Q: Show how to prepare the following aromatic amines by aromatic nitration, followed by reduction. You…
A: The following c) m-bromoaniline (d) m-aminobenzoic acid can be prepared by aromatic nitration,…
Q: What is the most likely organic product of the exhaustive hydrolysis of PhCN? A. benzoic acid B.…
A: The question is based on the concept of organic reactions. We have to identify the product formed…
Q: Which alkyl halide cannot be used effectively in a Gabriel synthesis of amines? Select one: a.…
A: Gabriel synthesis is a method which is used for the preparation of primary amines. In this reaction,…
Q: „NH2 CH3 - aniline target compound CH3
A: In the given reaction the aniline is converted into the target molecule by several steps.
Q: a. Propose a mechanism for the formation of succinic anhydride in the presence of acetic…
A: a. The mechanism for the formation of succinic anhydride is as follows: The succinic acid…
Q: Show how you would use anhydrides to synthesize the following compounds. In eachcase, explain why an…
A: a.
Q: What must be the starting material to carry out the following reaction? a) butanamide b)…
A: This is a reduction reaction.
Q: Several additional amine syntheses are effectively limited to making primary amines. The reduction…
A: (a) Reaction between allyl bromide with sodium azide to give allyl aide. This allyl azide is reduced…
Q: Which nitrogen atom in each compound is more basic?
A:
Q: Which alkyl halide cannot be used effectively in a Gabriel synthesis of amines? Select one: a.…
A: The Gabriel synthesis is a chemical reaction that transforms primary alkyl halides into primary…
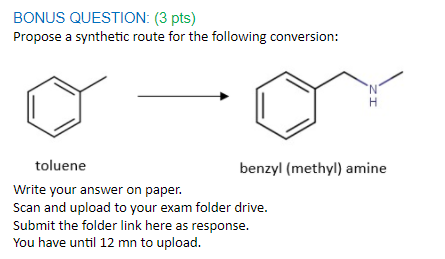
Q: Propose a synthetic route for the following conversion: toluene benzyl (methyl) amine
A: The conversion required: toluene to benzyl methyl amine.
Q: Arrange the members of each group in order of decreasing basicity: (a) Ammonia, aniline, methylamine…
A: Note: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question…
Q: Propose a synthesis using a reaction of a diazonium salt in at least one step of each conversion. a.…
A: We are authorized to answer three subparts at a time, since you have not mentioned which part you…
Q: ČOEt 1. EtO-Na+ OEt 3. NAOH, H,O, heat EtO 4. HCI, heat 2-Carbethoxy- 4-Butanolactone…
A:
Q: Propose the reaction mechanism for the two possible products formation from the following reaction…
A: The two possible products formation from the reaction of acetophenone and butanal and their…
Q: Show how you would convert aniline to the following compounds.(a) fluorobenzene
A: The conversion of aniline to fluorobenzene takes place via Balz-Schiemann reaction. In this reaction…
Q: Rank the following compounds in order of increasing basicity: L p-nitroaniline H.…
A: Basicity of a molecule is the tendency to donate the lone pair of electrons. Electron withdrawing…
Q: which of the following compounds would be the most basic? A. p-methoxyaniline B. p-nitroaniline C.…
A: Basicity means donate the lone pair of electron Basicity depends on 1) + I and + R
Q: Reduction of an alkyl azide results in the formation of —-. A. an imine B. an oxime C. a tertiary…
A: Alkyl azide have general formula R-N3
Q: 25. Quartenary ammonium salts with hydrocarbon chains are used as detergent BECAUSE the presence of…
A: 25. The structure of quaternary ammonium salt is as shown below: A detergent contains both…
Q: 3. How might you prepare pentylamine from the following starting materials? a) Pentanamide b)…
A: Organic reaction mechanisms.
Q: „CHO NH3 H2. Ni A -H20 60 °C, 90 atm CI
A: Since the N in NH3 is having lone pair electron and C attached to O in the benzene reactant is…
Q: By means of a suitable reaction, show how each of the compounds can be prepared from propionic acid.…
A:
Q: Based on this retrosynthetic analysis, propose a synthesis of the anticoagulant (a substance that…
A: Diphenadione can be produced from the retrosynthetic reaction of diethyl pthalate and the given…
Q: H. エ-Z
A:
Q: Propose a stepwise mechanism for the following reaction of a β-keto ester. Suggest a reason why this…
A: The stepwise mechanism for given reaction is shown below, First step is formation of enolate. The…
Q: Which of the following compounds is the most basic? A. p-nitroaniline B. p-chloroaniline C.…
A: Basicity depends on the stability of conjugate acid forms after accepting the proton.
Q: a. Propose a mechanism for the reaction of acetic anhydride with water.b. How does this mechanism…
A: (a) The reaction of acetic anhydride and water produces two moles of acetic acid. The nucleophile…
Q: Prozac may be prepared via a Mannich reaction using which amine? Benzylamine Methylamine Ethylamine…
A: A chemical reaction in which organic compounds take part is called organic reactions. Organic…
Q: Show the reagent needed to convert butanoic acid to its derivative of i. butanoyl chloride ii.…
A: In chemistry, reagent can be defined as the substance which is used to convert reactant into…
Q: Match each of the following with the correct answer. v Benzoyl chloride and propan-2-amine A. Butyl…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: The sex attractant A produced by Anomala cuprea is derived from oleic acid. Propose a biosynthetic…
A: Oleic acid is obtained in a natural way from animal and vegetable oils. Thus, it is also known as…
Q: Propose a synthesis for diphenhydramine starting from benzophenone, benzoic acid, and 2-(N,…
A: Synthetic method for diphenhydramine: Benzophenone undergoes reduction using sodium borohydride on…
Q: Rank the following compounds in order of increasing basicity: 1.p-nitroaniline I p-aminobenzaldehyde…
A:
Q: Propose a mechanism for the reaction of bisphenol A with phosgene.
A: Structure of bisphenol A and phosgene are as follows.
Q: Propose a synthesis of the topical anesthetic cyclomethycaine from 4-hydroxybenzoic acid,…
A:
Q: Show how to prepare the following aromatic amines by aromatic nitration, followed by reduction. You…
A: The structure of p-bromoaniline is shown below.
Q: Show how to prepare the following aromatic amines by aromatic nitration, followed by reduction. You…
A: Nitration is chemical process in which nitro group is introduced into an organic compound.
Q: Write the equation of the reaction of the following reagent with acetaldehyde: a. 2,4-DNP b.…
A: Hey, since you are posted a question with multiple sub-parts, we will answer first three sub-parts.…
Q: In the presence of a trace of acid, d@hydroxyvaleric acid forms a cyclic ester…
A:
Q: a) What are the difficulties with the following reaction to synthesize an amine. + HBr Br + NH NH2…
A: Given reaction is :
Q: Which of the following reaction or reagent does not produce an amine molecule? A. Curtius…
A: The given reactions are A. Curtius rearrangement B. Reaction of oximes with metallic sodium in…
Q: 1. Predict the products or the reaction of (i) phenylacetaldehyde and (ii) acetophenone with the…
A: Organic reactions are those in which organic reactant react to form organic products.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- please quickly thanks ! 3.Please write out the major reaction and side-reaction in the preparation of ter-butyl.chloride, and write out the key points to use separation funnel in this process.I am working on a practice assignment for my organic II course and am having difficulty with a question that asks to identify the reaction sequence used to synthesize isopropylcyclopentane. I would really appreciate the help!How do I report this data? Blue is background data, red is compound 1-bromo-3-phenylpropane, synthesized from 3-phenyl-1-propanol using Nabr and h2so4.
- Need help showing the reaction mechanism between LDA and 2-chloropyridine. Show all reaction steps and transition states. Also include interactions and brackets as well as show all products and byproducts. Thank you :)1. Draw a complete mechanism for the acid-catalyzed esterification of acetic acidwith ethanol. Then answer the follow-up questions:a) Every step in this reaction is an equilibrium. Comment on all of the possibleways to drive the equilibrium (there should be four ways.)b) If some carboxylic acid remains at the end of the reaction, what are somepossible ways to get rid of it?c) If excess alcohol remains at the end of the experiment, what are some possibleways to get rid of it?Calculate the theoretical yield of virstatin ethyl ester and virstatin. Procedure – Step 1 Synthesis of virstatin ethyl ester (with notes and musings about procedural steps in italics) 1,8-naphthalimide (0.25 g) and potassium carbonate (0.26g) are added to a 10 ml round bottom flask containing a stir bar. 1.3 ml DMF (N,N-dimethylformamide) is added to the flask. The DMF will be in a “sure seal” bottle with a septum. You will obtain the DMF using a syringe and needle. Wear gloves when obtaining and dispensing the DMF. Add ethyl 4-bromobutanoate (0.27 ml) (it might say ethyl 4-bromobutyrate on the bottle) to the reaction flask, using an automatic pipet. Heat under reflux, with stirring, for 1 hour. What will the sand temperature have to be to get DMF to boil?? When would you start heating the sand!? After cooling to room temperature add 5 ml water to the reaction solution and cool the resulting mixture in an ice bath. Using a Buchner funnel, vacuum filter the ice cold…
- How different would step 4 be if the amine was secondary? Show curved arrows and explain the mechanism!An organic chemistry experiment involves a nucleophilic substitution (SN2) reaction using 1-iodobutane and 2-naphthoxide ion. The 2-naphthoxide ion is generated from 2-naphthol and sodium hydroxide in an ethanol solvent. The experiment includes steps like generating the nucleophile, conducting the SN2 reaction, and analyzing the product using 1H NMR spectroscopy. Question: Write the structure of the undesired side product if CH3CH2O‾ (ethoxide) reacts with 1-iodobutane.Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…
- Draw the product of the reactions of succinic anhydride with each of the listed reagents. Assume in all cases that the reagent is present in excess and an aqueous workup takes place after each reaction. A. Reaction with (CH3)2CHOH. \table[[Select,Draw,Templates More,,],[/,//,I//,H,Erase]] B. Reaction with NH3. \table[[Select,Draw,tes More,Erase],[/,//,III,C,H,N,0]] Question Source: Vollhardt 7e0 Organic Chemistry: Structure And Function Publishęr: W. Freemar C. Reaction with phenylmagnesium bromide in THF, followed by H+,H2o. \table[[Select,Drav,v T,lates,,,Erase],[1,II,III,C,H,0,]] D. Reaction with LiAlH4 in ether, followed by H+,H2O \table[[Select,Draw,Templates More],[/,//,III]] \table[[3,0]] \table[[,2,QA. Give a product that contains carboxylic acid found in the market. Name of product Carboxylic acid present and its uses B. Give a product that contains ester found in the market. Name of product Ester present and its uses I WILL UPVOTE BUT PLS BE INFORMATIVE, THANK YOUUUSelf-condensation of acetone is a possible side reaction for the aldol condensation(Reaction 1). Draw the structure of this side product. How is this side reaction minimizedin our procedure written below? Reaction 1 - Experimental Procedure: In a clean, small Erlenmeyer flask, combine your unknownaromatic aldehyde (350 μL (400 mg if it is a solid)), 95% ethanol (2.0 mL), and 5 N aqueoussodium hydroxide (0.6 mL). Swirl to mix, and then add acetone (100 μL). Stir/swirl vigorously for30 minutes, until precipitation is complete. If no precipitate forms after 15 minutes, heat theErlenmeyer flask gently in on the sand bath in the hood for 15 minutes, and then allow it to coolto room temperature. Whether heated or not, cool your Erlenmeyer flask in an ice bath for 10minutes to ensure complete crystallization. Collect the product by vacuum filtration. (If you donot have any solid at this point, evaporate all solvent in the sand bath. Recrystallize if solidresidue is obtained; otherwise stop…



