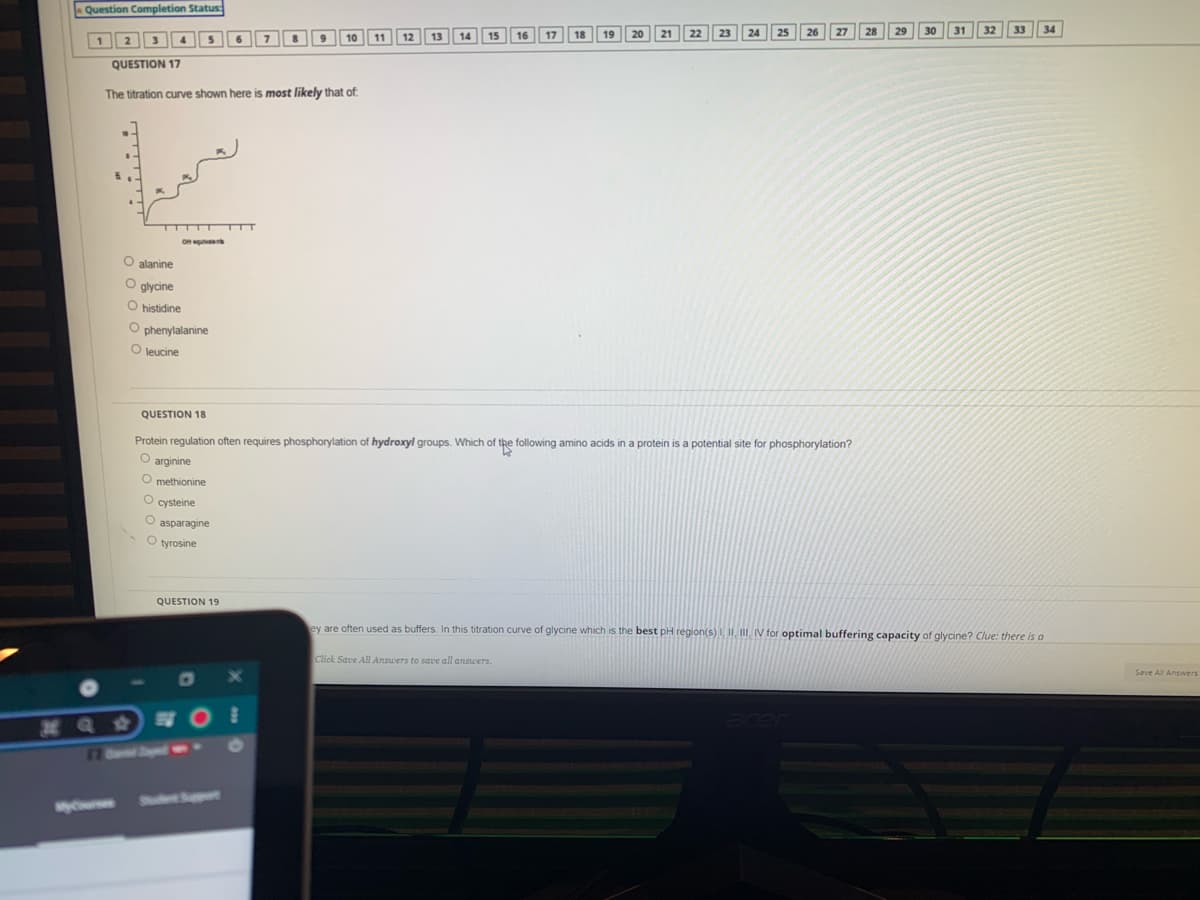
Question Completion Status 30 31 32 33 34 12 13 14 16 17 1 2 10 7 11 QUESTION 17 The titration curve shown here is most likely that of Oquiv O alanine O glycine O histidine O phenylalanine leucine
Q: The base that is circled in red could be adenine thymine guanine None of the provided answer choices…
A: The molecular basis of heredity is DNA. It can be thought of as a genetic information reserve bank.…
Q: Be sure to answer all parts. Give the amino acid sequence of an octapeptide that contains the amino…
A: Amino acids are the simplest unit for the formation of peptides or proteins. They are joined by a…
Q: Question 11 Match the different carbohydrates' nomenclaturel glycan representation with their…
A: There are 4 biomacromolecules; Proteins, Nucleic acids, lipids and carbohydrates. The polymer…
Q: Question 4 a-D-galactose from B-D-glucose can be differentiated using which method of analysis? O…
A: Alpha - D - Glucose and Beta - D - Glucose are the two cyclic hemiacetal forms of D - Glucose, which…
Q: QUESTION 1 Regarding the following peptide sequence, which of the following statements is FALSE?…
A: *Note: Since you have asked multiple question, we will solve the first question for you. If you want…
Q: Modified TRUE or FALSE. Write the word TRUE if the statement is correct. If the statement is false,…
A: Proteins are made up of amino acids, which are a type of molecule. The basic components of life are…
Q: Question 4. You must cleave the following peptide into smaller fragments. How many peptides would…
A: Proteins are complex molecules formed by the polymerization of amino acid residues. Amino acids are…
Q: 3. Minimum number of carbon required for a ketose sugar to have cyclic structure is - Explain in 2-3…
A: Carbohydrates are composed of carbon, oxygen, and hydrogen which are connected by the…
Q: Base pairs Data – all had Temp = 250C PH = 7 Binding Constant A & X [A] = 0.00373221M [X] =…
A: The DNA double helix is made of two linked strands that are wound in a helical shape. The two…
Q: chymotrypsin
A: Chymotrpsin: It is a digestive enzyme which breaks down polypeptides. It consists of a catalytic…
Q: 2. Glycosidic bond is sucrose is this answer. Explain in 2-3 sentences why you chose
A: Sucrose can be defined as a type of dissacharide. It comprises one unit of glucose and fructose…
Q: Question 15 gal(a1-6)gal(a1-6)glc(a1-2B)fru. Which of the complete IUPAC of this tetrasaccharide?…
A: This is the Tetrasaccharide structure of carbohydrates.
Q: Modified true or false. Write the correct answer if the underlined word is false. 1. In…
A: Chromatography is the process of separating a mixture of chemicals into its constituent parts based…
Q: - An aldo hexose will have stereoisomers. Explain in 2-3 sentences why you chose this answer. A. 8 B…
A: Six carbon atoms make up hexose, a monosaccharide (simple sugar). Hexoses play a critical role in…
Q: Base pairing means a purine bonds to a pyrimidine true False
A: Answer- There are two different types of base pairs purine and pyrimidine that have differences in…
Q: Question 13 Which of the following can determine location of glysosidic linkages? O Polarimetric…
A: A glycosidic linkage or bond is a type of covalent bond that joins the carbohydrate or sugar…
Q: QUESTION 9 Macrolides: O Bind to the 50S subunit O Contain a lactone ring O Include azithromycin (Z…
A: Hi! Thank you for the question. As you have posted multiple questions, I will be answering the first…
Q: Using the following coding DNA sequence:TAC GCA CCT TTA GAT CAT AAC TTT GGA ACT1. Write the…
A: Deoxyribose nucleic acid (DNA) is the genetic material composed of four nucleotide bases, namely…
Q: Modified true or false. Write the correct answer if the underlined word is false. 1. The zinc ion…
A: A zinc finger is a tiny protein structural motif defined by the coordination of one or more zinc…
Q: Question #28 Polysaccharides are different from polypeptideș in which of these Ways? A)…
A: Carbohydrates are biomolecules made up of carbon, hydrogen, and oxygen that's why also known as…
Q: 2. Which of the following is not a type of specificity?
A: If we talk about specificity there is many feilds where the specificity is play there role and all…
Q: All amino acids are optically active (contain except chiral alpha carbon) a Possible Answers:…
A: Amino acids are the units of protein which contains a central chiral carbon. Chiral carbon is the…
Q: GTP GDP, CO2 the task is to DRAW a reasonable mechanism for reaction listed. Not all products or…
A: Conversion of oxaloacetate to phosphoenolpyruvate is the major step in gluconeogenesis.…
Q: In 3-5 sentences, discuss the similarity and the difference between chitin and cellulose.
A: Chitin and cellulose are homopolysaccharides. Polysaccharide is of two types homo and…
Q: 3-phosphoglycerate serves as the precursor of the glycerol backbone during TAG biosynthesis. True…
A: All eukaryotic organisms and even a few prokaryotes are able to synthesize triacylglycerol. The…
Q: DISCUSSION - lodine (I) and its role in human body 5 5 Now try to discuss the role of iodine (I) in…
A: Iodine is a naturally occuring minerals present on the Earth. Humans get the iodine through the food…
Q: Modified true or false. Write the correct answer if the underlined word is false. 1. A prosthetic…
A: Proteins' principal role is to act as enzymes—catalysts that help cells speed up nearly all chemical…
Q: 1.) What are the main structural features of the mucupolysaccharides chitin? 2.) How do this aid in…
A: Chitin is a carbohydrates polymer that is composed of N-acetyl D-glucosamine units attached together…
Q: Draw 3 isomers of C7H12O2 then state where you wil disposal should only be repeated once).
A: Heptanedial is represented by C7H12O2. The molar mass of the given compound is found to 128.2 g/mol.…
Q: (In 10 sentences) Can disaccharide be completely assimilated and used by human cells as a direct…
A: All disaccharides are dimeric product of monosaccharide linked together with glycosidic bond.…
Q: Question: Glutamic acid is the parent compound of monosodium glutamate (known as MSG), which is used…
A: Glutamic acid is a polar acidic amino acid. In addition to the amino and carboxyl groups attached to…
Q: Question 1 Example of a Protein Purification Scheme: Purification of the Enzyme Xanthine…
A:
Q: 1.) What are the main structural features of the polysaccharides starch? 2.) How do this aids in its…
A: Polysaccharides are major classes of biomolecules. Polysaccharides are long chain of carbohydrate…
Q: 5. The minimum number of carbon required for a monosaccharide. Explain in 2-3 sentences why you…
A: Carbohydrates are divided into 3 classes monosaccharide, disaccharide, and polysaccharide.…
Q: BONUS QUESTION! Protein 1 has a pl value of 7.2. Protein 2 has a pl value of 5.6. Which protein is…
A: The isoelectric point (pI) is equal to the pH at which the protein exists in a double-ionic state…
Q: Intrinsically disordered proteins may:
A: An intrinsically disordered protein is a type of protein that primarily lacks a fixed or ordered…
Q: Snake venom contains phospholipases which cause hemolysis of the red blood cells of the victims by a…
A: Hi! Thank you for the questions. As you have posted multiple questions, I will be answering the…
Q: Question 23 Explain the structure of your protein. (Up to 100 words) For the toolbar, press ALT+F10…
A: When asked to explain the structure of a protein, we must describe the primary, secondary and…
Q: IV. PROTEIN QUESTIONS: 1. Classify each of the following amino acids as nonpolar, polar uncharged,…
A: Amino acids are the compounds that contain a carboxyl group, an amine group, a hydroxyl group, and…
Q: Question 6 Identify the three true statements related to pyrimidine synthesis: The initial steps of…
A:
Q: 1.) What are the main structural features of the polysaccharides starch? 2.) How do this aid in its…
A: Starch is a homopolysaccharide. It is the carbohydrate reserve of the plants. It is an important…
Q: ) Amino acid sequence of a heptapeptide gave the following information Asp, leu, lys, ,met, phe and…
A: Introduction Amino acids are the building blocks of proteins. The building blocks of life are amino…
Q: Be sure to answer all parts. Give the amino acid sequence of an octapeptide that contains the amino…
A: Proteins are composed of amino acids. They are linked together by peptide linkages. Proteins have…
Q: 10. Procaine is known to undergo faster metabolic hydrolysis than procainamide. Give explanation.…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 1. Maltose is a disaccharide of, Explain in 2-3 sentences why you chose this answer. A. glucose and…
A: Disaccharides are formed when two monosaccharides are joined together and a molecule of water is…
Q: What are the main structural features of the polysaccharides cellulose? How do this aid in its…
A: Cellulose - Polysaccharide made up of the monomer glucose
Q: Question Explain briefly why isn't the Alfa Carbon in ?Glycine considered Chiral
A: There are 20 naturally occuring amino acids. All amino acids contain at least one chiral center (the…
Q: Answer the following multiple-choice questions and EXPLAIN in 3-5 sentences why you chose that…
A: Enzyme is basically biocatalyst that increase the rate of chemical reaction without itself being…
Q: QUESTION 16 Protein maturation in the ER includes. OA Disuifide bend formation Li proteolytic…
A: Protein processing and modification occur in endoplasmic reticulum which has two sub units. Rough…
Q: the correct answers since there are no more keys to correction. MULTIPLE CHOICE: WRITE the letter of…
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since…
Step by step
Solved in 2 steps

- QUESTION NO. 1Targeting a protein to be degraded within proteasomes usually requires ubiquitin. In the function of ubiquitin all of the following are true except: A. ATP is required for activation of ubiquicin. B. a peptide bond forms between the carboxyl terminal of ubiquitin and an ε-amino group of a lysine . C. linkage of a protein to ubiquitin does not always mark it for degradation. D. the N-terminal amino acid is one determinant of selection for degradation. E. ATP is required by the enzyme that transfers the ubiquitin to the protein to be degraded QUESTION NO. 2Much of procollagen formation occurs in the endoplasmic reticulum and Golgi apparatus which requires signal peptide. All of the following statements about targeting a protein for the ER are true except. A. signal peptide usually has a positively charged N-terminus and a stretch of hydrophobic amino acids. B. signal peptide emerging from a free ribosome binds signal recognition…Question: A decapeptide composed of ser, ala, IIe. his, trp, phe was treated with 1-flouro- 2,4-dinitrobenzene. It gave a DNP-his on the N terminal and free trp when treated with carboxypeptidase. Upon partial hydrolysis of the peptide, the following fragments were obtained. a. his-lle-phe-ala c. his-ala-phe e. ser-lle-his b. ala-phe-trp d. phe-ala-ser Give the amino acid sequence of the above decapeptide.student question 1. How do both Hemoglobin and S-adenosylmethionine synthetase form hydrophobic pockets? explain in detail. 2. how does the structure of S-adenosylmethionine synthetase make it resistant to heat denaturation and why, explain in detail
- 510. Trypsin cleaves proteins after lysine and arginine residues. Trypsin itself has 13 lysine and 2 arginine residues in its primary structure. Why does trypsin not cleave itself into 16 smaller peptides?Question 1 options: The specificity pocket of the serine protease chymotrypsin, which interacts with Tyr and Phe-containing peptide sequences, contains a Ser residue. A research group is trying to modify chymotrypsin such that it has a low KM with Trp-containing peptides. Enter the name or abbreviation of an amino acid that the Ser could be mutated to that would likely have the desired effect. (Hint: look at the diagrams of the specificity pockets shown in the course slides, and consider how the Ser would need to change to account for the difference between Tyr/Phe and Trp.)Question:- Based on the figure below, predict what peptide bond could be the substrate of each protease(The bond marked in blue is where hydrolysis occurs, choose 2 peptides per protease type) Chymotrypsin:_________ Trypsin:_________ Elastase:_________ 1. SR−SG 2. SF−SG 3. SK−SG 4. SA−SG 5. SV−SG 6. SM−SG
- Time remaining: 00:08:55 Chemistry Consider a peptide with the following amino acid sequence: H2N-ASENHLDGCPYTKSRG-COOH Analyze the predominate protonation state and thus dominate charge (+1, 0, -1) of the residues in this peptide at pH 3, 6, and 10 by filling in the table below, identifying the relevant pKa appropriate for each residue. (Note that choice of pKa and its. application will be graded independently; e.g., if you can choose the wrong pKa but apply it correctly, you will lose points only for the pKa.) AA Residue pKa Charge pH 3 Charge pH 6 Charge pH 10 A S E N H L D G C P Y T K S R GCan Please Help Me: Bio- Chemistry 60 minutes only the given time. Wish you could help me. I will give UPVOTE and GOOD FEEDBACK. QUESTION/// Diisopropyl fluorophosphate (DFP) inhibit acetylcholinesterase activity by reacting covalently to a serine residue found within the active site of the enzyme. The physiological effect of this inactivation is interference with neurotransmitter inactivation at the synapses of nerves, resulting in the constant propagation of nerve impulses, which can lead to death. What type of inhibitor is DFP? Explain. ///QUESTIONQuestion 11. // Hint: Isoelectric focusing separates proteins based on their pI values, and can separate proteins that only differ by a net charge of ±1.±1. Recall that an amino acid residue with a negatively charged R group has a relatively low isoelectric point (pI) where it has zero net charge. Likewise, an amino acid residue with a positively charged R group has a relatively high isoelectric point (pI) where it has zero net charge. Order from Low pH to High pH
- Question for protein crystallography- 1. While performing a routine protein crystallization screening, you observe that one of your well drops has doubled in size compared to the remaining wells. Note: the drop in question was set up by taking 1 µL of the protein solution (10 mg/mL LDH in 20 mM TRIS pH 7.5, 0.5 M NaCl and 10% PEG 4000K) and adding it to 1 µL of the crystallization solution (10% PEG 4000K, 50 mM NaCl and 20 mM TRIS pH 8.0). Why did this drop grow larger in size compared to when you initially set it up?Enzyme: Crystal Structure of Wild-Type Human Phosphoglucomutase-1 (PGM1) the description of the mechanism of how this enzyme is regulated (e.g., depending on the enzyme, the mechanism could range from being solely dependent on gene regulation to protein structure-based mechanism).Situational task: As a result of intoxication, enzymes that provide splicing are not synthesized in liver cells. What is the reason for stopping protein biosynthesis in this case? Justify the answer



