14.9 g ice cube is placed into 332 g of water. Part A Calculate the temperature change in the water upon complete melting of the ice. Hint: Determine how much heat is absorbed by the melting ice and then use q= MCAT to calculate the temperature change of the 332 g of water. AT = PC Submit Previous Answers Request Answer
14.9 g ice cube is placed into 332 g of water. Part A Calculate the temperature change in the water upon complete melting of the ice. Hint: Determine how much heat is absorbed by the melting ice and then use q= MCAT to calculate the temperature change of the 332 g of water. AT = PC Submit Previous Answers Request Answer
Nutrition Through The Life Cycle
7th Edition
ISBN:9781337919333
Author:Brown, Judith E.
Publisher:Brown, Judith E.
Chapter4: Nutrition During Pregnancy
Section: Chapter Questions
Problem 23RQ
Related questions
Question
Transcribed Image Text:I Review Constants Periodic Table
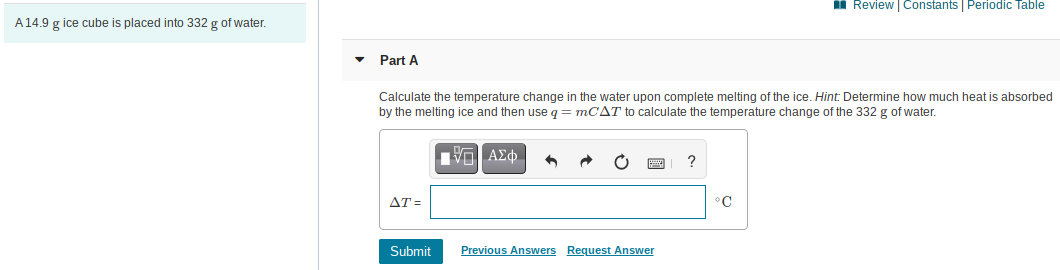
A14.9 g ice cube is placed into 332 g of water.
Part A
Calculate the temperature change in the water upon complete melting of the ice. Hint: Determine how much heat is absorbed
by the melting ice and then use q = MCAT to calculate the temperature change of the 332 g of water.
ο ΑΣφ.
AT =
°C
Submit
Previous Answers Request Answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Nutrition Through The Life Cycle
Health & Nutrition
ISBN:
9781337919333
Author:
Brown, Judith E.
Publisher:
Cengage Learning,



Nutrition Through The Life Cycle
Health & Nutrition
ISBN:
9781337919333
Author:
Brown, Judith E.
Publisher:
Cengage Learning,



Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781305073951
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning


Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781337408332
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning