Q: Explain the attached statement is true or not ?
A: Given:
Q: it is the correct order of stability of the following radicals (more stable > less stable)?
A: Carbon forms four bonds to complete its octet. In the formation of free radical,carbon has formed…
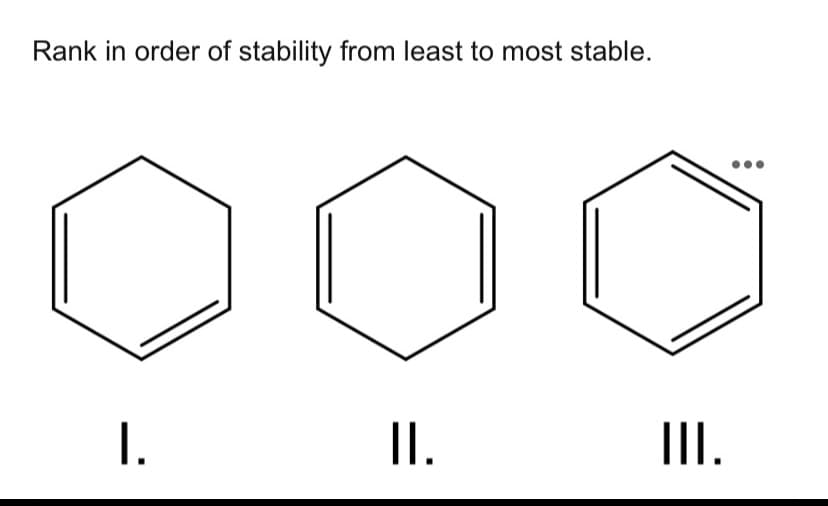
Q: Rank the alkenes shown below based on stability, should be indicated as (most stable (left) to least…
A: The correct order of the stability of alkene can be done on the basis of substitution. Higher the…
Q: Complete energy diagram for the two-step conversion of A–B + C:−→ A:− + B–C
A: SOLUTION: Step 1: The overall reaction given is as shown below: A-B + C: -→ A: - +B-C A possible…
Q: LEAST stable
A:
Q: Which product(s) will predominate if the reaction is under kinetic control?
A: According to the reaction mechanism or temperature dependence. Two types of products are formed in…
Q: Sulfonation of naphthalene, C1,Hg , results in two products. One product is kinetically favored and…
A:
Q: Rank the following in order of increasing stability (least stable to most stable): A В D
A: Interpretation- To determine the order of increasing stability of all the given alkenes ( less…
Q: Arrange the following compounds in order of stability by dragging them into the space below.
A:
Q: a) Alkene (menkovnikov) Bn (Anti - menkovnikov) Step2 b) HBP Menkounikov Br HBr Bn Antimenkovnikov…
A: Given reactions: We have to show the mechanisms of the Markonikov and anti Markonikov additions.
Q: Which alkene has (E) configuration? а. b. c. d.
A: E–Z configuration is the method of describing the absolute stereochemistry of double bonds.
Q: Place from high stability to lowest stability for 2,4 dimethyl pentane
A: The stability of any compound is inversely proportional to the steric repulsion between the groups…
Q: regiochemistry and/or configuration
A: The given blank has to be filled correct regiochemistry and/or configuration
Q: A в Which of the alkenes above is the least stable (highest in energy)? Which is the most stable…
A: More substituted alkene is more stable. Alkyl group is electron donating and it stabilize alkene
Q: a convenient method For making the bollowing Give conversions cyclopropyne a dibromocyclopropane
A: Given: cyclopropyne and 1,1-dibromocyclopropane To find: convert cyclopropyne and…
Q: next compound according to IUPAC rules?
A: While writing IUPAC name, give priority to the functional groups and list them in alphabetical…
Q: Rank the following radicals in order of decreasing stability. most stable least stable (CHдС-снсн,…
A: Free radicals are electron deficeint. the stability of tertiary radicals is more than that of…
Q: Rank the following radicals in order of decreasing stability. most stable least stable (сны,снінсн,…
A: The stability of carbon free radicals can be determined by considering the number of hypercojugative…
Q: Arrange carbocation in order of increasing stability
A: More the hyperconjugation, more is the stability of the carbocation.
Q: Rank the carbocations in the Figure below in order of decreasing stability. ["~" = approximately…
A:
Q: Rank the attached radicals in order of increasing stability
A: Free radicals refer to the type of reactive intermediate in which a homolytic cleavage of bond takes…
Q: Rank the following alkenes in order of increasing stability.
A:
Q: Which one is more stable S2-, S4-, S6-?
A: The given ions of Sulphur are S2-, S4-, and S6-.
Q: Match each reaction with its correct energy diagram. он он H2SO, H2SO, HCI он A) B) C) ..... DIAGRAM…
A:
Q: b. С. + + + a.
A: The reactive intermediate species in which carbon atom forms only three bonds and contains a…
Q: Arrange the following radicals in order of INCREASING stability. II IV
A:
Q: ,including all stereoisomers of the product.
A: This reaction is Dieles Alder reaction because it's have diene and dienophile Compound in the…
Q: Explain why compound A is much more stable than compound B.
A: The compare the stbaility of the two compounds above we check whether the compound is aromatic or…
Q: Rank the following alkenes in order of increasing stability.
A: Increasing order of stability of alkenes has to be given.
Q: Rank the following carbocations in terms of stability from lowest to highest. to A B C D
A:
Q: A) Which one is more stable? B) Which one is more stable?
A: For comparing the stability of cyclohexane, firstly we have to convert the cyclic form into chair…
Q: What is the % of H2O if 102g H2) is produced from 14.6g H2 and excess O2?
A: Given Mass of H2O = 102 gram Mass of H2 = 14.6 gram Percentage yield =?
Q: Rank the following alkenes in order of increasing stability (least to most stable)
A:
Q: Rank the following radicals in order of increasing stability.
A: Since the stability of free radicals depends on two things 1) resonance => more is the resonance…
Q: Br or Br Br C or CI || CI or CI ||
A:
Q: What are the following alkenes, E or Z? if neither label as such as with both
A: Welcome to bartleby ! We have to tell whether these alkenes are E or Z or neither
Q: For the pairs of compounds in figure, which is more reactive in an SN2 reaction?
A: Since in SN2 reaction, the nucleophile attacks on the carbon with Br and removes it. So the attack…
Q: А. В. С.
A:
Q: Which one is the most stable?
A: The more substituents the alkenes have, the more stable they are. Thus, a tetra substituted alkene…
Q: List them in order of stability from higher to lower
A: The order for the stability of the carbocation - Conjugated carbocation > tertiary carbocation…
Q: is this z or E?? If it is E. please in details explain why cycloproyl is greater than OH group??
A:
Q: Rank the followings carbocations in terms of increasing stability (least to most)
A: Stability of carbo cation depends upon the type of structure of carbocation , primary are least…
Q: Explain why1-bromobicyclo [2.2.2] octane is not likely to undergo an Sn1 or an SN2 substitution…
A:
Q: Rank the following radicals in order of increasing stability.
A: The stability of free radical is governed by various factors such as: Allylic / benzylic radical 3°…
Q: Using the principles described in this course, explain (using sentences and drawings) why…
A: The more stable the carbocation ,the lower the activation energy for reaching that inter mediate…
Q: Which is more stable?
A:
Q: Arrange the following in order of increasing stability A в
A: Stability of diens depends upon conjugation and no. of alpha hydrogens.
Q: Rank the following from the most to the least stable.
A: Stability of alkenes can be explained using hyperconjugation effect.
Q: Rank the following alkenes in order of increasing stability:
A: To solve this problem we have to know about the stability of alkene with substitution .
Q: most stable?
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images


