Quick overview of our lesson: Our topic is all about Second Law of Thermodynamics. Gibbs’ free energy, G is defined by G = H – TSwhere H is the enthalpy, T is the temperature (in Kelvins), and S is the entropy. In a chemical reaction,R ↔P (R are reactants and P are products) at aconstant temperature we have ∆G = ∆H – T∆S.If ∆G < 0 the reaction may proceed spontaneously tothe right.If ∆G = 0 the reaction is in equilibrium.If ∆G > 0 the reaction may proceed spontaneously to the left. please do help me with the questions on the picture PLEASE EXPLAIN ALSO PLS
Quick overview of our lesson: Our topic is all about Second Law of Thermodynamics. Gibbs’ free energy, G is defined by G = H – TSwhere H is the enthalpy, T is the temperature (in Kelvins), and S is the entropy. In a chemical reaction,R ↔P (R are reactants and P are products) at aconstant temperature we have ∆G = ∆H – T∆S.If ∆G < 0 the reaction may proceed spontaneously tothe right.If ∆G = 0 the reaction is in equilibrium.If ∆G > 0 the reaction may proceed spontaneously to the left. please do help me with the questions on the picture PLEASE EXPLAIN ALSO PLS
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter10: Entropy And The Second Law Of Thermodynamics
Section: Chapter Questions
Problem 10.33PAE: Limestone is predominantly CaCO3, which can undergo the reaction CaCO3(s)CaO(s)+CO2(g). We know from...
Related questions
Question
100%
Quick overview of our lesson:
Our topic is all about Second Law of
Gibbs’ free energy, G is defined by G = H – TSwhere H is the enthalpy, T is the temperature (in Kelvins), and S is the entropy. In a
please do help me with the questions on the picture
PLEASE EXPLAIN ALSO PLS
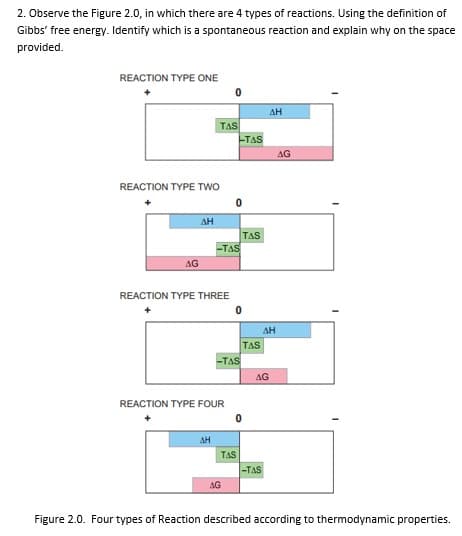
Transcribed Image Text:2. Observe the Figure 2.0, in which there are 4 types of reactions. Using the definition of
Gibbs' free energy. Identify which is a spontaneous reaction and explain why on the space
provided.
REACTION TYPE ONE
AH
TAS
TAS
AG
REACTION TYPE TWo
AH
TAS
-TAS
AG
REACTION TYPE THREE
AH
TAS
-TAS
AG
REACTION TYPE FOUR
AH
TAS
-TAS
AG
Figure 2.0. Four types of Reaction described according to thermodynamic properties.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps with 4 images

Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning