Proteins are characterized by their isoelectric point (pl), at which they carry no net charge. Proteins dissolved in a buffer solution with pH larger than the isoelectric point are negatively charged and positively charged when they are present in solution with a pH smaller than their isoelectric point. Consider a protein mixture consisting of protein A with pl-5.1 (MW 10,000), protein B with pl-7.1 (MW 10,000) protein C with pl-9.6 (MW 10,000) which will be separated by Capillary Zone Electrophoresis. Determine the migration order (fastest to slowest) in a buffer solution at neutral pH (negative potential applied to outlet and positive potential applied to the inlet, assume negative charge at capillary wall).
Proteins are characterized by their isoelectric point (pl), at which they carry no net charge. Proteins dissolved in a buffer solution with pH larger than the isoelectric point are negatively charged and positively charged when they are present in solution with a pH smaller than their isoelectric point. Consider a protein mixture consisting of protein A with pl-5.1 (MW 10,000), protein B with pl-7.1 (MW 10,000) protein C with pl-9.6 (MW 10,000) which will be separated by Capillary Zone Electrophoresis. Determine the migration order (fastest to slowest) in a buffer solution at neutral pH (negative potential applied to outlet and positive potential applied to the inlet, assume negative charge at capillary wall).
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter20: Molecular Spectroscopy And Photochemistry
Section: Chapter Questions
Problem 67CP
Related questions
Question
6
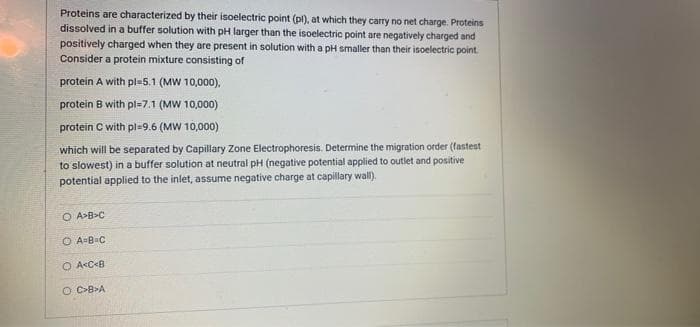
Transcribed Image Text:Proteins are characterized by their isoelectric point (pl), at which they carry no net charge. Proteins
dissolved in a buffer solution with pH larger than the isoelectric point are negatively charged and
positively charged when they are present in solution with a pH smaller than their isoelectric point.
Consider a protein mixture consisting of
protein A with pl=5.1 (MW 10,000),
protein B with pl=7.1 (MW 10,000)
protein C with pl=9.6 (MW 10,000)
which will be separated by Capillary Zone Electrophoresis. Determine the migration order (fastest
to slowest) in a buffer solution at neutral pH (negative potential applied to outlet and positive
potential applied to the inlet, assume negative charge at capillary wall).
O A>B>C
O A=B=C
O AcC<8
O C-B>A
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning