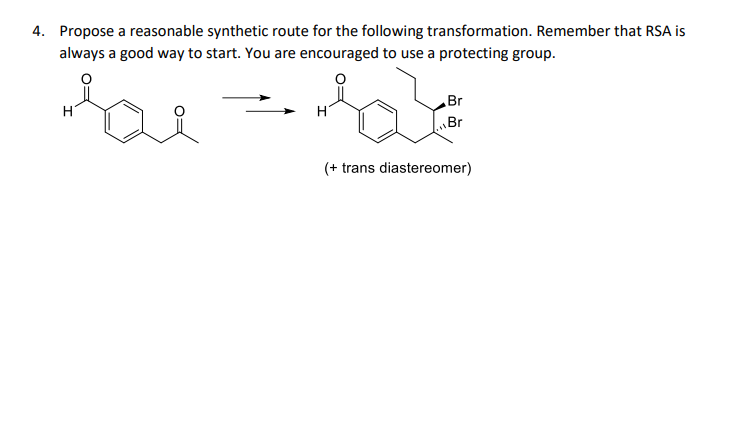
4. Propose a reasonable synthetic route for the following transformation. Remember that RSA is always a good way to start. You are encouraged to use a protecting group. Br H Br (+ trans diastereomer)
Q: What is the aim of submerging the electrode in a potassium chloride solution? Must this be stored in...
A: Potassium chloride (KCl) goes about as a wellspring of chloride particles for the anode. The upside ...
Q: change in the internal energy
A:
Q: Which of these molecules contains fewest high energy electrons (i.e. in which of these molecules is ...
A: The carbon becomes more oxidized when it is the carbonyl(C=O) carbon.
Q: 100 90 80 70 50 40 30 20 10 3500 3000 2500 2000 1500 1000 500 Wave number (cm -1) Band Intensity W o...
A:
Q: Does it possible to use gravimetric analysis to quantiify certain parameters as indicators of quali...
A:
Q: Five different solutions were prepared in the laboratory. One was prepared by dissolving 7.1 g sodiu...
A:
Q: 13C NMR PROBLEM 1 How many signals are expected in the decoupled 13C NMR spectrum of butyl acetate? ...
A: The NMR spectroscopy is an important tool for the determination of the structure of an organic compo...
Q: A weather balloon is inflated to a volume of 26.4 L at a pressure of 740. mmHg and a temperature of ...
A:
Q: Based on the table, which of the samples contain double bonds?
A:
Q: What is the pH of a 0.450 M solution of Ca(NO₂)₂ (Ka of HNO₂ is 4.5 × 10⁻⁴)?
A:
Q: A 2 molal hydrochloric acid (HCl) solution freezes at - 8.86 C. Calculate the apparent percentage of...
A:
Q: Before proceeding to the analysis, the ____________ of the sample must be considered. a. size b. c...
A: Answer: In the chemistry lab, samples are often being analyzed to gather the certain information abo...
Q: Unsymmetrical bond breaking is a mechanism for electrophilic or nucleophilic substitution reactions....
A: A covalent bond is formed by sharing of electrons between the atoms. When the covalent bond breaks, ...
Q: CH Br CH,CH2C=CH NANH2 1. ( 2 =-CH3 (3) Lindlar's catalyst
A: When sodium amide reacts with alkyne, sodium replaces the terminal hydrogen. Further reacting with a...
Q: Which of the properties below apply to the following polymer? CH CH2 CH CH2 Choose all that apply.Ch...
A:
Q: Like many metals, aluminum reacts with a halogen to give a metal halide: 2 Al(s) +3 Br2 (e) → Al, Br...
A: Molar mass of Al = 26.98 g/mol Molar mass of Br2 = 159.8 g/mol Molar mass of Al2Br6 = 533.4 g/mol Ma...
Q: The resulting chromatogram showed the separation of two component in the sample. Component A is clos...
A: Given that, for a chromatogram separation of two components in the sample, where component A is clos...
Q: What is the equilibrium concentration (in M to three decimal places) of HBr for the following reacti...
A: Given, H2(g) + Br2(g) ⇌ 2HBr(g) Equilibrium constant (Keq) at 25°C = 0.000351 Initial concentration...
Q: Which of the following will forin a basic solution in water? [Select all that apply) NaCIOa(aq) O Li...
A:
Q: Correct Let's calculate the number of moles of each reactant: 1 mol CH4 1370 g CH4X = 85.4 mol CH, 1...
A: Number of moles is calculated by dividing the given mass with molecular mass.
Q: If the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the neg...
A:
Q: When a solid dissolves in water, heat may be evolved or absorbed. The heat of dissolution (dissolvin...
A: heat of dissolution is the heat which may be releases or absorbed when a solid is dissolved in water...
Q: 1. Calculate the potential as a function of titrant volume when 50 mL of 0.100 M Fe* is titrated wit...
A: Given, Titration of 50mL 0.100M Fe2+ solution is done with 0.100M Ce4+ solution. Before the equival...
Q: Which intermediates are formed during the mechanism of the below reaction? Select all that apply. 2 ...
A:
Q: Remaining Time: 26 minutes, 33 seconds. Question Completion Status: > A Moving to the next question ...
A: The bcc lattice can be considered as a unit cubic cell with two lattice points per cell.
Q: Determine the OH- and H+ concentration and calculate the pH, pOH of a solution that is 0.70 M NH3 so...
A: pH of a solution is a measure of acidity of a solution. pH less than 7 indicate that solution is aci...
Q: Br2 1) Mg/ether 2) CH,CH,CH,CHO 3) H,O* H3C. FeBr3 d) CH3 Cro3 H3C. HNO, H,SO4, A H,SO4 e) CH3
A: Both are Multi step synthesis reaction . Detail mechanistic pathway is given below
Q: )A 315.5 g of sodium chloride is dissolved in 900 g of water at 25 degree Celsius. Find: a. Final va...
A:
Q: CH3 1.Hg(ОАC)2, Н,0, THF 2. NaBH4 (d) CH2=CH-CH2-CH-CH3 1. NaBH4 (e) CH2=CH-CH2 CH3 2. НаО 1. (CH3)2...
A:
Q: 2. Circle all the compounds below that will racemize when treated with a weak acid or base. Explain ...
A:
Q: A sample contains unknown amount of Na2CO3 and NaHCO3 . The sample was titrated with 0.103 N HCl sol...
A:
Q: Correct 1. Calculate the amounts of each reactant. 2. Compare the ratio of reactants amounts to the ...
A:
Q: 2. What quantity of heat is associated with the synthesis of 35.6 g of NH3 according to the followin...
A: Nitrogen oxide reacts with water to form ammonia and oxygen. According to the given reaction when on...
Q: 1) Outline reasonable synthetic schemes to convert pentane to 1-tritio-1-butyne 2) Draw a reasonable...
A: Here we have to synthesize 1-tritio-1-butyne from pentane by multistep reactions.
Q: Identify the following properties of arsenic pentabromide and arsenic pentafluoride. a. Chemical for...
A:
Q: What mass of Ba(IO3)2 (487 g/mol) can be dissolved in 1 L of water at 25°C? Ba(IO3)2 = Ba2+ + 2 IO3 ...
A: Ksp is the product of concentration of ions of sparingly soluble salt.
Q: How much energy (in kJ) is required to heat 17.73 g of Au(l) from 2254.1 K to 3080.2 K (this substan...
A: Here we are required to find the heat energy required from gold from 2254.1K to 3080.2K
Q: 1. Explain why the behavior of H2 (positive deviation) is different from the behavior of CH4 and NH3...
A: Behavior of H2 is different from that of other gases like NH3,CH4 etc. Z in thermodynamic factor tha...
Q: Manganese commonly occurs in nature as a mineral. The extraction of manganese from the carbonite min...
A: First step : 2MnCO3(s) + O2(g) -------> 2MnO2(s) + 2CO2(g) Second step : 3MnO2(s) + 4Al(s) ---...
Q: 4. Using resonance structures, show how the intermediate is stabilized in the reaction of p-chloroni...
A: It is an example of Nucleophilic aromatic substitution reaction Here we are required to show stabili...
Q: Which of the properties below apply to the polymer that forms when the following monomers react? H H...
A: Monomers which have double or triple bond are react with each other by addition reaction without l...
Q: ). To produce 3.00 L of a 1.90 M solution of sodium hydroxide, how many grams of sodium hydroxide mu...
A: Dear student since you have asked multiple questions but according to guidelines we will solve 1st q...
Q: C. 8.12 d. 3.44 e. 5.28 3. Determine the solubility of the ions that is calculated from the Ksp for ...
A:
Q: - the AH value
A:
Q: Consider only the first dissociation constant of a 10-4 M solution of boric acid (H3BO3): a) develop...
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for you...
Q: Which member of each of the following pairs would you expect point, and why? O2 or Brz NO2 or CO2 HF...
A: Boiling point is the temperature at which vapor pressure of liquid is equals to atmospheric pressure...
Q: 12 of 22 I Review | Constants | Periodic Table Part B The following system is at equilibrium:A (s) +...
A: In the given question we have to solve the problem by Applying Le chatelier's principle on the follo...
Q: Balance the reaction, do not leave any fractions, don't leave anything blank, then answer the questi...
A: Answer: This is a reaction in which Al has been gradually added into the nitric acid solution.
Q: Consider the following reaction for formation of nitric acid from the pollutant nitrogen dioxide rea...
A:
Q: Which of the following is a sugar alcohol? Mannitol Xylulose Trehalose
A: Sugars refer the carbohydrates.Carbohydrates can be defined as the organic compounds made up of carb...
Step by step
Solved in 2 steps with 2 images

- Chemistry Find the most efficient synthetic route for the followinf transformations.1. Which of the following is the strongest nucleophile? Refer to the diagram below with an MeOH, MeSH, etc. 2. Which of the following is true about the stereochemistry of SN1 reaction?a.retention of configuration at the electrophilic centerb.50:50 mixture of retention and inversion of configuration at the electrophilic centerc.inversion of configuration at the electrophilic centerd. slightly more inversion than retention at the electrophilic centerDraw the major organic product of this E1 elimination reaction. Ignore byproducts.
- Draw a detailed mechanism for the chlorination of benzene using Cl2 and FeCl3.1. propose a mechanism for each step of the reported synthetic route, except for the steps marked in redDraw the molecule: Under second-order conditions (strong base/nucleophile), SN2 and E2 reactions may occur simultaneously and compete with each other.Show what products might be expected from the reaction of 2-bromo-3-methylbutane (a moderately hindered 2∘∘ alkyl halide) with sodium ethoxide. Do not worry about sterechemistry inther SN2 product for this problem. Don't forget about stereochemistry.
- Why we need step 3 before step 4? a. Because the nitro group increases the electrophilicity at the ortho positions which is where the bromine is added. b. Because the amine group is a strong ortho, para director which is what controls the regiochemical outcome of this bromination. c. Step 4 is unessesary. The symmetry of compound 3 allows for the bromination to be regioselective and give compound 5. 5. There will be a mixture of products because there is no selectivity for a major product.How does changing the alkyl halide from CH3CH2Br to (CH3)2CHBr affect the rate of an E2 reaction?1. Draw a reasonable arrow-pushing mechanism for the transformation shown along. 2. Identify nucleophiles and electrophiles 3. Name any type of reactions taking place like E2 or E1 4. Account for any regio- or stereoselectivity

