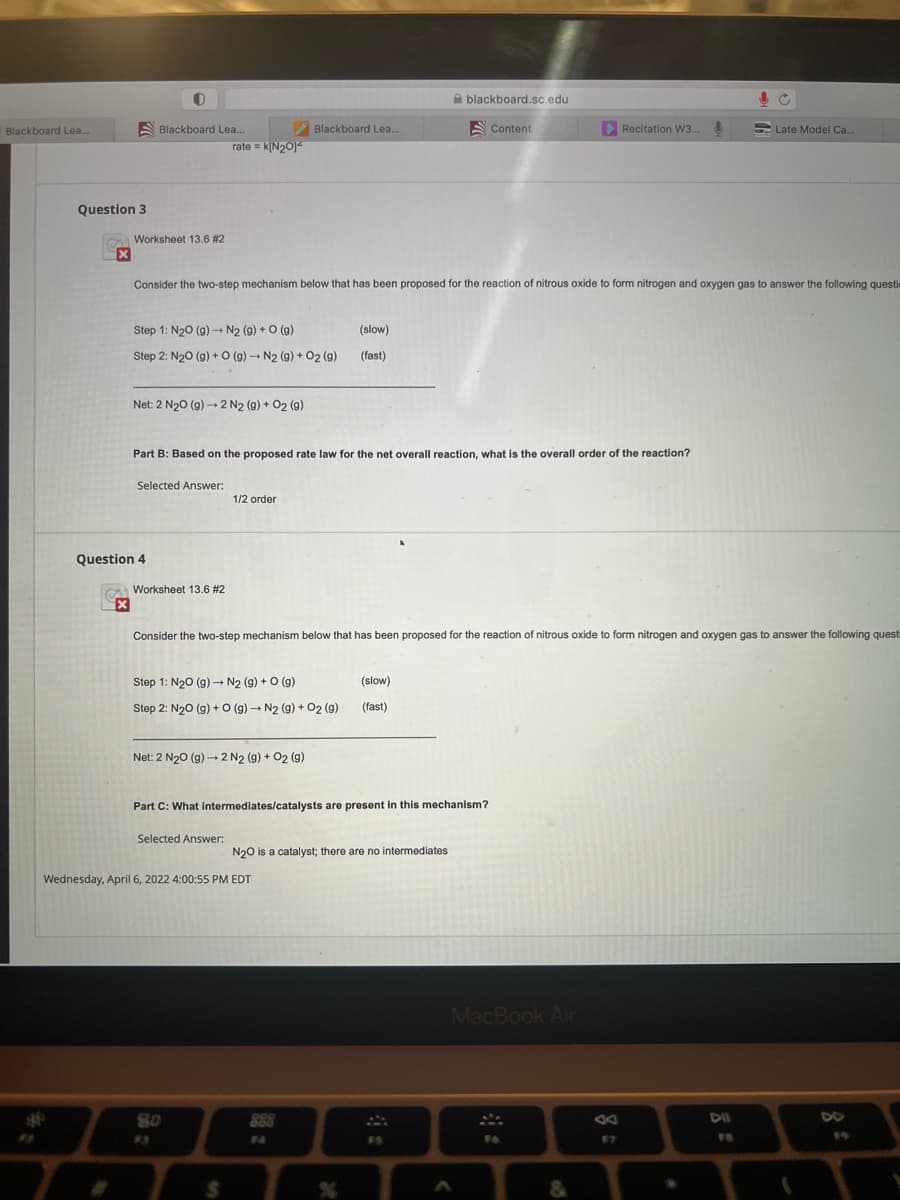
Question 3 Worksheet 13.6 #2 Consider the two-step mechanism below that has been proposed for the reaction of nitrous oxide to form nitrogen and oxygen gas to answer the following questi Step 1: N₂0 (9)→ N2 (9) + 0 (g) (slow) (fast) Step 2: N₂0 (g) + O (g) → N₂ (g) + O2(g) Net: 2 N₂0 (g) → 2 N₂ (9) + 02 (9) Part B: Based on the proposed rate law for the net overall reaction, what is the overall order of the reaction? Selected Answer: 1/2 order Worksheet 13.6 #2 X Consider the two-step mechanism below that has been proposed for the reaction of nitrous oxide to form nitrogen and oxygen gas to answer the following quest Step 1: N₂0 (g) → N₂ (g) + O(g) (slow) (fast) Step 2: N₂0 (g) + O (g) → N₂ (9) + 02 (9) Net: 2 N₂0 (g) → 2 N2 (9) + 02 (9) Part C: What intermediates/catalysts are present in this mechanism? Selected Answer: N₂O is a catalyst; there are no intermediates nesday, April 6, 2022 4:00:55 PM EDT Question 4
Question 3 Worksheet 13.6 #2 Consider the two-step mechanism below that has been proposed for the reaction of nitrous oxide to form nitrogen and oxygen gas to answer the following questi Step 1: N₂0 (9)→ N2 (9) + 0 (g) (slow) (fast) Step 2: N₂0 (g) + O (g) → N₂ (g) + O2(g) Net: 2 N₂0 (g) → 2 N₂ (9) + 02 (9) Part B: Based on the proposed rate law for the net overall reaction, what is the overall order of the reaction? Selected Answer: 1/2 order Worksheet 13.6 #2 X Consider the two-step mechanism below that has been proposed for the reaction of nitrous oxide to form nitrogen and oxygen gas to answer the following quest Step 1: N₂0 (g) → N₂ (g) + O(g) (slow) (fast) Step 2: N₂0 (g) + O (g) → N₂ (9) + 02 (9) Net: 2 N₂0 (g) → 2 N2 (9) + 02 (9) Part C: What intermediates/catalysts are present in this mechanism? Selected Answer: N₂O is a catalyst; there are no intermediates nesday, April 6, 2022 4:00:55 PM EDT Question 4
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 11.65PAE
Related questions
Question
Transcribed Image Text:C
blackboard.sc.edu
Content
Blackboard Lea...
> Recitation W3... J
Late Model Ca...
rate = k[N₂01²
Question 3
Worksheet 13.6 #2
X
Consider the two-step mechanism below that has been proposed for the reaction of nitrous oxide to form nitrogen and oxygen gas to answer the following questi
Step 1: N₂0 (g) → N2 (9) + O (g)
(slow)
(fast)
Step 2: N₂O(g) + O (g) → N2 (g) + O2 (9)
Net: 2 N₂O (g) → 2 N2 (9) + O2 (9)
Part B: Based on the proposed rate law for the net overall reaction, what is the overall order of the reaction?
Selected Answer:
1/2 order
Worksheet 13.6 #2
Consider the two-step mechanism below that has been proposed for the reaction of nitrous oxide to form nitrogen and oxygen gas to answer the following questi
Step 1: N₂O (g) → N₂ (g) + O (9)
(slow)
(fast)
Step 2: N₂O(g) + O (g) → N₂ (9) + 02 (9)
Net: 2 N₂0 (g) → 2 N2 (9) + 02 (9)
Part C: What intermediates/catalysts are present in this mechanism?
Selected Answer:
N₂O is a catalyst; there are no intermediates
Wednesday, April 6, 2022 4:00:55 PM EDT
80
Blackboard Lea...
Question 4
Blackboard Lea...
999
F4
*
<
MacBook Air
F6
&
8
17
DII
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning