Q: What is the major product for the following reaction (A) (B) CH3CH₂CH₂CH₂Cl AlCl3 (C) (D)
A: The given reaction is Friedel craft alkylation in which aromatic proton replace with alkyl group.…
Q: CH; +CH CH; I II III IV
A: We have to rank the following given carbocation in order of increasing stability as follows in step…
Q: 1. Rank the following alkenes in order of increased stability. 2 5
A:
Q: 6. Rank the alkenes in order of increasing stability. (1 = least stable, 4 = most stable) |
A: Stability of alkene ∝ Number of hyper conjugative structure ∝ Number…
Q: Draw an Energy diagram for an E1 reaction: (CH3)3CI + H2O ⇢ (CH3)2C=CH2+ H3O+ + I−
A: E1 is the unimolecular elimination reaction, the removal of HX from the substrate leads to the…
Q: 4. (2 pts) Circle the most stable carbocation and cross out the least stable carbocation among the…
A:
Q: Rank the following alkenes in terms of increasing stability. (A) (B) (C) OC<A<B OA< C < B OA < B < C…
A: Stability of the alkenes increases on increasing the substitution which means highly substituted…
Q: Rank the following carbocations in order of increasing stability. + CH,CH,CH2CH2CH=CH b.…
A: To rank the given carbocation in the order of increasing stability.
Q: Draw an energy diagram for an E2 reaction: (CH3)3CBr + −OH →(CH3)2C=CH2 + H2O + Br−
A:
Q: Rank the following carbocations in order of increasing stability. CH2 CH3 CH3
A: The carbocation is basically a trivalent carbon whose hybridization is sp2. The higher the…
Q: Rank the following alkenes in order of increasing stability: CH,=C(CH3)CH,CH3, CH2=CHCH(CH3)2, and…
A: Stability of alkene is directly proportional to number of hyperconjugation structures and number of…
Q: stereoisomers. NaOEt 1. Br NaOH Br 2. NaOtBu 3. A Меон 4. Br 2. Which reaction will proceed faster A…
A: 1) Elimination 2) SN2 reaction 3) Elimination 4) SN1 reavtion Reavtion B is faster than A due 1,3…
Q: 2. Arrange these carbocations in order of increasing stability. CH2 CH3 -CH3 C. b. a.
A: Stability of carbocation can be compared using hyperconjugation effect. Carbocations with having…
Q: a. Rank in order of S2 reactivity (1= most reactive) b. Rank in order of leaving group ability (1=…
A:
Q: Which of the following carbocations is the most stable? (A) (B) (C) (D)
A:
Q: 1. What will be the major product for the following reaction according to the Markovnikov's rule?…
A: Note : As per our guidelines ,we are supposed to answer first question , Please resubmit the other…
Q: Draw an energy diagram for the following SN2 reaction. Label the axes, the starting materials, and…
A: The reaction is assumed to be exothermic and so, the energy of the reactants is more than the energy…
Q: Which of the following carbocations (A or B) is more stable? Explain your choice.
A: In the given two carbocations, the stability is decided by the electronegativity factor of O and…
Q: Looking at the reaction below. Is A or B expected to be the major product and why? HO H H3C H3C. OH…
A: Base catalyzed ring opening of epoxide(oxiranes) takes place from the sterically less crowded side…
Q: What is the major product formed in the following reaction? A A В в C C D D
A: Elimination reaction is a type of reaction in which substituents are removed from the molecule in…
Q: CH3 *CH cH2 || C A. B
A: Ans is C
Q: А. В. С. D.
A:
Q: 8A. Draw a reasonable mechanism for the following reaction, with clear indication of…
A:
Q: Classify the following carbocations as 1o, 2o, or 3o and b) rank the following carbocations in order…
A:
Q: 9. Draw the four allylic carbocation intermediates of the following reaction and rank their relative…
A: Since your question has multiple parts, we will answer the first part for you. If you want residual…
Q: carbocations in decreasing order of stability:
A: Stability of carbocation increases by electron releasing groups and decreases by electron…
Q: Rank the following alkenes in order of increasing stability (least to most stable) I II III IV
A: A question based on alkene, which is to be accomplished.
Q: Rank the following carbocations in order of decreasing stability (most → least): CH₂CHCH₂…
A:
Q: 4. Supply the missing reagents/conditions to make the following transformations. но OTf CN
A: In the first step the OH is basically replaced by OTf. OTf is basically the triflate functional…
Q: 7. Which of the following carbocations is most stable (circle your answer)
A: In this question, we will Identify which Compound have most stable. You can see details explanation,…
Q: Rank the following carbocations in decreasing stability (most stable to least stable). [Note: your…
A: Carbocations are carbon ions which have a positive charge on it and its hybridization is sp2.
Q: 3. Which of the following is most likely to rearrange via 1,2-hydride shift to form a more stable…
A:
Q: ne major product c
A:
Q: What are the reagents A and B in the scheme below? CH, CH,CH,CH,C-CH, ČI в A H,C CH H,C CH,
A:
Q: Please Prouide the missing Feagents, NH2 Please Prouide the missing reagents. OH Br
A:
Q: 2. Draw all significant resonance contributors for the following carbocations. CH2 CH3 H2C: CH
A: Resonance contributors are those resonance structures which are formed by the shifting of bonding…
Q: Q4. The major product(s) of the following E2 reaction is/are: CH CH,ONa Br CH3 E2
A: Biomolecular elimination (E2) reaction:E2 reaction involves a one-step mechanism in which…
Q: Rank the following carbocations in terms of their energy. A Highest Energy Lowest Energy
A:
Q: 7. Rank the following carbocations in order of stability. (1-most stable). ++ 0
A: The reactive intermediate species in which carbon atom forms only three bonds and contains a…
Q: Rank the following carbocations from least to most stable: 1 2 O1<3<2 O 2<1< 3 O1<2<3 O 2<3 <1
A: Carbocation stability depends upon the number of alkyl groups attached to it. The alkyl groups have…
Q: . Rank the following enols in order of increasing stability: HO- OH HO- II III %3D
A: The compounds given are,
Q: Which of the following is the most effective way to stabilize carbocations? a. Zaitzev's rule O b.…
A: Carbocation stablized by electron donating group or resonance when positive charge is in conjugation…
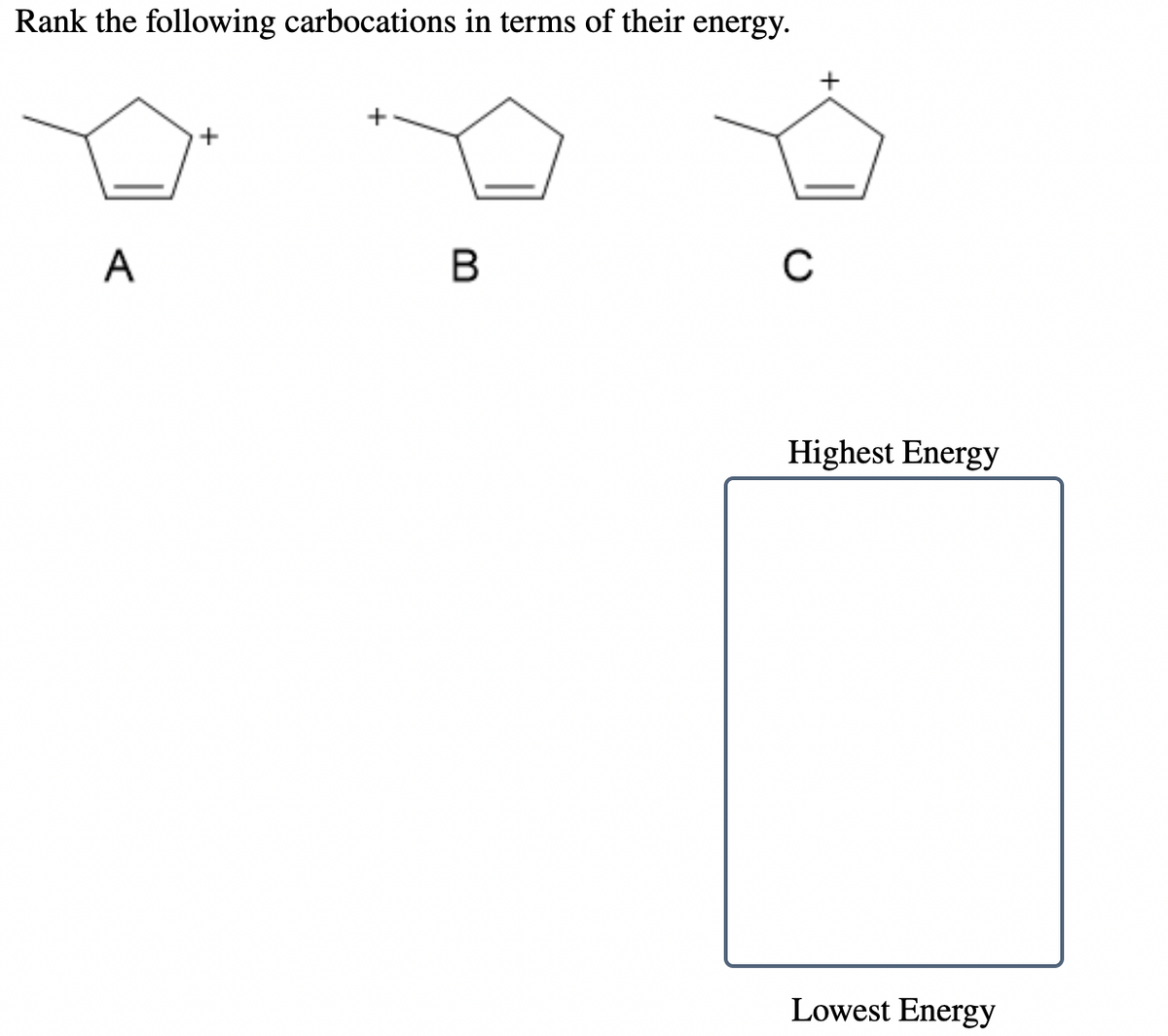
Q: Rank the following carbocations in terms of their energy. + A В Highest Energy Lowest Energy
A:
Q: 6. Draw the reagent that converts the alkene below to the epoxide (oxirane) c=CH2 CH2 H,CH,C°…
A:
Q: OH OH OH II III
A:
Q: Which compound is more reactive in an SN1 reaction? In each case, you can assume that both alkyl…
A: Since in SN1 reaction, the stability of carbocation formed decides the rate of the reaction. So if…
Q: Identify the least stable and the most stable of the following carbocations B A
A: Carbocation is species in which carbon carries positive charge.
Q: 7. For each reaction, circle the correct product. Br N2OCH3 or NaOCH3 or E2 E2
A: A species with a larger size can easily accommodate negative charge. In other words, in a large size…
Q: Rank the following carbocations from most stable to least stable:
A: Since more is the resonance of +ve charge on Carbon, more it will be delocalised. Hence more will be…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Draw all resonance structures for the carbocation formed by ortho attack of the electrophile +NO2 on each starting material. Label any resonance structures that are especially stable or unstable.Draw all resonance structures for the carbocation formed by ortho attack of +NO2 on each starting material. Label any resonance structures that are especially stable or unstable with reasonsCircle the electrophiles in the following reagents: AlCl3 NO3– NO2+ H3O+ OH-
- Rank the following radicals in order of increasing stability.Which alkene in each pair is more stable?1. Which among these would be the most stable carbocation?a. Allylicb. Vinylicc. Primaryd. Secondary2. What is the primary reason for the stability of tertiary carbocations?a. Free rotationb. Resonancec. Hyperconjugation

