Q: [References] a. Will dichromate ions oxidize chloride ions to free chlorine in acidic solution?…
A: Oxidising agent can be defined as the species that oxidises other species. Oxidation means loss of…
Q: 3. Dry ice is the name for solid carbon dioxide. Instead of melting, solid carbon dioxide sublimes…
A: Answer: This question is based on energy conservation where heat lost by water during cooling will…
Q: Determine the lattice constant, a (in nm), of a metal conforming to the BCC structure having an…
A:
Q: 3. Chloroform, an important solvent, is prepared by the chlorination of methane at 721 K. CHa(g) +…
A: Given reaction: CH4 (g) + Cl2(g) ----> CH3Cl(g) + HCl(g) The thermodynamic standard gibbs…
Q: In the reaction shown, the Cm is known as the nucleus, and the Pu as the nucl 242 Cm 96 238 94 Pu…
A:
Q: For each reaction, calculate AHn, ASn , and AGn at 25 °C and state whether or not rxn rxn the…
A:
Q: A 48.2 mL sample of gas in a cylinder is warmed from 28°C to 82°C. What is the volume at the final…
A:
Q: Initial Final Electrode A: 10.345 g 10.278 g Electrode B: 14.010 g 14.887 g Current 0.3942 A time 39…
A: Given: Mass lost by electrode = 0.067 grams (g)Current = 0.3942 ATime of electrolysis = 39min 50s =…
Q: x-1-2 = 0- x = +3 NH3 LANH3 NH3 ONO ove Name of complex #3: Pentamminenitro-O-Cobalt (III) Chloride…
A: We have to find the oxidation state of central metal atom then see the linkage isomers and according…
Q: ОН ÇH3 ÇH3 CH3 H3C-O. `CH3 10 H3C-O `CH3 ОН This compound is called type your answer...
A: This is a compound having high molecular weight and having phenolic-OH and methoxy group.
Q: What does the microphone and transducers in your cellphone do with your voices?
A:
Q: A voltaic electrochemical cell is constructed using the following reaction. The half-cell components…
A:
Q: 33. Which of the following equations would correspond to the bicarbonate ion, HCO3', behaving as a…
A: In this question we have to tell the equation in which bicarbonate ions work as weak acid.
Q: Question 9: Predict the product of the following reaction. CI AICI3 A. В. С. D. CI B.
A: In this question we have to tell product of the reaction.
Q: Using the thermodynamic information, calculate the standard reaction free energy for the reaction…
A:
Q: 1.2. If you have 50.0 mL of 0.456 M Mgl2 solution, what is the concentration of iodide ions in the…
A: Given, Concentration of MgI2 solution = 0.456 M = 0.456 mol/L Volume of the solution = 50.0 mL =…
Q: Suppose a gas is in a vessel connected to both an open-end and a closed-end manometer. The…
A: Solution - According to the question - Mercury level are at = 225mm Pressure of the gas sample =…
Q: Calculate the total binding energy in kJ per mole nuclide and in kJ/mol nucleons for the following…
A: To calculate the total binding energy in kJ per mole nuclide and in kJ/mol nucleons for the…
Q: Which substances in the equation below are classified as Bronsted-Lowry ACIDS: H2S (aq) + H2O0 → H30…
A:
Q: 40. You mix 50.0 mL of 0.10 M HCl with 40.0 mL of 0.10 M NaOH. What do you know about the final…
A:
Q: A) What is the Chemical Formula and number of unshared electron PAIRS in the skeletal structure…
A:
Q: Alkenes are a(n) hydrocarbons. a) saturated b) unsaturated
A: General formula for alkenes : CnH2n If n = 2 , formula is C2H4 If n = 3, formula is C3H6
Q: What is the calculated value of the cell potential at 298K for an electrochemical cell with the…
A:
Q: For numbers 6-10, the process steps of an ideal cycle operating between two heat reservoirs are laid…
A: In thermodynamics, an isothermal process is a type of thermodynamic process in which the temperature…
Q: Suppose a tank of oxygen-enriched air prepared for scuba diving has a volume of 11.04 L and a…
A: Partial pressure of oxygen can be used to calculate the number of moles of oxygen present in the…
Q: Determine the major product of the given reaction below by drawing the complete reaction mechanism.…
A: Detail mechanistic pathway is given below
Q: Determine the type of the following chemical reaction: CuCl2(aq) + NaOH(aq) -------------->…
A:
Q: 45. If a chemical reaction has a positive change in entropy, AS, then: A) the reaction is…
A: As it is a multiple question and no specific question is asked to answer so i am solving first…
Q: T Write out the mechanism for the following reaction. Show all steps and use 1. curved arrows. H20 +…
A: Given,
Q: EXPERIMENTATION - Exercise 1 Observing Boyle's Law In this exercise, you will increase the pressure…
A: The solution is given below -
Q: enthalpy of fusion (melting) of ice is +6.01 kJ/mol at 0.00°C. What is the energy change when 49.0 g…
A: Given, Mass of ice = 49.0 g Molar mass of Ice = 18 g Moles of ice =mass/molar mass = 49.0/18 =…
Q: The following reaction is endothermic: 30,g)= 20,g) If the reaction is at equilibrium, tell whether…
A:
Q: Question 3 Which of the following is the definition of the rate-determining step of a reaction? a)…
A: Definition of rate -determining step of a reaction:
Q: hd See Periodic Table See Hint Calculate the pH of 1.25×10M solution of selenocysteine (pK,1 = 2.21.…
A:
Q: F. B(OH)2 Ni(OTf)2 DPPE K2CO3 (a) Give the product of the reaction and suggest a plausible catalytic…
A:
Q: A student is using a calorimeter to determine the specific heat of a metallic sample. She measures…
A: Given, For metal Mass of metal (mmetal) = 128.1 g Specific heat of metal (Cpmetal) = ? Initial…
Q: Use the space provided below to draw a flow chart showing how you could separate a mixture…
A: The flow chart for the extraction of given compounds has been shown below:
Q: Which of the following statements is/are correct? I. Positron emission decreases the mass number of…
A: Positron emission occurs when a proton in a radioactive nucleus changes into a neutron and in that…
Q: The unknown compound that gives the following IR and NMR spectra has the formula C4H9B1. Based on…
A: IR shows the different functional group present in the compound. NMR shows different peaks for…
Q: 4. Propose a synthesis scheme for the amine shown below using the starting material indicated. No…
A: Here we have to prepare isopropyl amine from phthalimide by multistep reactions.
Q: How many o and a bonds are in each compound? :C number of o bonds: number of r bonds: number of o…
A: σ bond: σ bonds are formed by end to end overlapping of atomic orbitals. Overlapping occurs along…
Q: In order to determine the identity of a metal, its specific heat is found. When 28 g of the metal…
A:
Q: Given the rate constant, k, for the decay of a radioactive substance, how would you find the…
A: Given :- rate constant for decay of radioactive substance = k To find :- half-life (t1/2)
Q: Aside from repeating the experiment, search literature and suggest what can be done to remedy a…
A: Remedy for a sample that is ‘over-titrated' 1 - Over-titration is a state where there is more…
Q: 6. Write abbreviated formulas for the six isomeric tripeptides of aspartate, glutamate, and proline.
A: Six isomeric tripeptides of aspartate, glutamate and proline is given below in step no. 2:
Q: 2. How many of the following statements are true? I. Kinetically fast reactions always have large…
A: According to rate law rate of reaction = Kf [reactant]a Where a could be number like 0,1,2,3 .…
Q: The density of Cr is 7.20 g cm3, It its unit cell is a cube with an edge length of 0.289 nm,…
A: Formula for this is Density = Mass of unit-cell / volume of unit cell.
Q: 4. A peptide bond is classified as a(n) C. phosphate bond. A. ester B. amine D. amide E. none of…
A:
Q: 5. Base pairing in DNA is accomplished through A. disulfide bonds B. hydrogen bonding C. peptide…
A: Base pairing in DNA accomplished through
Q: Current Attempt in Progress Calculate the binding energy per nucleon for the carbon-12 nucleus,…
A:
Step by step
Solved in 2 steps with 1 images

- Answer the following questions about the mechanism for the acid-catalyzed hydration of an alkene: a. How many transition states are there? b. How many intermediates are there? c. Which step in the forward direction has the smallest rate constant?What other alkene is also formed along with Y in Sample Problem 9.3? What alkenes would form from X if no carbocation rearrangement occurred?An alkene G (C6H12) reacts with cold basic KMnO4 to produce H (C6H14O2). Hydrogenation of Gproduces I (C6H14). Ozonolysis of G produces propanone (CH3COCH3) and propanal(CH3CH2CHO).a) Draw the structure of G, H, I.b) State the reagent(s) and condition for ozonolysis reaction
- A hydrocarbon of unknown structure has the formula C8H10. On catalytic hydrogenation over the Lindlar catalyst, 1 equivalent of Η2 is absorbed. On hydrogenation overa palladium catalyst, 3 equivalents of Η2 are absorbed. (a) How many degrees of unsaturation are present in the unknown structure? (b) How many triple bonds are Present? (c) How many double bonds are present? (d) How many rings are present? (e) Draw a structure that fits the data.What steps are needed to convert Y to the alkene Z ?Replacing a kin, a word each of the following our kids results in a significantly lower reaction. Identify the reasons for this decrease in rate for each alkene D – F. Select all that apply. 
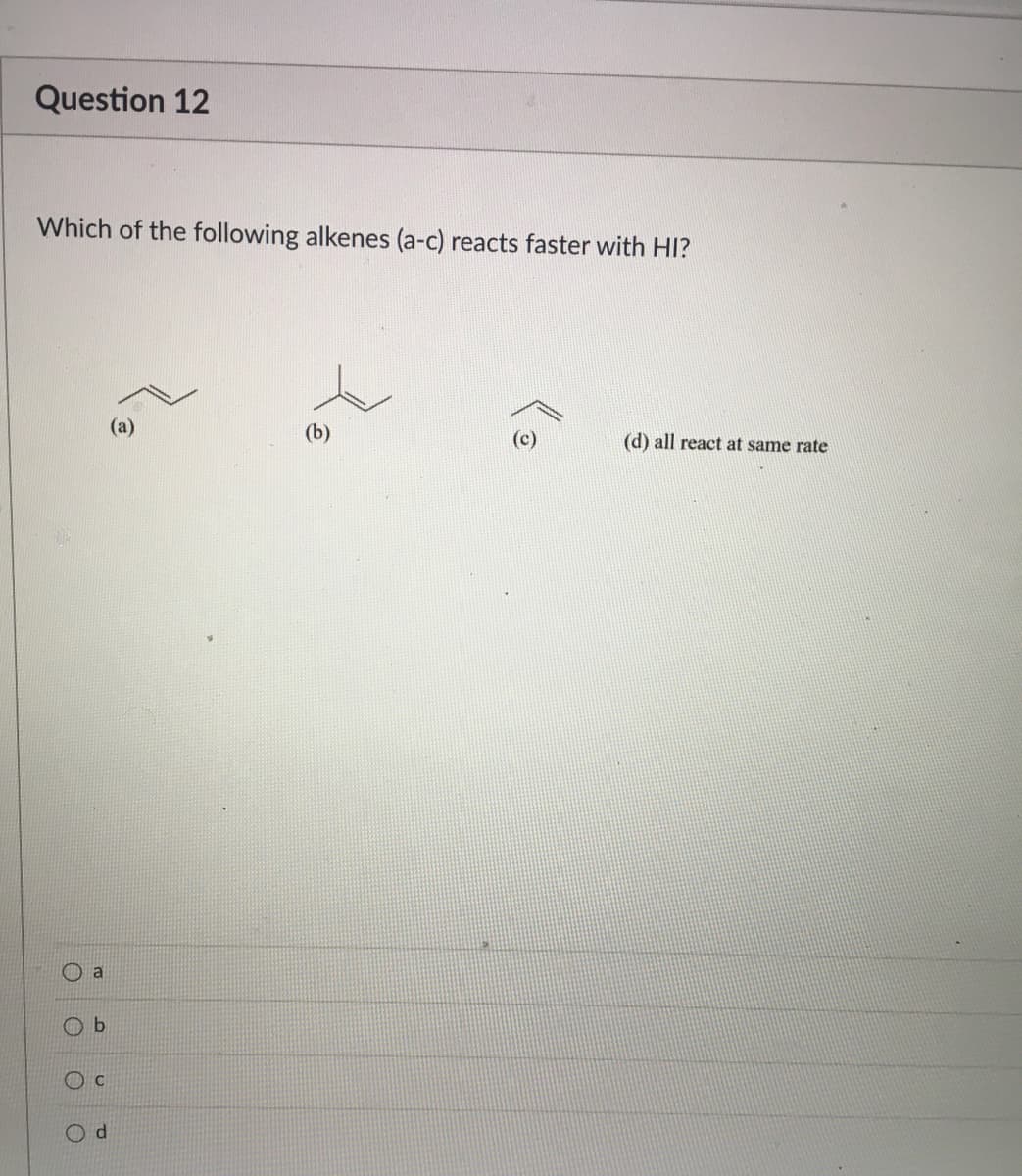
- Consider a reaction where cis-but-2-ene is treated with sO followed by NaHSO/H, O. Draw the structure of one product that is formed in the reaction, including correct stereochemistry.Which of the following alkenes (a-c) reacts faster with HI?13.30c3 Add curved arrow(s) to draw step 3 of the mechanism. Modify the given drawing of the product as needed to show the intermediate that is formed in this step.
- Identify each of the following species as either nucleophilic or electrophilic: Cl-, H+, and carbon in H3C+Following is a balanced equation for bromination of toluene.(a) Using the values for bond dissociation enthalpies given in Appendix 3,calculate ∆H0for this reaction.(b) Propose a pair of chain propagation steps and show that they add up to theobserved reaction.(c) Calculate ∆H0for each chain propagation step.(d) Which propagation step is rate-determininThere are two isomeric cyclohexa-1,4-diene products when toluene undergoes the Birch reduction (see Problem 25.24). (a) Draw the mechanism that leads to the formation of the major product. (b) Will the Birchreduction of toluene occur faster or slower than the Birch reduction of benzene itself? Hint: Is –CH3 an electron-donating or an electron-withdrawing group?

