QUESTION 17 What is the equation that relates Keq with AE'o? (Hint: Rearrange two different equations). 'In (Keq) = (NFAE'o)/(RT) 'In (Keq) = (RT)/(NFAE') 'In (Keq) = (nF)/(AE'•RT) In (Keq) = (n)/(FAE'oRT) QUESTION 18 Calculate the equilibrium constant for this reaction at 25°c by using the standard reduction potentials given in the given Table. 1.45 x 104 1.62 x 105 6.19 x 10-6
QUESTION 17 What is the equation that relates Keq with AE'o? (Hint: Rearrange two different equations). 'In (Keq) = (NFAE'o)/(RT) 'In (Keq) = (RT)/(NFAE') 'In (Keq) = (nF)/(AE'•RT) In (Keq) = (n)/(FAE'oRT) QUESTION 18 Calculate the equilibrium constant for this reaction at 25°c by using the standard reduction potentials given in the given Table. 1.45 x 104 1.62 x 105 6.19 x 10-6
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter18: Glycolysis
Section: Chapter Questions
Problem 23P: Examine the ActiveModel for alcohol dehydrogenase and describe the structure and function of the...
Related questions
Question
Can someone help with this question?
|
L-Malate + NAD⁺ ← → Oxaloacetate + NADH |
Above is what i selected for the equation, but I don't know how to find the equilibrium constant.
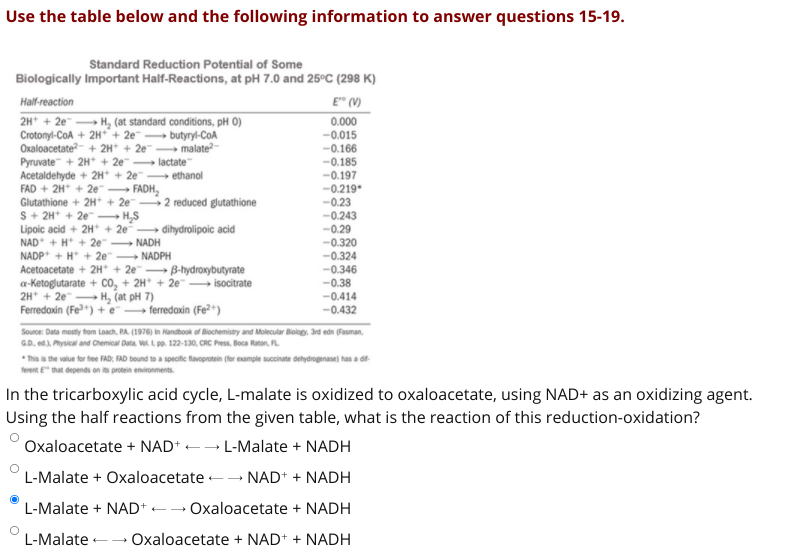
Transcribed Image Text:Use the table below and the following information to answer questions 15-19.
Standard Reduction Potential of Some
Biologically Important Half-Reactions, at pH 7.0 and 25°C (298 K)
Half-reaction
E" (V)
0.000
2H* + 2e H, (at standard conditions, pH 0)
Crotonyl-CoA + 2H* + 2e¯ → butyryl-COA
Oxaloacetate- + 2H* + 2e malate-
Pyruvate + 2H* + 2e lactate
Acetaldehyde + 2H* + 2e
FAD + 2H + 2e
Glutathione + 2H* + 2e 2 reduced glutathione
S+ 2H* + 2e H,S
Lipoic acid + 2H* + 2e dihydrolipoic acid
NAD* + H* + 2e" NADH
NADP* + H* + 2e" NADPH
Acetoacetate + 2H* + 2e B-hydroxybutyrate
a-Ketoglutarate + co, + 2H* + 2e"→ isocitrate
2H* + 2e -
Ferredaxin (Fe*) + e– ferredaxin (Fe²*)
-0.015
-0.166
-0.185
→ ethanol
-0.197
--0.219*
FADH,
-0.23
-0.243
-0.29
-0.320
-0.324
-0.346
-0.38
-0.414
-0.432
H, (at pH 7)
Source: Data mosty from Loach, PA. (1976) in Nandbook of Blochemistry and Molecular Blology. 3rd edn (Fasman,
GD. ed). Physical and Chemical Duta, Vol. L po. 122-130, CRC Press, Boca Raton, FL
sis the value for fee FAD; FAD bound to a specific favoprotein (for example succinate dehydrogenase) has a d
RE" that depends on s protein enironments.
In the tricarboxylic acid cycle, L-malate is oxidized to oxaloacetate, using NAD+ as an oxidizing agent.
Using the half reactions from the given table, what is the reaction of this reduction-oxidation?
Oxaloacetate + NAD+ -- L-Malate + NADH
L-Malate + Oxaloacetate -- NAD+ + NADH
L-Malate + NAD+ -- Oxaloacetate + NADH
L-Malate -- Oxaloacetate + NAD+ + NADH

Transcribed Image Text:QUESTION 17
What is the equation that relates Keq with AE'o? (Hint: Rearrange two different equations).
° In (Keq) = (NFAE'•)/(RT)
O In (Keg) = (RT)/(NFAE'o)
O In (Keq) = (nF)/(AE'•RT)
In (Keq) - (n)/(FΔΕ'ο RΤ)
QUESTION 18
Calculate the equilibrium constant for this reaction at 25°c by using the standard reduction potentials given in the given Table.
1.45 x 104
1.62х 105
6.19 х 10-6
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 6 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning