Question6 Consider the phase diagram given below. What phases are present at points A through H? Identify the triple point, normal boiling point, normal freezing point, and critical point. Which phase is denser, solid or liquid? B. 1.0 atm (ignore point H).
Question6 Consider the phase diagram given below. What phases are present at points A through H? Identify the triple point, normal boiling point, normal freezing point, and critical point. Which phase is denser, solid or liquid? B. 1.0 atm (ignore point H).
Biology 2e
2nd Edition
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:Matthew Douglas, Jung Choi, Mary Ann Clark
Chapter33: The Animal Body: Basic Form And Function
Section: Chapter Questions
Problem 40CTQ: On a molecular level, how can endotherms produce their own heat by adjusting processes associated...
Related questions
Question
6.
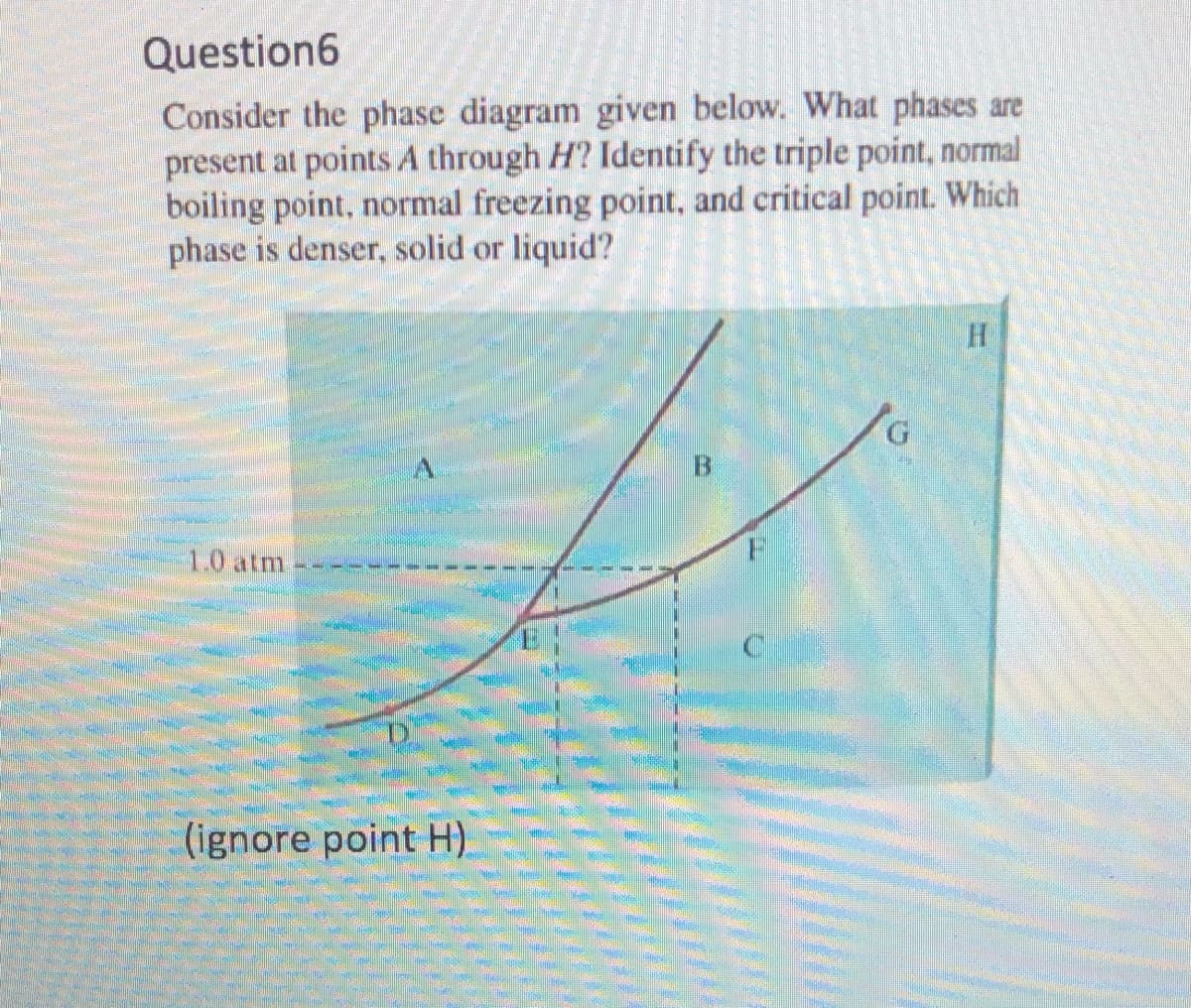
Transcribed Image Text:Question6
Consider the phase diagram given below. What phases are
present at points A through H? Identify the triple point, normal
boiling point, normal freezing point, and critical point. Which
phase is denser, solid or liquid?
G.
1.0 atm
(ignore point H)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax



Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax


