you begin with 60 g of AGNO3 and 15 g of NaCl (sodium chloride). Create a pair of stoichiometric setups as shown in the graphic below: given amount of substance molar mass (g/mol) molar mass mole (mol/g) ratios Complete the setups to compare the theoretical yield of each reactant. Use those predictions to determine whether AGNO, or NaCl is the limiting reactant in the formation of NANO, (sodium nitrate).
Problem #1 Consider the following balanced equation: AgNO3 + NaCl → NaNO3 + AgCl Assume that you begin with 60 g of AGNO3 and 15 g of NaCl (sodium chloride). Create a pair of stoichiometric setups as shown in the graphic below: given amount of substance molar mass (g/mol) molar mass mole (mol/g) ratios Complete the setups to compare the theoretical yield of each reactant. Use those predictions to determine whether AGNO, or NaCl is the limiting reactant in the formation of NANO, (sodium nitrate).
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter3: Stoichiometry
Section: Chapter Questions
Problem 13ALQ: What is true about the chemical properties of the product? a. The properties are more like chemical...
Related questions
Question
100%
I need help with my chem hw
Transcribed Image Text:ems 2021.docx
ert
Layout
References
Review
View
Help
QTell me what you want to do
Editing v
E A ov a
ebuchet MS
A
A
...
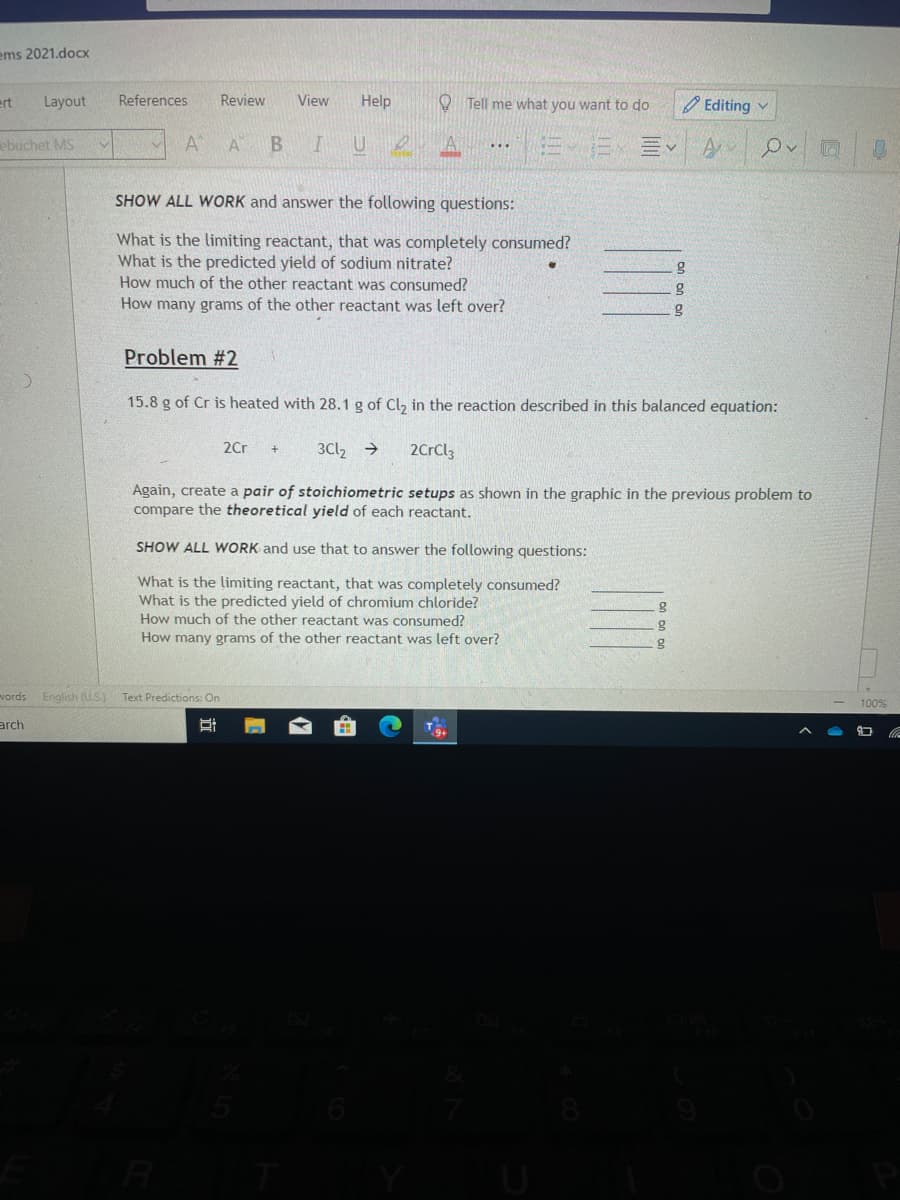
SHOW ALL WORK and answer the following questions:
What is the limiting reactant, that was completely consumed?
What is the predicted yield of sodium nitrate?
How much of the other reactant was consumed?
g
How many grams of the other reactant was left over?
Problem #2
15.8 g of Cr is heated with 28.1 g of Cl, in the reaction described in this balanced equation:
2Cr
3Cl, →
2CrClz
Again, create a pair of stoichiometric setups as shown in the graphic in the previous problem to
compare the theoretical yield of each reactant.
SHOW ALL WORK and use that to answer the following questions:
What is the limiting reactant, that was completely consumed?
What is the predicted yield of chromium chloride?
How much of the other reactant was consumed?
How many grams of the other reactant was left over?
g
vords English (U.S.) Text Predictions: On
100%
arch
R
Transcribed Image Text:s 2021.docx
Layout
References
Review
View
Help
O Tell me what you want to do
Editing v
chet MS
v 20
A
В
U
ev Av
三vニv三 ov
...
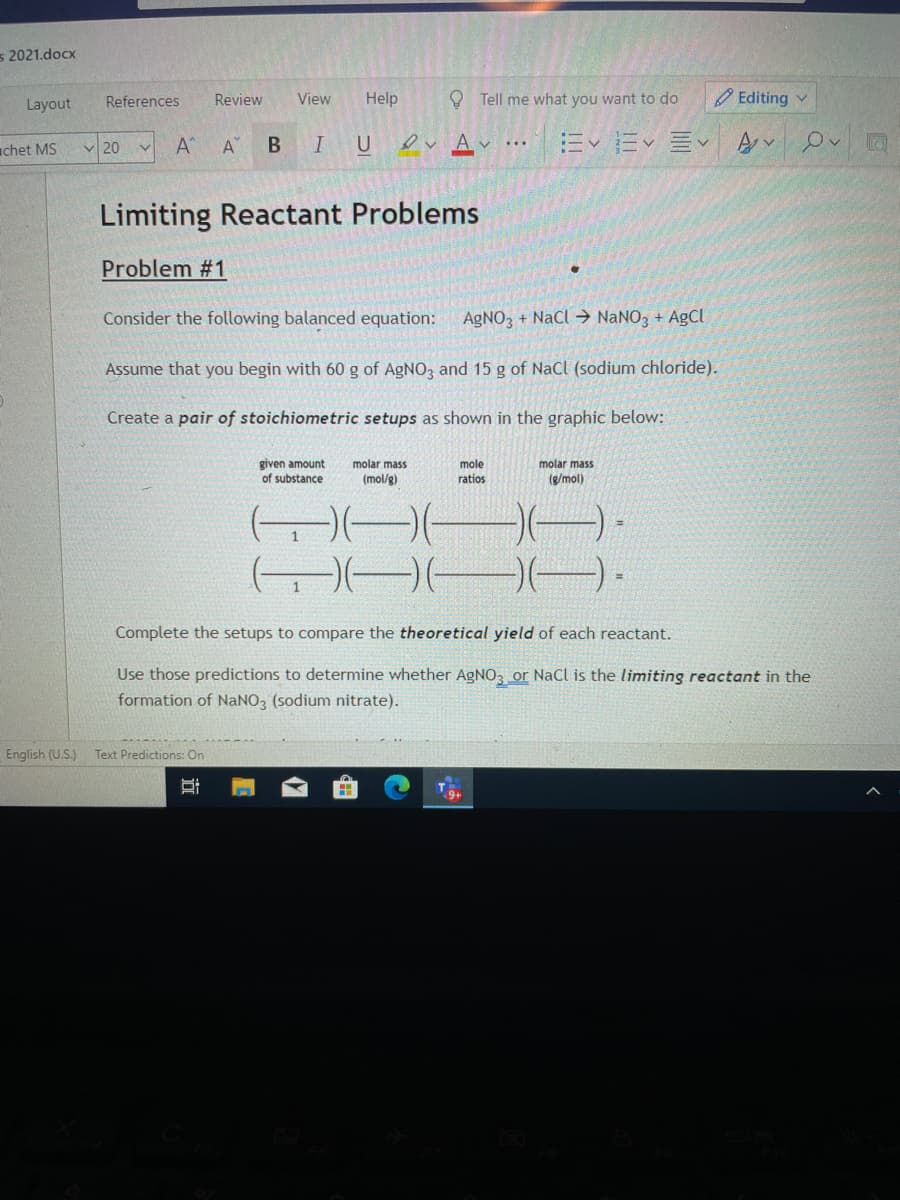
Limiting Reactant Problems
Problem #1
Consider the following balanced equation:
AGNO3 + NaCl → NANO3 + AgCl
Assume that you begin with 60 g of AGNO, and 15 g of NaCl (sodium chloride).
Create a pair of stoichiometric setups as shown in the graphic below:
given amount
of substance
mole
ratios
molar mass
molar mass
(mol/g)
(g/mol)
Complete the setups to compare the theoretical yield of each reactant.
Use those predictions to determine whether AgNO3 or NaCl is the limiting reactant in the
formation of NaNO, (sodium nitrate).
English (U.S.)
Text Predictions: On
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning