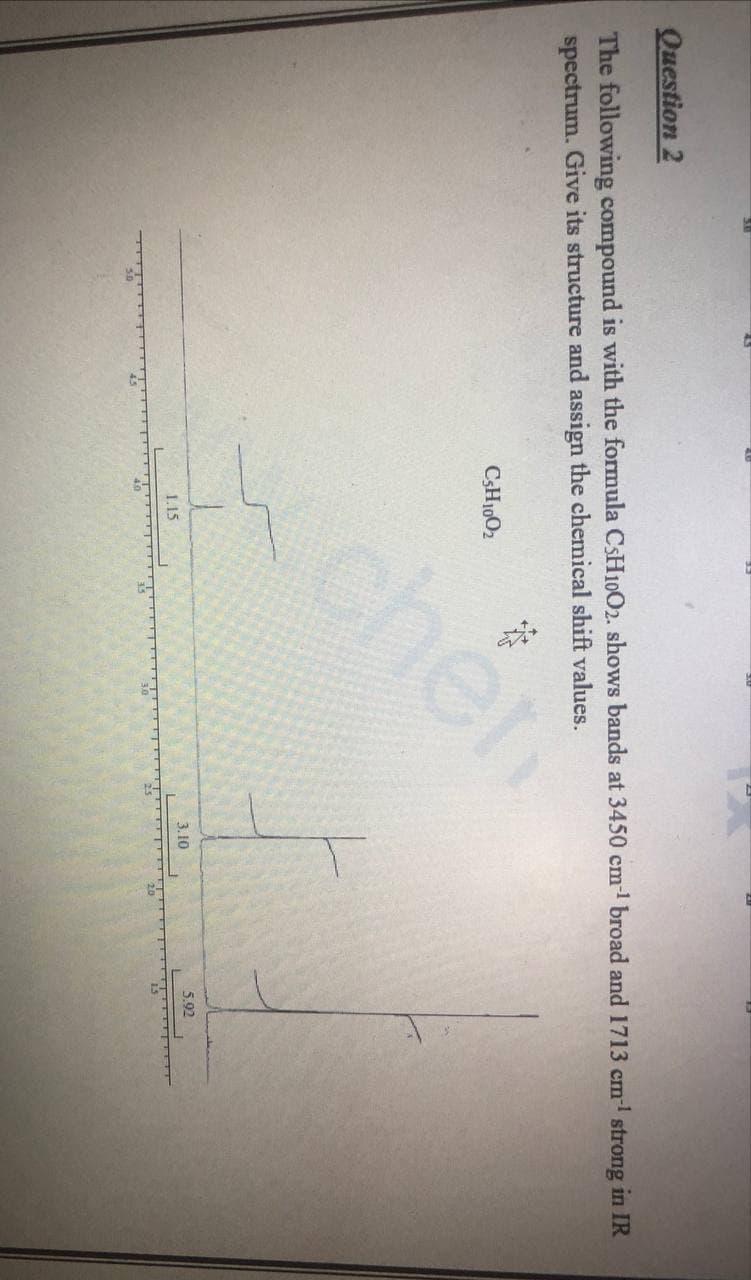
Question 2 The following compound is with the formula C$H10O2. shows bands at 3450 cm1 broad and 1713 cm- strong in IR spectrum. Give its structure and assign the chemical shift values. CSH1002 chen 1.15 3.10 5.92 20
Q: 5. IR Spectrum Identify three bands on the spectrum that are consistent with your structure. b…
A:
Q: Assign the important peaks in the IR spectrum below and identify the name of the functional group.…
A:
Q: What is the IR frequency or range best distinguish between the two compounds shown. Specify which…
A: There are many types of spectroscopy like UV Visible spectroscopy, IR spectroscopy, NMR…
Q: The IR spectrum of a compound with molecular formula C 6H 10 is shown below. Suggest a possible…
A: To solve this type of question we must know some characteristic peaks .
Q: (a) A sample of aspirin (acetyl salicyclic acid) of molecular mass 180, on analysis was found to…
A: Given: % of C = 60 % . % of H = 4.4 % . % of O = 35.6 % . Moles = weight / molecular weight…
Q: (9) C2H4B1, 3 1 4 1 2.
A: Since, you have provided the NMR spectra of the given and not the IR spectra, we will provide you…
Q: INFRARED SPECTRUM 0.8 0.6 0.4 0.2 0.0 3000 2000 1000
A: On finding out the characteristic peaks from the IR, compound will be identified.
Q: Examine the IR spectra below. Each arrow points to a significant absorption that indicates a…
A: IR spectra is one of the very important spectroscopic tool to identify the functional groups of…
Q: ОН PhMgBr H3O+ EtzN, CH2CI, Et20 C3H1202
A:
Q: how would the Rf values change if eluted with hexanes using an alumina TLC plate?
A: In chromatography, a stationary phase is a solid, or a liquid supported on a solid (e.g. silica) and…
Q: SOLUBILITY TEST distilled water Litmus paper test OBSERVATIONS clear, colorless solution no change…
A: A question based on qualitative analysis, which is to be accomplished.
Q: Problem attached
A: The absorption of ultraviolet radiations in the UV-visible spectroscopy results in the excitation…
Q: Compound 48 is a low-melting solid (melting point 55-57 °C), that is soluble in dilute aqueous base,…
A: Let's start with the elemental analysis to check the ratio in which different elements are present…
Q: Discuss peak splitting, and draw H(proton) NMR spectra for CH3CH2CH2F structure, label and explain…
A: For the above given molecule, presence of F atom play a key role for deciding the nature of the…
Q: CDCI3 80 75 70 65 60 55 50 45 40 35 30 25 15 10
A: The question is based on the concept of Spectroscopy. we have to assign chemical shift value to…
Q: Below are two molecules, cyclohexylamine (A) and aniline (B). When analyzed by Infrared Spectroscopy…
A: The exact wave number at which vibration occurs depends on the strength of the involved bond. A…
Q: Question 10 Given is teh mass spectogram of an unknown compoound. This unstable cyclic compund is a…
A: Note : The compound name is cyclohexene ( see below).
Q: The product was tested for its ability to absorb UV light by illumined a solution of the compound…
A: Given: The product was tested for its ability to absorb UV light by illuminating a solution of the…
Q: Using IR Spectroscopy to Distinguish Isomers How can the two isomers having molecular formula C2H6O…
A:
Q: Give a parallel description or analogy between proper paraphrasing of textual information and the…
A: Isomerism is the phenomenon in which more than one compound have same molecular formula but…
Q: what is the unknown and please give the reason of your choice using the IUPAC name, structure,…
A: Given, Refractive index = 1.413866 boiling point correction is 119.8 °C We have to…
Q: Question 1. A researcher isolated 10 g of a Natural product from a local plant. In an attempt to…
A:
Q: 1. Look at the spectrum shown and answer the following questions: 1.4 1.2 - 0.4 0.2 24n 280 320…
A: electronic transition from lower energy level to higher energy level leads to formation of energy…
Q: Provide the correct answers in the given statement with the given compounds in the image. A. In…
A: The reactions are as follows:
Q: 1. Theoretical questions a. If two compounds have the same Rf under identical TLC conditions, does…
A: since you are posted a multiple questions we are entitiled to answer only first question, if u get…
Q: A colorless solid that melts at 103 to 105 C was subjected to IR Spectroscopy to determine the…
A: C5H11NO (DBE = 5- (11-1/2)+1 = 6-5 = 1. The compound is primary amide. Therefore the compound is…
Q: The absorbance (A) of a solution is related to its concentration (C) according to the Beer-Lambert…
A: Lambert Beer's law gives the relation of concentration with respect to absorbance as; A=εC where A…
Q: „CN and ČN
A: Conformational sterioisomers wrt X and 2 hydrogens, the groups are the Axial(Upwards) position…
Q: Questions 27 to 31 concern the following organic compounds (A) HOOCCHCHCOOH (B) C:lICHOHCH (C)…
A: 31. The reaction of ammonium cuprous chloride with terminal alkynes gives a red-brown ppt. The…
Q: Which structure fits the following IR-spectrum? 3363 3090 1600 770 690 1528 1352 ENUHO но, Но. CEN…
A: Answer. A option is correct. Given IR spectrum shows the characteristics peaks for the N-O bond…
Q: IR Peaks for different functional group?
A:
Q: 4) Compare the IR spectra of both isomers and show the bond vibrations for functional groups, decide…
A: Linkage isomerism occurs with ambidentate ligands that are capable of coordinating in more than one…
Q: QUESTION 2 An unknown compound B is a saturated compound having a molecular formula of C3H&O.…
A:
Q: Question: 1, Based on Beer-Lambert law, A = ɛ. c. L, where A, absorbance; ɛ, extinction coefficient;…
A:
Q: What is the check digit for the Flamin’ Hot Cheetos with the following UPC: 0 28400 58989
A: A check digit is a form of redundancy check used for error detection on identification numbers,…
Q: Provide the correct structure of a compound that gave the data below MS: M m/2 144.04 relative…
A:
Q: Which set of IR bands (in cm-1) would you expect to see for the following compound?
A: The correct answer about IR stretching frequencies is given below
Q: Question 3: Identify the structure for each of the following cases: 1. Structure with the general…
A: Two questions based on IR spectroscopy that is to be accomplished.
Q: Question 1. a) Describe the effect of conjugated chromophores in a molecules UV-Vis absorption.
A: Conjugation is the system in which one single bond is combined to one double in a long chain. Or…
Q: The measured optical rotation of polyglutamic acid as a function of wavelength (2) can be fitted by…
A:
Q: 5. Can you predict all chromophores from the following UV-Vis spectra 3 200 -Sixth hour (b) 2.5…
A: Chromophore refers to a sepcific group present in a compound that absorbs light and is responsible…
Q: Answer the question below directly. Identify the two geometric isomers of stilbene C6H5CH=CHC6H5…
A: UV spectroscopy is branch of chemistry in which we deal with Woodward Fischer rule. From this rule,…
Q: (3) C2H;Cl3 1 3 PPM 2 1 2. 4, 6,
A: The NMR spectrum provides the following details about the structure of the molecules: The number of…
Q: need chemoselectivity. Under this 1. The relative reactivity of two different functional groups,…
A:
Q: 5. Which of the following materials (B- E) could be used to carry out the optical resolution of…
A: Whenever a particular enantiomer is needed in pure form, an optically pure chiral molecule can be…
Q: Ultraviolet and visible molecular absorption spectroscopy is used primarily for quantitative…
A: There are two statements given Ultraviolet and visible molecular absorption spectroscopy is used…
Q: What type of compound corresponds to the IR spectrum shown here? MICRONS 25 2.62.72.82.9 3 100 3.5…
A: IR spectrum is used to study and identify chemical substances or functional groups present in them…
Q: Unknown #: 4 s,9H d, 3H 9, 1H s,1H 11 10 8 ppm 180 160 120 100 80 20 200 140 60 40 ppm nasicITIKEIU
A:
Q: 4. The IR and the 'H NMR spectra of an unknown with a molecular formula of C₁1H14O3 are shown below.…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- A colorless solid that melts at 103 to 105 C was subjected to IR Spectroscopy to determine the functional groups present and partially determine its nomenclature based on the molecular formula given. What are the functional groups present based on the major infrared absorptions? (Select Possible Answers)Mesityl oxide( (CH3)2C=CHCOCH3) shows two bands at 230 nm (12600) and 327 nm (98). But its isomer, ( CH2=C(CH)3-CH2COCH3) does not show any bands above 210 nm. Explain.Acrolein shows two bands at 210nm (11,500) and 315 nm(15).assign these bands.
- This is a ATR spectrum. In which caffeine/PVA films were measured. Line a) correspond to a pure pva film and the other lines are films at increasing caffeine concentration. Notice that the OH band at around 3200cm-1 decreases at higher caffeine concentrations. However at a certain concentration (line d) such band starts to increase back to the initial intensity. What does that mean? Waht is the chemical meaning for that behavior?Would a component with a larger Rf value have a higher or lower affinity for the mobile phase compared to a component with a smaller Rf value? Explain, using specific color spots from your data.A compound Y (150.0 g/mol) in a 1.00 cm quartz cell has an extinction coefficient (ε) of 23,150 at λmax=235 nm. What is the energy absorbed by the compound at the maximum of the peak? Note that hc=1.98644586×10−25J*m. Report your answer in KJ/mole to the correct number of significant figures. Do not include the units in your answer. Please write out steps, will rate. Thank you.
- 2. What are some of the factors that can make interpreting IR spectra more difficult and that should be considered to avoid band misinterpretation?Compound X was dissolved in a 5-mL volumetric flask. A 1.00-mL aliquot of the compound was placed in a 10-mL volumetric flask and diluted to the mark. The solution was read in a UV-Vis spectrophotometer and an absorbance of 0.427 in a 1.000-cm cuvette was observed at 340 nm. Solve the following question. Data for Compound X:Molecular mass = 292.16 g/molMolar absorptivity (ɛ) at 340 nm= 6130 M -1 cm -1 a. Calculate the concentration of compound X in the cuvette. b. How many milligrams of compound X were used to make the 5-mL solution?Compound X was dissolved in a 5-mL volumetric flask. A 1.00-mL aliquot of the compound was placed in a 10-mL volumetric flask and diluted to the mark. The solution was read in a UV-Vis spectrophotometer and an absorbance of 0.427 in a 1.000-cm cuvette was observed at 340 nm. Solve the following question. Data for Compound X:Molecular mass = 292.16 g/molMolar absorptivity (ɛ) at 340 nm= 6130 M -1 cm -1 1. What was the concentration of compound X in the 5-mL flask?
- Im having a hard time distinguishing peaks on an IR spectrum. Theyre often very crowded, and i dont know if broad peaks get combined with sharp peaks. On this IR spectrum, it seems there is a broad range, medium strength band extending from 4000 to (about) 2000 cm-1, with several sharp peaks on top of that range. Does this mean that there is a carboxylic acid O-H present as well as the other bonds determined by the sharp peaks?Silica gel is very polar stationary phase. Therefore, more polar molecule sticks to silica gel on the TLC plate and has higherRfvalue. True FalseTo resolve the Na D lines 591.0 and 591.5 nm, what resolution is required if this is first order?

