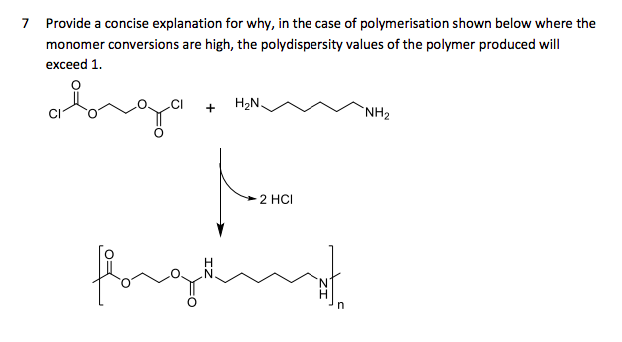
Provide a concise explanation for why, in the case of polymerisation shown below where the monomer conversions are high, the polydispersity values of the polymer produced will exceed 1. H2N. `NH2 2 HCI
Q: Write the repeating unit of the polymer formed from the following reaction and propose a mechanism…
A: Given: The repeating unit contains diepoxide and diamine groups so the polymerization of these…
Q: а. Describe the polymerisation of 2-hydroxy ethylmethacrylate, in the form of chemical equations.…
A: 2 hydroxy ethyl methyl Acrylate polymerises to form long chain polymer. Given in the image.
Q: How many grams of H2O2 would be needed to yield 1 kg of a polypropylene, (C3H6 )n, with an average…
A: The degree of polymerization is= MwMoMw is the average molecular weight of the polymer and Mo is the…
Q: Illustrate, with chemical formulae, the influence of the size of the side groups of a polymer…
A: Polymer is a large molecule which is made up of the small repeating units. These molecules have the…
Q: #5: Estimate the full contour length of a low-density polyethylene (LDPE) (CAS# 9002-88-4) chain…
A: average degree of polymerization is a weighted mean of the degrees of polymerization of polymer…
Q: 1. List the following group of monomers in decreasing order of the ability to undergo cationic…
A: 1-The ability of monomers to undergo cationic polymerization is based upon the formation of the…
Q: What is the first step in the cationic polymerization of alkenes?
A:
Q: For a monomer of general structure of CH2=CHX, complete the following table. Indicate + if the…
A: Polymerization occurs through cationic pathway if electron donating group (EDG) is present. This is…
Q: Explain why radical polymerization of styrene forms branched chains with 4° carbons as in A, but…
A: The radical polymerization of styrene results in the formation of benzyl radical which is stabilized…
Q: . Show 2 repeating units of the polymer produced by styrene - refer the diagram attached H₂C-C H-O…
A:
Q: 4. Consider the free radical polymerization of methyl methacrylate (shown below) using AIBN as the…
A: Free radical polymerization of Methyl methacrylate occurs in three steps using AIBN as a radical…
Q: Provide a concise explanation for why, in the case of polymerisation shown below where the monomer…
A: A question based on polymer chemistry, which is to be accomplished.
Q: 17. Rank the following monomers in order of increasing reactivity toward cationic polymerization…
A: fullscreen_exit
Q: Draw the repeat unit of the polymer produced by cationic polymerization of 1,3-dioxolane using HBF4,…
A: It is asked to draw the repeating unit of the polymerization of Tetrahydro furan.
Q: 1. Draw 2D chemical representations of atactic, syndiotactic, and isotactic polystyrene. 2.…
A: First three question answered below:
Q: High pressure, high temperature free-radical p
A: The repeating structural units are usually obtained from low molecular mass simple compounds called…
Q: On the same axes, sketch the extent of reaction (p) vs time for (i) uncatalyzed and (ii) catalyzed…
A:
Q: 2. For the monomers below, state whether they will undergo chain growth or step growth…
A:
Q: #2: Draw/sketch a polyacrylonitrile (CAS# 25014-41-9) architecture with a i) linear structure and…
A: The degree of polymerization (DP) is the number of monomeric units in a macromolecule or polymer or…
Q: Poly(ethylene terephthalate) (PET) can be prepared by this reaction. Propose a mecha- nism for the…
A: Poly(ethylene terephthalate) is made by reacting Dimethyl terephthalate and ethylene glycol in the…
Q: ROOR n RO
A: The initiation and propagation step of this radical polymerization reaction given below .…
Q: Questicn 3: Vinyl alcohol is polymerized by free radical polymerization to form long polymer chains.…
A: Polymers are the macro molecules which are made by the repetition of smaller units. These small…
Q: You are preparing polyesters from the following two monomers: CH,OH CH3CH2 C-CH,OH ČH,OH Co.H HO,C…
A: Polymerization can be described as a chemical process that results in the creation of polymers in…
Q: vill more likely involve addition polymerization reaction or condensation olymerization reaction:…
A: Addition polymerization involves only one kind of monomer whereas condensation polymerization…
Q: 3.2 Using acrylic acid as a monomer, CH₂CH(COOH), (or look up the structure on-line), illustrate A.…
A:
Q: Provide the structure of a region of the polymer that is produced when the following monomers are…
A: The structure of the polymer -
Q: Explain why cationic polymerization is an effective method of polymerizing CH2 = C(CH3)2 but not CH2…
A: One type of chain growth polymerization process is cationic polymerization. It involves…
Q: 7. When asymmetric alkenes polymerize (such as chloroethene to produce PVC), the pendant group like…
A: Sporadic arrangement is the random arrangement of the polymeric chains / branches . Where Atactic…
Q: One common type of cation exchange resin is prepared by polymerization of a mixture containing…
A: The structure formula of the polymer, the resin is shown in the acid or protonated form.
Q: Provide a concise explanation for why, in the case of polymerisation shown below where the monomer…
A: Polymerization reaction is classified generally into two types that is addition polymerization and…
Q: نقطة واح The degree of polymerization .determines the weight of polymer True False O محو التحديد…
A:
Q: CH2 -CH2 -0- -(CH)4 One or more of the monomers below are needed to make the polymer Choose which…
A:
Q: >1,3-Cyclopentadiene undergoes thermal polymerization to yield a polymer that has no double bonds…
A:
Q: Q1. (a) In each of the following cases (I. to III.) identify which polymer in each pair has the…
A: a) pair 1- polymer 2 has higher Tg. Reason- because the presence of aromatic, cyclic structure in…
Q: -(CH2 -(CH) One or more of the monomers below are needed to make the polymer Choose which one(s). но…
A:
Q: Explain why radical polymerization of styrene forms branched chains with 4° carbons as in A, but…
A: Given: products of the radical polymerization of styrene.
Q: Outline methods for the production of vinyl polymers using bulk methods
A: Vinyl polymers are polymers made from vinyl monomers i.e. small molecules containing carbon-carbon…
Q: Draw the structure of the polymer that results from anionic polymerization of…
A: Given,Anionic polymerization of p-trichloro methyl styrene (CCl3C6H4CH =CH2) using ethylene oxide as…
Q: for CH2 CH2 O- (CH)a n One or more of the monomers below are needed to make the polymer Choose which…
A:
Q: Identify which is suitable for radical, cataionic, anaionic, condensation polymerization, and give…
A: Here we have to predict the types of polymerization occur and structure of polymer formed.
Q: Write the mechanism of acid-catalyzed polymerization of ethylene to polythene.
A: This is the polymer reaction of ethylene
Q: 5.In the polycondensation of H2N(CH2)6NH2 and HOOC(CH2)4COOH, in equimolar quantities, to form…
A: Although the efficiency of a reaction can be measured, by calculating the yield (percentage yield).…
Q: .5(a) Which is the first conductive polymer? How intrinsic conductive polymer can be prepared. (b)…
A: a.1st conductive polymer is polyacetelene or polyethyne. Polymers that conduct electricity is called…
Q: For an alkene to polymerize easily via anionic polymerization... A) Atoms adjacent to the…
A: The correct option is given as:
Q: Explain why radical polymerization of styrene forms branched chains with 4° carbons as in A, but…
A: The radical formed in radical-Polymerization of styrene is The radical formed in X is benzyl free…
Q: Which of the following provides an absolute measure of the molecular weight of polymers: (a)…
A:
Step by step
Solved in 2 steps

- Give at least two methods for the preparation of 2-methylpropene. What happens if it is treated with: a) H2O/H+; b) HBr/ROOR; c) polymerization. In the case of the reaction with H2O/H+ write the mechanism.What are the fundamental differences between a good solvent and a theta solvent, and how do they effect polymer configuration?Describe the polymerization of 2,2-dimethyloxirane by an anionic mechanism.
- A3 Anionic polymerization of styrene is generally known to produce polymers with a narrow molecular weight distribution. Explain the reason for this.9a. Give a complete reaction scheme for the polymerization of styrene with tert-butylhydroperoxide as an initiator and termination by disproportionation.9a. Give a complete reaction scheme for the polymerization of styrene with tert-butylhydroperoxide as an initiator and termination by disproportionate.
- Which polymer is more flexible at room temperature: poly(methyl acrylate) (CAS# 9003-21-8) or poly(methyl methacrylate) (CAS# 9011-14-7)? Why? Use your knowledge of the relationship between polymer structure and its Tg to help explain and support your answer8.For end-group analysis, 0.8632 g of a carboxyl terminated polybutadiene (CTPB) sample dissolved in 1:3 mixture of ethanol and toluene consumed 5.2 ml of 0.1240 N alcoholic potassium hydroxide solution in titration using phenolphthalein as the indicator. Calculate the molar mass of the polymer.One common type of cation exchange resin is prepared by polymerization of a mixture containing styrene and 1,4-divinylbenzene . The polymer is then treated with concentrated sulfuric acid to sulfonate a majority of the aromatic rings in the polymer. Q.) Show the product of sulfonation of each benzene ring.
- Drawing on what you know about the stereochemistry of alkene addition reactions, a. write the mechanism for the reaction of 2-butyne with one equivalent of Br2. b. predict the configuration of the product of the reaction.What prpduct would you expect from the reaction of cyclopentene with NBS and water? show the stereochemistry? Please draw up the reaction and all the chemistry formed.I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?

