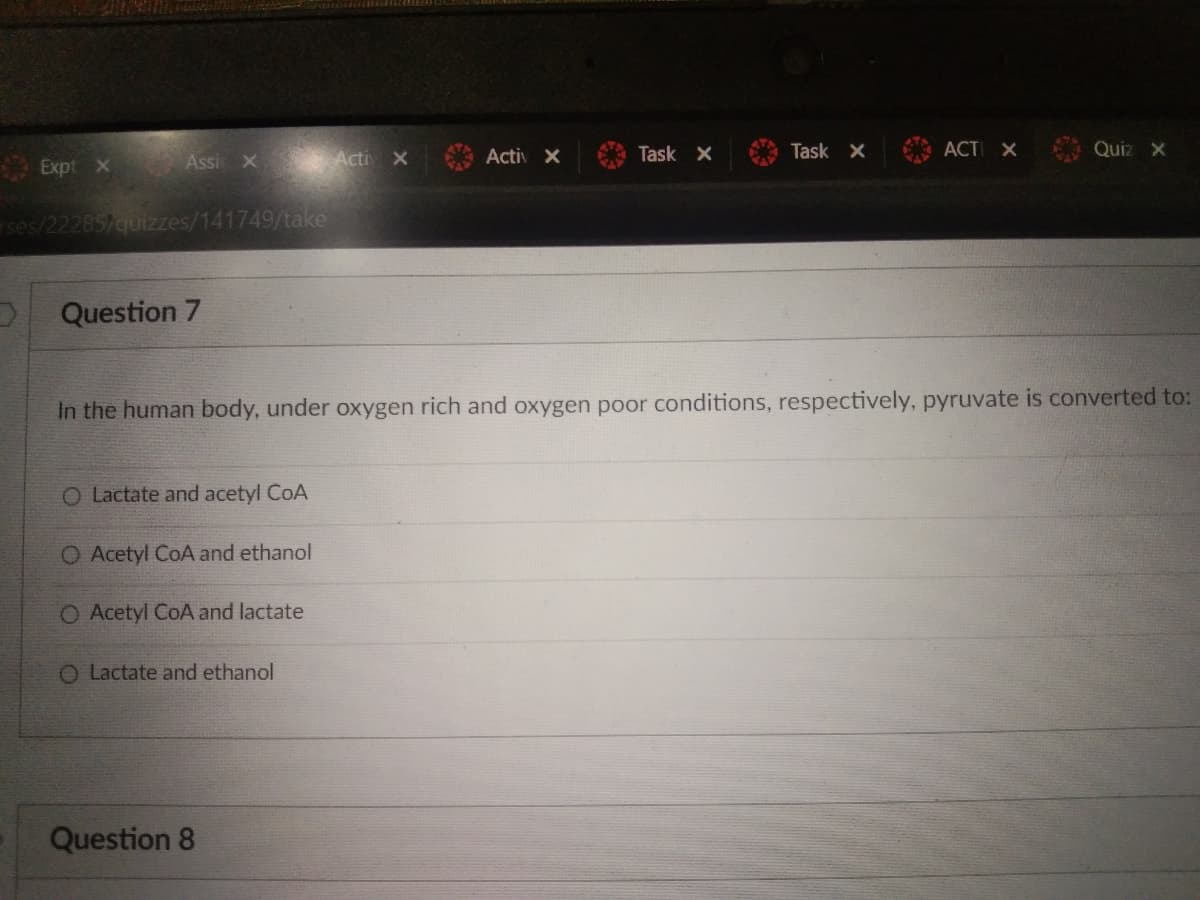
Question 7 In the human body, under oxygen rich and oxygen poor conditions, respectively, pyruvate is converted to: O Lactate and acetyl CoA O Acetyl CoA and ethanol O Acetyl CoA and lactate O Lactate and ethanol Questic
Question 7 In the human body, under oxygen rich and oxygen poor conditions, respectively, pyruvate is converted to: O Lactate and acetyl CoA O Acetyl CoA and ethanol O Acetyl CoA and lactate O Lactate and ethanol Questic
Anatomy & Physiology
1st Edition
ISBN:9781938168130
Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Chapter24: Metabolism And Nutrition
Section: Chapter Questions
Problem 15RQ: Acetyl CoA can be converted to all of the following except ________. ketone bodies fatty acids...
Related questions
Question
Transcribed Image Text:Expt x
Assic X
ses/22285/quizzes/141749/take
Question 7
O Lactate and acetyl CoA
O Acetyl CoA and ethanol
O Acetyl CoA and lactate
O Lactate and ethanol
Activ X
Question 8
Activ X
Task X
In the human body, under oxygen rich and oxygen poor conditions, respectively, pyruvate is converted to:
Task X
ACTI X
Quiz X
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning