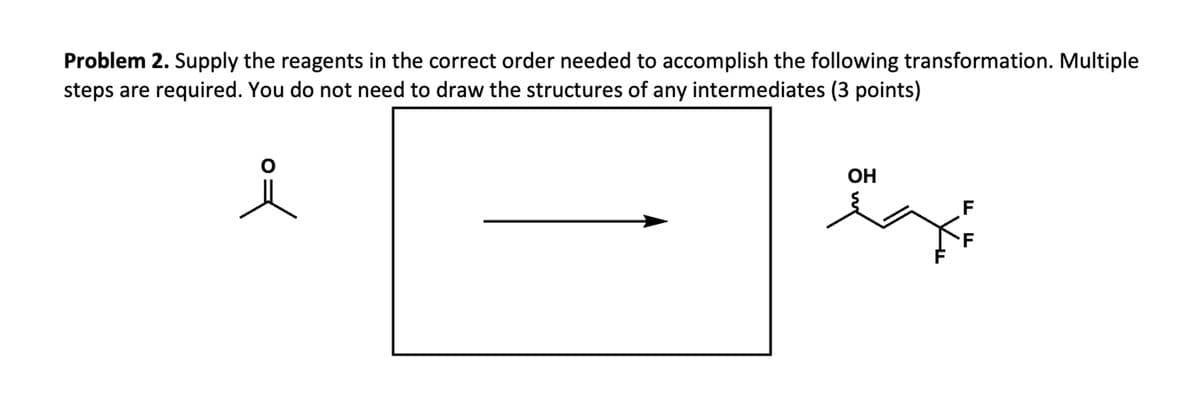
Problem 2. Supply the reagents in the correct order needed to accomplish the following transformation. Multiple steps are required. You do not need to draw the structures of any intermediates (3 points) 요 F F OH HH ہجر
Q: Heating D-altrose with dilute acid produces a nonreducing anhydro sugar (C6H1005). Methylation of…
A: The objective of the question is to understand the structure of the anhydro sugar formed by heating…
Q: Is there a minimum standard reduction potential that the half- reaction used at the cathode of this…
A: Approach to solving the question: there is a minimum standard reduction potential, but there is no…
Q: Course: College Chemistry Course code: CHM 141 Complete the table that follows, indicate changes in…
A: Approach to solving the question:Le Chatelier's principle is used to identify the change in the…
Q: an efficient synthesis for each of the following compounds using the 22.39 Propose acetoacetic ester…
A: Step 1: a)1. Acetoacetic ester with sodium ethoxide, deprotonates the acidic hydrogen forming…
Q: Drow thise reaction mechanism
A:
Q: 4.00 g NaOH(s) (39.996 g mol–1) dissolves in 100.0 g of water in a constant–pressure calorimeter.…
A: Step 1:Moles of NaOH= molarmassmass= 39.996g/mol4.00g= 0.100 mol.Given,…
Q: You are burning organic brush in your backyard. The molecular formula for the brush is: C204 H325…
A: Now, let's perform the calculations: b. Number of moles of compost that burns:We'll first find the…
Q: For the substrate below write all possible reactions. Ethanol is a weak nucleophile. Please verify…
A: Step 1: Step 2: Step 3: Step 4:
Q: Provide a synthesis of the target molecule. All of the carbon atoms in the final product must come…
A: Step 1: Wittig reaction:Methyl bromide with triphenyl phosphine and n-BuLi gives phosphorus…
Q: Fast expert solutions please
A:
Q: What is the IUPAC name for the compound shown in the figure?
A: Step 1: Find the longest carbon chain containing the alkyne.Number the chain to give the alkyne the…
Q: A 1.864 g sample of vinegar requires 12.65 mL of 0.1403 M NaOH solution to reach the endpoint. If…
A: Given: masssample1=1.864g;V1=12.65mL;[NaOH]=0.1403Mmasssample2=1.774g;V2=???mLStep 1: Write the…
Q: Use the References to access important values if needed for this quest Hybrid orbitals are formed by…
A: a) The electron pair geometry of this molecule is Trigonal Bipyramidal The geometry of electron…
Q: A weak base has a K₁ of 5.6x104, determine the pK₁ of the base. 3.25 7.64 10.75 6.36
A: The pKb of a base is the negative logarithmic of Kb value of the base. pKb = - log Kb As per the…
Q: 1) Provide the structure of the major product, which results from Diels-Alder reaction. +…
A:
Q: What is the product of this acid base reaction assuming a 1:1 reaction? YY + CH3
A:
Q: 2. In the synthesis of peptides, carboxylic acids are condensed with amines in the present of a…
A:
Q: Part A Account for the difference in the products of the following reactions: ф-оф-о Check all that…
A:
Q: (a-f) best describes the major product(s) you would expect form? Choose only one. d) C a Ob O Br…
A: option ( e ) product is major product because it is more substituted alkene which is major alkene…
Q: Heating D-altrose with dilute acid produces a nonreducing anhydro sugar (C6H1005). Methylation of…
A: The objective of the question is to understand the structure of the anhydro sugar formed by heating…
Q: Draw the tautomer product of each molecule shown below (NOTE: the reaction conditions result…
A: Step 1: When two molecules have the same molecular structures but different interconvertible…
Q: 4 1/1 point The solubility of the ionic compound M2X3, having a molar mass of 288 g/mol, is 3.60…
A: Step 1:Given solubility of M2X3 = 3.60 × 10-7 g/L molar mass of the compound = 288 g/mol.Conversion…
Q: Please don't provide handwritten solution...
A: The objective of the question is to determine the number of signals that would be observed in the…
Q: Imagine you are tasked with designing a building in a seismically active area. You have met with…
A: Question 2:(a) Definitions:Void ratio (e): The ratio of the volume of voids (Vv) to the volume of…
Q: Can you explain the problem? How do you identify them?
A: Step 1: SolutionThe removal of one proton (H+) from a given compound gives us a conjugate base. We…
Q: Consider a reaction that has a negative AH and a negative AS. Which of the following statements is…
A: 1.The relation between ΔG, ΔH, and ΔS can be described by the equation below:wherein T is the…
Q: Draw a mechanism for the following transformation
A: Step 1:Electrophilic substitution reaction of benzene:Benzene behaves as a good nucleophile, and…
Q: 4. Propose a synthesis for the transformations shown below (no mechanisms needed, just…
A: Here are the proposed syntheses for each transformation: 1. 3-methylbut-1-yne →…
Q: Each row of the table below describes an aqueous solution at 25 °C. The second column of the table…
A: Step 1: A. The initial solution contains only water (H2O), making it neutral. When KClO4 is added,…
Q: Choose the best reagents to complete the following reaction.
A:
Q: road map
A:
Q: Please don't use hend raiting
A: Step 1:carbon (B) atom that participates in resonance typically has lower electron density compared…
Q: The officers of a high school senior class are planning to rent buses and vans for a class trip.…
A: Detailed explanation:Step 1: Understand the Problem There are two categories of vehicles to think…
Q: Explore relative reducing/oxidizing ability. Use the half-reactions shown below to answer the…
A: Step 1:Step 2:
Q: Draw a plausible mechanism for the following transformation: te=& OH H3O+ HO Step 0 × Your answer is…
A:
Q: III O Chemical Reactions Solving for a reactant in solution 3/5 B One way in which the useful metal…
A: 7.9 x 10^2 g/l
Q: Please correct answer and don't use hend
A: Step 1:If there were acetal present with oxygen atoms in place of the sulphur, then the hydrolysis…
Q: Huhuhuhuhuu
A: C) The ring with methyl group is activated one because the CH3 group donated electron density inside…
Q: name the product of the reaction excess H,, Ni
A:
Q: Consider the reaction:CaCO3(s)CaO(s) + CO2(g)Using standard thermodynamic data at 298K, calculate…
A: Step 1:Given reaction:CaCO3(s) → CaO(s) + CO2(g) ΔH0=∑ΔHf0(products)−∑ΔHf0(reactant)…
Q: Choose the best reagents to complete the following reaction.
A: Step 1: Step 2: Step 3: Step 4:
Q: 12. In the hydrogenation reaction shown, which of the alkenes below would yield the observed…
A: In a hydrogenation reaction involving H2 Pd/C, both the hydrogen atoms attach on the same side. This…
Q: 2. Which of the following compounds would be considered anti-aromatic?
A: Step 1:Hückel's rule for aromaticity, antiaromatic and non aromatic compound are :-…
Q: None
A: The reaction given is a hydroboration-oxidation reaction.In this type of reaction, an -OH group is…
Q: The write-up says “A C18-silica column is used in this experiment, a 0.46 x 15 cm column with 5-µm…
A: Column dimensions: Particle size: Flow rate: Buffer concentration: 0.010 MBuffer pH: 7.2
Q: What is the pH of the resulting solution when 0.456 g of sodium acetate are dissolved into 1 L of…
A: Given: massNaCH3CO2=0.456g;MNaCH3CO2=82.0343g/mol;V=1.00L;Ka=1.8x10−5;pH=???Step 1: Solve for…
Q: In each compound below, rank the labeled protons in order of increasing acidity.
A: In general, the acidity of a compound increases with the number of oxygen atoms directly bonded to a…
Q: 3. Draw compounds A-G in the space below the scheme using line notation ΟΞ A: hexanoic acid B:…
A:
Q: Learning AA prod03-cnow-owl.cengagenow.com Login Learning Learning × Online tea... y dr. marlow...…
A: Given:G°f(BrF3(g)) = -229.4KJ/molG°f(Br2(g)) = 3.1 KJ/molG°f(F2(g)) = 0.0 KJ/mol Balanced chemical…
Q: 2Fe(s) + 3Cl2(g)2FeCl3(s)Using standard thermodynamic data at 298K, calculate the entropy change for…
A: The objective of the question is to calculate the entropy change for the surroundings when 1.74…
Please don't provide handwritten solution ...
Step by step
Solved in 2 steps with 3 images
