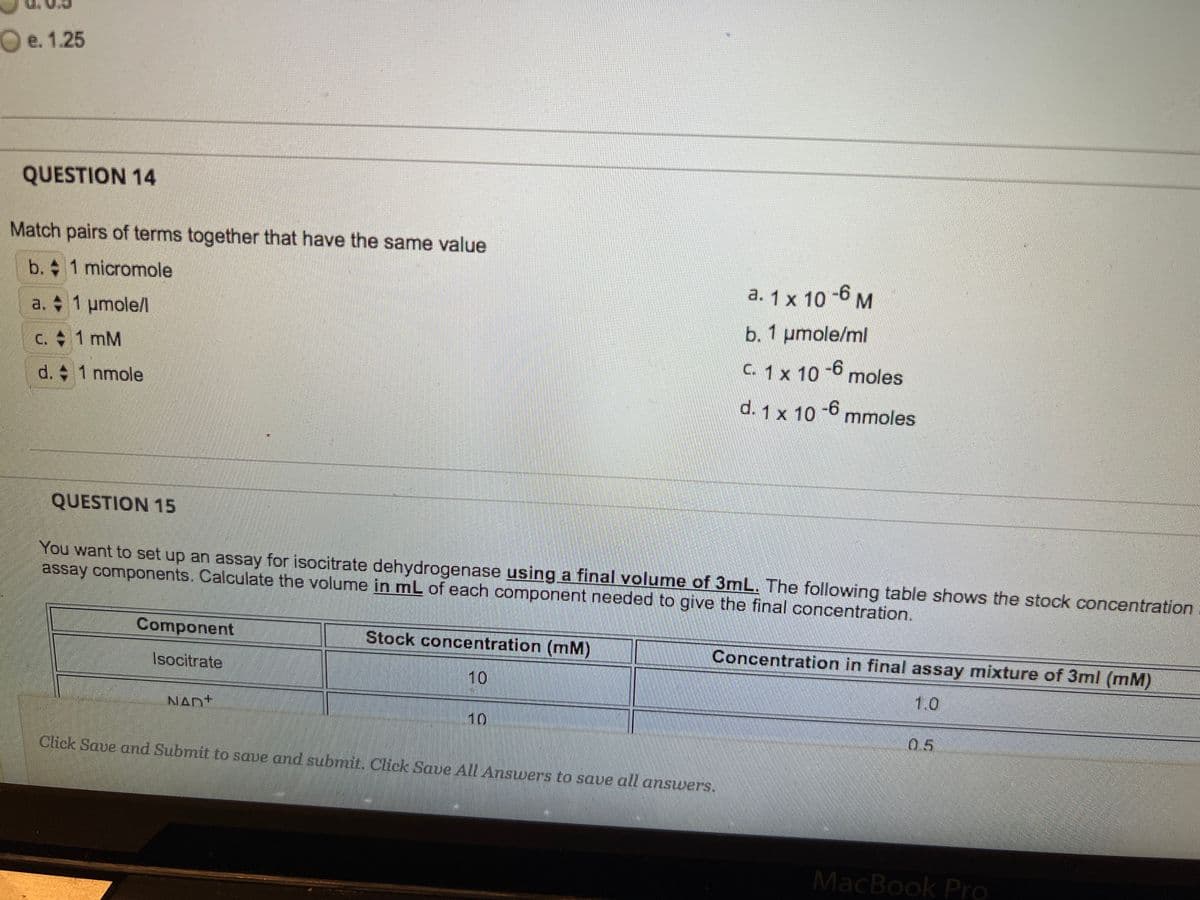
QUESTION 14 Match pairs of terms together that have the same value b. 1 micromole 1 μmole/l 1 mM 1 nmole a. c. d. a. 1 x 10-6 M b. 1 µmole/ml C. 1 x 10-6 moles -6 mmoles d. 1 x 10
Q: From the various reactions you have learned in catabolism of amino acids; list three reactions in…
A: Catabolism of proteins releases amino acids which composed of mainly two parts-amino nitrogen (in…
Q: For the amino acids listed below, what tertiary/quaternary interaction can each of the amino acids…
A: Tertiary structure is the three dimensional conformation of a protein. Proteins with more than one…
Q: The apical surfaces of hair cells are exposed to an unusual extracellular fluid called endolymph.…
A: Endolymph is an extracellular fluid present in the inner ear. It is also known as Scarpa fluid. This…
Q: Which of the following is true of the TCA cycle? It is independent of aerobic conditions. It is the…
A: Catabolism is the process by which complex molecules (consumed in the form of food) are broken down…
Q: How to calculate the Km based on this data?
A: hghfjThe tricarboxylic acid (TCA) cycle, also known as the Krebs or citric acid cycle, is the main…
Q: Integral membrane proteins can exist as all of the following, except: a. A single helix A beta…
A: An integral or intrinsic membrane protein abbreviated as IMP is one type of membrane protein that is…
Q: Table 1, Effect of enzyme concentration Amount of lodine Test Results (color) Amylase Enzyme 0 drops…
A: There are various tests used to detect the presence of polysaccharides including disaccharides and…
Q: In a mammalian liver, glucose 1-phosphate can go in three different direction; glycolysis, replenish…
A: The end product of glycogenolysis (i.e. break down of glycogen) is Glucose 1-phosphate. The enzyme…
Q: QUESTION 8 You need to make 500mL of a stock solution of sodium chloride at a final concentration of…
A: Given Values: The volume of the sodium chloride stock solution = 500 ml=0.5 Litre Concentration of…
Q: Riboflavin gives rise to cofactors: Select one: O a. All of them O b. FAD; FADH2 O c. NAD+; NADH d.…
A: Introduction: Riboflavin is likewise known as Vitamin B2. It is an important food supplement.…
Q: Which is more hydrophilic, cholesterol or phospholipids? Defend your answer
A: Organic substances known as lipids are not water soluble. They are non-polar and are soluble in…
Q: Explain the structure of Human muscle fructose 1,6-biphosphate aldolas complexed with fructose…
A: Introduction: Fructose 1,6 bisphosphate aldolase is a ubiquitous cytosolic enzyme involved in the…
Q: Which of the following is incorrect about the transition state analogs? O a. They are synthetic…
A: Transition state analogs or analogues, are chemical compounds with a structure resembling the…
Q: Using appropriate diseases as examples, describe three molecular techniques that can be used in…
A: Three molecular techniques involved in disease diagnosis are: 1. Various amplification techniques…
Q: The following carbohydrates are disaccharide, except:* A. Maltose B. Galactose C. Lactose D.…
A: Carbohydrates are the primary building blocks of all living organisms. Carbohydrates are all made up…
Q: Identify A, B and C. Show how the conversion from A to B takes place. HN A: Name? B: Enzyme? C:…
A: Vitamins and cofactors undergo several different small chemical changes to become different…
Q: CH₂-0-0 _-(CH₂)-CH₂ Triglycerides can be classified by the number of double bonds present in the…
A: A triglyceride has a glycerol backbone in which all the hydroxyl groups are esterified to fatty…
Q: 3. If we alter the kinetics of a unimolecular reaction by increasing Km but leaving Vmax alone, how…
A: Km reflects the enzyme’s dissociation constant so a high Km means the enzyme is likely to dissociate…
Q: ve me solution Using the half-reaction technique, write the molar stoichiometric equation for…
A: vbbbbbbbbbbbbbbbbbbbbb
Q: Which of the following is incorrect? a. The ratio "catalyzed rate/uncatalyzed rate" is known as…
A: The Enzymes as biological catalysts. In the enzyme catalyzed reaction, the substrate is converted to…
Q: Which of the following are considered predominantly catabolic metabolic pathways? (Choose all…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: What are the components of Blood?
A: Blood is a fluid tissue that is major component of circulatory system. It supplies the essential…
Q: of the following is incorrect about the sphingolipids? a. The backbone is sphingosine which has an…
A: Sphingolipids are a type of lipid present in cell membranes containing a backbone of sphingoid…
Q: An enzyme has 10 times greater affinity for substrate "A" than for substrate "B". Which of the…
A: Enzymes are bio-catalyst that participate in biochemical process and they are highly specific in…
Q: A compound was suspected of being a negative allosteric regulator of enzyme A. Which of the…
A: Allosteric regulation is the regulation of an enzyme in the body by binding an effector molecule at…
Q: What mutation can be prevented when we exercise?
A: As our body grows, damages, and repairs with its progression so does our genetic material. The DNA…
Q: Which of the following is incorrect about the peptide Val-Leu-Ala-Ser-Gly? It has a net charge of…
A: H3+N-Val-Leu-Ala-Ser-Gly-COO- a) We have 1 positive charge at the N-terminal and one negative charge…
Q: 1/V, min/umol 5.50 5.00 4.50 4.00 3.50 answer. 3.00 2.50 2.00 0.00 y = 0.9474x + 2.6649 0.50 1.00…
A: Double reciprocal plot is also called as lb plot that is drawn by taking inverse of Substrate…
Q: Which of the following is incorrect about lipids? a. Some contain polar groups referred to as "polar…
A: Lipids are biomolecules that are made up of fatty acids esterified to Glycerol backbone. Lipids have…
Q: Under what conditions will lactic acid accumulate in skeletal muscle? A. when citric acid enzymes…
A: Lactic acid is the end product of anaerobic Glycolysis where Glucose is oxidised to lactic acid.
Q: Question Completion Status: A 2.0kb bacterial plasmid 'BS1030' is digested with the restriction…
A: Restriction enzymes cleave the DNA at palindromic sequences. Plasmids are circular DNA molecules.…
Q: Which of the following is incorrect about an omega-3 fatty acid whose hydrocarbon chain has only one…
A: Omega-3 fatty acids are defined as polyunsaturated fatty acids (PUFA) which have a double bond…
Q: discuss the significance of the different aseptic techniques utilized in maintaining sterility of…
A: The laboratory that carries out growth of prokaryotic cells or eukaryotic cells in physiological…
Q: Question Completion Status: 2,0 1,6- 1,2 0,8 0,4 2000- CH₂ 31 CH-COO 260 300 340 400 nm If you…
A: Isocitrate dehydrogenase is an enzyme involved in the TCA cycle. The TCA cycle occurs in the…
Q: Which of the following depicts the mild alkalosis? Select one: a. pH=7.33 Ob. pH=7.45 O c. H=7.65…
A: The collecting duct of kidney participates in reabsorption and secretion of selective ions into…
Q: 2 When an oxygen molecule binds to the deoxyhemoglobin, multiple conformational changes happen that…
A: Hemoglobin is a protein that carries oxygen from lungs to tissues, and CO2 from tissues to lungs.…
Q: Which of the following amino acid residues cannot play a role in acid-base catalysis? a. Aspartate…
A: Acid-Base Catalysis is reaction process involving the movement of a proton from one molecule to…
Q: Conjugated proteins which are a combination of amino acids and carbohydrates O A. nucleoproteins OB.…
A: Based on the composition, proteins are of two types, simple and conjugated proteins. The simple…
Q: In glycolysis, pyruvate kinase produces what high energy product and what low energy product?
A: Glycolysis is a pathway in which the Glucose (C6) is broken down into a 3 carbon product i.e. 2…
Q: Consider the following reaction. CH₂-CH-COO-CH₂-C-Coo- он b Which group of enzymes catalyzes this…
A: The six functional classes of enzymes are hydrolases, oxidoreductases, lyases, transferases, ligases…
Q: protein A on-rate += 25 °C binding Determinat and C э Kon = 1.6 x 106 M²ts² 8-1 free energy in…
A: Association constant tells us the rate at which to molecules can combine with each other. Whereas,…
Q: A child weighing 59 lb and 48 inches tall is receiving an anti-inflammatory agent at a regimen of 60…
A: Dosage represents the amount of drug a person takes or consumes. Dosage can be calculated from…
Q: A chemical reaction is more likely to occur spontaneously if A. the products of the reaction are…
A: For a reaction to occur spontaneously, the free energy change i.e delta G should be negative meaning…
Q: or the following amino acids: Isoleucine and Tyrosine a. Draw its complete protonic equilibria.…
A: Hi, thank you for the question. As per the honor code, we are allowed to answer three sub-parts at a…
Q: can be broken down by amylase.* A. glycogen B. amylopectin C. catalase D. amylase
A: Glycogen is a homopolysaccharide composed of glucose units that are linked through glycosidic…
Q: Which of the following is the characteristic that is common to all lipids?* A. They are made up of…
A: The simplest form of lipid is fatty acids which are a long chain of hydrocarbons. A biological…
Q: Hello! Can you please answer the four questions below directly? This is related to GLUTEN from WHEAT…
A: Answer for the following questions are :
Q: See attached, you have a table that is perhaps required for some of the questions. Answer question…
A: The food that we eat contains proteins which need to be broken down and absorbed for its…
Q: Regarding light spectroscopy, which of the following statements is correct? The spectral lines are…
A: Introduction: Spectroscopy is the study of light emission and absorption of light and other…
Q: How many net molecules of nucleoside triphosphate (ATP and equivalent molecules) are produced by…
A: As you have posted multiple unrelated questions we are supposed to answer only the first question…
Question
Step by step
Solved in 2 steps

- Serum blood of a patient with dislipoproteinemia type 1 has milky appearance even in fasting. If serum stays at low temperature (40) for several hours fatty layer appears on its surface. What are the possible causes of these symptoms? To explain this, answer the questions and do the following tasks: a) what compounds of serum must be tested for that patient in biochemical lab? b) write the reaction which does not occur properly in patient’s blood; c) write down the schemes, explaining how the products of the previous reaction are used in adipose tissue and heart in healthy person 2 hours after a meal.Briefly comment on the differences of using a fixed-time assay versus a kinetic assay to measure enzyme activity. Is it reasonable to assume that the reaction velocity obtained by measuring the amount of product after 30 minutes in a fixed-time assay is directly proportional to absorbance? How could you determine whether this was the case? Word limit 180 words including citation and referenceQ. You have now carried out the Somogyi Nelson determination of reducing sugars and measured the absorption spectra of Tube 9. What is the optimum wavelength you should use to monitor the Somogyi Nelson Assay why? Table 1._SOMOGYI-NELSON DTERMENATION OF REDUCING SUGARS Tube 1 2 3 4 5 6 7 8 9 10 D-Glucose (160 µg/mL), mL 0 0 0.125 0.125 0.25 0.25 0.375 0.375 0.5 0.5 H2O, mL 0.5 0.5 0.375 0.375 0.25 0.25 0.125 0.125 0 0 D-Galactose (160 µg/mL), mL - - - - - - - - - - D-Arabinose (160 µg/mL), mL - - - - - - - - - - Glucose-Galactose Mixture, mL - - - - - - - - - - Absorbance (710 nm) 0.049 0.051 0.138 0.151 0.271 0.26 0.382 0.395 0.454 0.444 Absorbance - blank 0 0 0.088 0.101 0.221 0.21 0.332 0.345 0.404 0.394 Final [Reducing sugar]…
- The initial velocity data shown in the table were obtained for an enzyme. Each assay at the indicated substrate concentration was initiated by adding enzyme to a final concentration of 0.01 nM. Derive Km, Vmax, kcat, and the specificity constant. [S] (mM) Velocity (x10^7) 0.10 0.96 0.125 1.12 0.167 1.35 0.250 1.66 0.50 2.22 1.0 2.63Explain how the Kirby-Bauer test works and what information it provides. Define minimum inhibitory concentration (MIC). Describe how the E-test works and what information it provides.200 ml of a 2% protein solution are available, containing an enzyme to be purified. Half of the sample is subjected to method A, consisting of fractionated precipitations, and 5 ml of final solution are obtained, with a protein concentration equal to 5 mg / ml and enzymatic activity equal to 2000 U / ml. The other half is subjected to method B, consisting of ion exchange chromatography, and a final solution of 10 ml is obtained, with a protein richness equal to 10 mg / ml, and with an enzymatic activity equal to 2000 U / ml. You want to know: a) Which of the methods has provided the purest enzyme. b) By which of the methods has the greatest amount of protein been obtained.
- Pyridoxal phosphate (PLP) is a coenzyme for the enzyme ornithine aminotransferase. The enzyme was purified from cells grow in PLP = deficient media as well as from cells grown in media that contained pyridoxal phosphate. The stability of the two different enzyme preparations was then measured by incubating the enzyme at 37°C for different lengths of time and then assaying for the amount of enzyme activity remaining. The following results were obtained. (a) Why does the amount of active enzyme decrease with the time of incubation? (b) Why does the amount of enzyme from the PLP deficient cells decline more rapidly?The enzymatic activity of PFK1 is generally measured by set- ting up a coupled enzyme assay system whereby aldolase, triose phos- phate isomerase, and glycerol-3-phosphate dehydrogenase are added to the assay mixture. For the latter enzyme, NADH is added and its change in concentration is readily monitored at 340 nm. Write the chain of reactions catalyzed by these enzymes using structural formulas, label substrates and products, and explain why the coupled en- zyme assay system leads to oxidation of NADH. While the chain of reac- tions is similar to those in glycolysis, there is a critical difference because of the dehydrogenase enzyme. Describe how this enzyme causes the chain of reactions to differ from those in glycolysis.a) Determine kcat (in units of sec-1) for a particular enzyme, given the following information: Vo = 144 mmol/min; [S] = 2 mM; Km = 0.5 mM; Enzyme Molecular weight = 40,000 mg/mmole; 8 mg of enzyme used in assay generating this data. b) In general, explain how the total enzyme concentration affects turnover number and Vmax?
- Calculate the Activity of an amylase enzyme which is diluted 1:100 times with phosphate buffer and incubated for 10 minutes at 37 degree Celsius. Given are the amount of maltose [mg] = 5.05 and the volume of enzyme used for the assay as 0.5ml.The turnover number for an enzyme is known to be 5000min-1. Given the following set of data, Substrate concentration(mM) 1, 2, 4, 6, 100, 1,000 Initial Rate(micromol/min) 167, 250, 334, 376, 498, 499 a) What is the Km of the enzyme for the substrate? (do this without using a calculator) b) What is the total amount of enzyme present in the assay?Enzyme X has a molecular weight of 48,000. It converts substrate Z into product Y. Z absorbs at 340 nm, and Y absorbs at 480 nm. A.) At what wavelength would you measure the change in absorbance to assay for enzyme X? Would the absorbance increase or decrease over time? B.)If Vmax = 60 μmol/min and you used 400 μL of a 0.1 mg/mL solution of enzyme, what is the turnover number?

